Study of Immunosuppressive Activity of a Synthetic Decapeptide Corresponding to an ACTH-Like Sequence of Human Immunoglobulin G1
E. V. Navolotskaya1*, T. A. Zargarova1, T. N. Lepikhova1, V. I. Turobov1, R. I. Nurieva1, N. V. Malkova1, V. M. Lipkin1, and V. P. Zav'yalov2
1Branch of Shemyakin and Ovchinnikov Institute of Bioorganic Chemistry, Russian Academy of Sciences, Pushchino, Moscow Region, 142292 Russia; fax: (0967) 79-0527; E-mail: navolots@fibkh.serpukhov.su2Institute of Engineering Immunology, Lyubuchany, Moscow Region, 142380 Russia; fax: (095) 546-1574
* To whom correspondence should be addressed.
Received February 11, 1999
The synthetic ACTH-like decapeptide H-Val-Lys-Lys-Pro-Gly- Ser-Ser-Val-Lys-Val-OH, corresponding to amino acid residues 11-20 of the variable part of the human IgG1 heavy chain (referred to as immunocortin) was found to have an immunosuppressive effect on cells in vitro: it inhibits blast transformation of mouse thymocytes and reduces spontaneous motility of mouse peritoneal macrophages as well as their bactericidal activity against the virulent bacterial strain Salmonella typhimurium 415. Tritium-labeled immunocortin binds with high affinity to ACTH receptors on thymocytes and macrophages (Kd 2.1 and 2.5 nM, respectively) and activates adenylate cyclase in these cells. Thus, the interaction of immunocortin with the target cell includes the following main steps: binding to the receptor, activation of adenylate cyclase, and elevation of the intracellular content of cAMP.
KEY WORDS: adrenocorticotropic hormone (ACTH), immunoglobulin G (IgG), peptides, receptors, adenylate cyclase, thymocytes, peritoneal macrophages, immune system
In the early 1980s immobilized antibodies to adrenocorticotropic hormone (ACTH) and beta-endorphin were studied as possible affinity adsorbents for isolation of the hormones from human placenta extract [1]. A 50-kD polypeptide was isolated, and it was found to be a heavy (H) IgG chain. Further study led to the discovery of sequences homologous to ACTH and beta-endorphin in the H-chain. The fragment 364-377 of the constant part of the IgG H-chain (SLTCLVKGFYPSD) was homologous (40%) to a beta-endorphin antigenic determinant (KSQTPLVTLFKNALIKN), and fragment 9-22 of the variable part of the IgG1 H-chain (AEVKKPCSSVKVSC) was homologous (36%) to ACTH amino acid sequence 11-24 (KPVGKKRRPVKVYP). An artificial peptide (14 amino acid residues long) corresponding to the beta-endorphin-like sequence of IgG was synthesized and found to interact with opioid receptors on rat brain cells [2].
We synthesized the ACTH-like decapeptide (H-VKKPGSSVKV-OH) corresponding to amino acid sequence 11-20 of the variable part of the human IgG1 H-chain (referred to as immunocortin). We showed that it binds with high affinity to ACTH receptors and activates adenylate cyclase of murine brain synaptic membranes [3].
The purpose of the present study was to investigate the effect of immunocortin on the activity of immunocompetent cells in vitro.
MATERIALS AND METHODS
Inbred CBA mice (16-18 g) and randomly bred white rats (200-250 g) were used for experiments.
The chemicals used in this study were: concanavalin A (Con A) from Pharmacia (Sweden); L-glutamine and Hepes from Flow (USA); penicillin and streptomycin from Gibco (USA); agarose, saccharose, BSA, EDTA, EGTA, Tris, and phenylmethylsulfonyl fluoride (PMSF) from Serva (Germany); scintillator Unisolv 100 and [methyl-3H]thymidine (76 Ci/mmole) from Amersham (England); Al2O3 according to Brockman II from Reanal (Hungary). Other chemicals were chemically pure or especially pure grade.
199 medium (Central Scientific Research Institute of Vaccines and Sera, Moscow, Russia) and RPMI-1640 (Serva, Germany) were used for cell cultivation.
Immunocortin and ACTH fragments were synthesized as described previously [3] using activated esters. The peptides were purified by HPLC on a Zorbax ODS column (4 × 150 mm, 5 µm particle size) with a linear gradient of aqueous acetonitrile (95%) in 0.2% trifluoroacetic acid (10-25%, 20 min, 1 ml/min flow rate). The content of the main substance was assessed by measuring the absorbance at 220 nm. The molecular masses of the peptides were determined by mass spectrometry.
[3H]Immunocortin (9.6 Ci/mmole) was obtained at the Institute of Molecular Genetics of the Russian Academy of Sciences.
The virulent strain of Salmonella typhimurium 415with typical morphological and functional properties was used in this study. The LD50 was approximately 100 bacterial cells injected intraperitoneally into white mice. S. typhimurium was grown in Hottinger's broth for 4-6 h at 37°C, then transferred to beef-extract agar and incubated at 37°C for 18 h.
The effect of immunocortin on mouse thymocyte blast transformation in vitro was assayed as in [4]. Each well of a 96-well microtiter plate contained 100 µl thymocyte suspension isolated from 3-4-week-old mice (5·105 cells/ml), 50 µl Con A (2 µg/ml), and 100 µl immunocortin solution of known concentration. The cultures in 3-4 replicates were used for each experimental point. The following samples served as controls: without Con A (a); with peptide, without Con A (b); with Con A, without peptide (c). The reaction mixture was incubated for 72 h at 37°C in 5% CO2. After 60 h each well was pulsed with 1 µCi [methyl-3H]thymidine. When incubation was terminated the reaction mixture from each well was filtered separately and rinsed with 100-fold excess of physiological solution. Radioactivity on filters was measured in a Minibeta 1211 liquid scintillation counter (LKB, Sweden). The reaction medium was RPMI 1640 supplemented with glutamine (2 mM), streptomycin and penicillin (100 µg/ml each), Hepes (20 mM), and heat-inactivated (56°C, 30 min) fetal calf serum (5%).
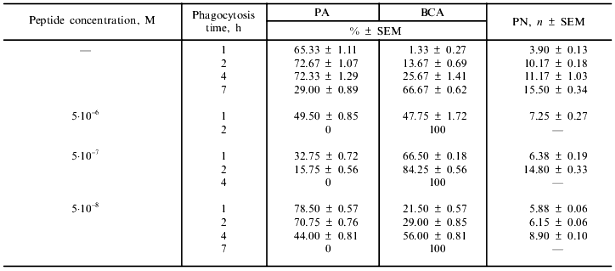
Mouse peritoneal macrophages were isolated and cultivated as in [5]. Phagocytic activity of macrophages was estimated according to our published procedure [6]. Macrophage monolayers on cover glasses were cultivated in sterile test tubes in 199 medium supplemented with streptomycin and penicillin (100 µg/ml each) and inactivated fetal calf serum (5%) at 37°C. After 24 h, macrophages were infected with 199 medium supplemented with serum and S. typhimurium 415 (108 bacterial cells/ml final concentration). After 2 h the contact between the bacteria and macrophages was interrupted by replacing the infection medium with fresh medium supplemented with antibiotics. To prevent the recapture by other phagocytes of bacteria released from broken phagocytes, the culture medium was replaced with fresh medium every 2 h. The macrophages on cover glasses (in triplicate for every time point, at 1, 2, 4, 6, 7, and 24 h) were fixed in methanol for 7 min. Then the preparations were stained with 0.1% aqueous azure II eosinate for 5 min. Cells (300 per cover glass) were examined using a light microscope (magnification ×1350) and analyzed for the following parameters: phagocytic activity (PA), the percentage of macrophages participating in phagocytosis; bacterial cytocidal activity (BCA), the percentage of phagocytes destroyed by intracellular bacteria; and phagocytic number (PN), the average number of bacteria per macrophage.
Macrophage motility was estimated by a micromethod for determination of spontaneous migration of mononuclear phagocyte lineage cells from an agarose drop [7]. Agarose solution (0.4%) was prepared as follows: 20 mg agarose was added to 3 ml 199 medium, melted in a boiling water bath, and cooled to 60°C. The solution was then mixed with 1.5 ml heat-inactivated fetal calf serum and 0.5 ml 199 medium supplemented with all necessary ingredients so that the resulting agarose solution contained 10 mM Hepes and 50 µg/ml each of streptomycin and penicillin. A macrophage suspension in 199 medium (109 cells/ml) was added to an equal volume of agarose and the mixture was heated to 37°C. The cell suspensions in agarose were put in flat-bottom plates (1 µl per well) and incubated at 4°C for 15 min. When the agarose congealed, 200 µl 199 medium supplemented with 10 mM Hepes, streptomycin and penicillin (50 µg/ml each), fetal serum (15%), and immunocortin (10-9-10-3 M) was added to each well. The plates were incubated for 18-20 h at 37°C in 5% CO2. Macrophage motility was assessed microscopically as follows: the diameter of the cell migration zone was determined using a micrometer and migration index (MI) was estimated from the equation:
MI = (1 - migration zone diameter in experiment/migration zone diameter in control) × 100%.
Binding of [3H]immunocortin to mouse thymocytes and peritoneal macrophages was assayed in 199 medium supplemented with Hepes (25 mM), NaN3 (20 mM), and PMSF (0.6 mg/liter) as follows: 100 µl labeled peptide (concentration range 10-10-10-7 M) and 100 µl medium (total binding) or unlabeled peptide (nonspecific binding) were put together with 800 µl cell suspension (107 cells/ml) into siliconized test tubes and incubated at 4°C for 1 h. The reaction mixture was then filtered through fiberglass filters GF/B (Whatman, England) to separate the labeled cell-bound peptide. Filters were rinsed three times with 5-ml volumes of ice-cold 25 mM Hepes, pH 7.4. Radioactivity of filters was counted using the Minibeta 1211 liquid scintillation counter. Nonspecific binding was measured in the presence of 0.1 mM unlabeled immunocortin.
Binding of [3H]immunocortin to plasma membranes isolated from thymocytes and macrophages was carried out in 25 mM Hepes, pH 7.4, supplemented with PMSF (0.6 mg/liter) at 4°C for 40 min. The protein concentration in the reaction mixture (1 ml) was 0.5 mg/ml. Nonspecific binding of [3H]immunocortin was assessed in the presence of 0.1 mM unlabeled peptide. Further procedures--filtration, counting of radioactivity, and data processing--were carried out as described above. Plasma membranes of freshly isolated thymocytes and macrophages were obtained as described in [8]. Protein concentration was determined by the Lowry method [9] using BSA as the standard.
To determine the parameters of labeled immunocortin specific binding to the receptors, i.e., the equilibrium dissociation constant Kd, binding capacity n (number of receptors per cell), and Bmax (maximum binding capacity per mg membrane protein), the profiles of the ratio between bound (B) and free (F) labeled immunocortin molar concentrations as a function of bound labeled peptide (B) were plotted. Binding capacity (n) was estimated from the equation:
n = (R0 · Avogadro's number)/cell number per liter,
where R0 is the molar concentration of receptors [10].
The ability of ACTH (13-24) to compete with immunocortin for the receptors was assessed by plotting curves of inhibition of 10 nM [3H]immunocortin-specific binding to thymocytes and macrophages by increasing concentrations of unlabeled ACTH (13-24), somatostatin, and ACTH (4-10) (concentration range 10-12-10-4 M). Receptor binding assay and all subsequent procedures were carried out as described above. Inhibition constant Ki was calculated from the equation [11]:
Ki = IC50/(1 + [L]/Kd),
where IC50 value was determined from the curve, [L] is the molar concentration of labeled ligand, and Kd is equilibrium dissociation constant of labeled ligand--receptor complex.
Adenylate cyclase activity was determined as in [12] using [alpha-32P]ATP. The reaction mixture contained: 40 mM Tris-HCl, pH 7.4, 50 µM ATP, 4 mM cAMP, 12 mM phosphoenolpyruvate, and 2 µg/ml pyruvate kinase. [alpha-32P]ATP (200,000-500,000 cpm) in 50 µl medium and 50 µl test substance or an equal volume of buffer (control) were put into conical siliconized glass test tubes residing in an ice-cold water bath. The test tubes were transferred to a thermostat and incubated at 34°C for 45 min. The reaction was terminated by adding 100 µl 0.5 N HCl. The test tubes were then put into a boiling water bath for 15 min, then transferred to an ice-cold bath and supplemented with 100 µl 1.5 M imidazole. The content of each test tube was passed through a separate column filled with 1 cm3 Al2O3 according to Brockman II and rinsed with 5 ml distilled water. The enzyme activity was determined as the decrease in substrate content expressed as nanomoles cAMP generated per milligram test substance protein per 10 min.
For determination of cAMP content in macrophages, their TCA (5%) extracts were used [13]. The cAMP content of extracts was determined using the Cyclic-AMP-Assay-Kit (Amersham) according to the manufacturer's instructions.
RESULTS
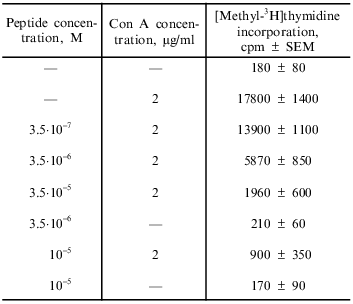
Effect of immunocortin on blast transformation of mouse thymocytes in vitro. The results obtained in this study indicate that immunocortin at doses ranging from 3.5·10-7 to 10-5 M inhibits Con A-induced blast transformation of thymocytes (Table 1). The suppressive effect of the peptide was not associated with its cytotoxicity: cell viability according to the trypan blue exclusion test remained at the 95-97% level.
Table 1. Effect of immunocortin on blast
transformation of mouse thymocytes in vitro
Effect of immunocortin on the activity of mouse peritoneal macrophages in vitro. Immunocortin inhibited the bactericidal activity of peritoneal macrophages in relation to S. typhimurium 415. The values of the main characteristics (PA, PN, BCA) of S. typhimurium phagocytosis in the absence (control) or in the presence of various concentrations of the peptide are given in Table 2. It is evident that in the presence of 5·10-6 M immunocortin phagocytes are completely destroyed by intracellular bacteria within 2 h. When the peptide concentration in the medium was 5·10-7 M a macrophage monolayer was destroyed within 4 h, and at the concentration of 5·10-8 M this process was completed within 7 h. At the same time, BCA in control did not exceed 70% within 7 h. ACTH (13-24) studied in parallel had no effect on phagocytosis: the main characteristics of the process did not differ from control values at peptide doses ranging from 5·10-10 to 5·10-5 M. However, ACTH (13-24) at 10-5 M (but not 5·10-8 M), when added in combination with immunocortin (5·10-7 M), canceled completely the phagocytosis-suppressing effect of the latter.
Table 2. Effect of immunocortin on the
interaction of mouse peritoneal macrophages with the virulent bacterial
strain S. typhimurium 415
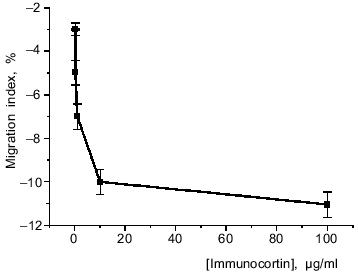
Study of the effect of immunocortin on mouse peritoneal macrophage spontaneous migration from an agarose drop revealed that increasing its concentration in the medium decreased the migration index (Fig. 1). This suggests that immunocortin inhibits macrophage motility.
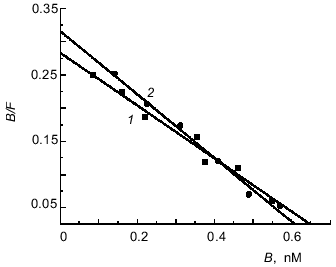
[3H]Immunocortin binding to mouse thymocytes and macrophages. Study of the interaction of [3H]immunocortin with mouse thymocytes and peritoneal macrophages revealed that the labeled peptide can bind specifically to these cells. Figure 2 shows Scatchard plots characteristic of specific binding of [3H]immunocortin to macrophages (plot 1) and thymocytes (plot 2). Both plots are linear, this suggesting that macrophages and thymocytes each possess only one receptor type (Kd 2.5·10-9 and 2.1·10-9 M, respectively).Fig. 1. Effect of immunocortin on spontaneous migration of mouse peritoneal macrophages.
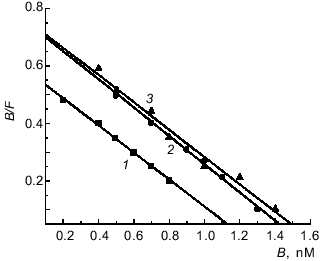
Similar Kd values were determined by Scatchard analysis of [3H]immunocortin specific binding to plasma membranes isolated from macrophages and thymocytes (Fig. 3). The Kd values of labeled immunocortin--receptor complex were 2.1·10-9 M for plasma membranes of rat (plot 1) and mouse (plot 3) thymocytes and 2.0·10-9 M for those of mouse macrophages (plot 2).Fig. 2. Scatchard analysis of[3H]immunocortin specific binding to mouse peritoneal macrophages (1) and thymocytes (2). B and F are molar concentrations of bound and free labeled peptide, respectively.
Table 3 presents data on the inhibition by unlabeled ACTH (13-24), somatostatin, and ACTH (4-10) of the specific binding of 10 nM [3H]immunocortin to mouse thymocytes and macrophages. It is evident that only ACTH (13-24) was able to inhibit the specific binding of labeled immunocortin to both thymocytes (Ki = 1.3 nM) and macrophages (Ki = 1.9 nM).Fig. 3. Scatchard analysis of[3H]immunocortin specific binding to rat thymocyte plasma membranes (1), mouse peritoneal macrophage plasma membranes (2), and mouse thymocyte plasma membranes (3). B and F are molar concentrations of bound and free labeled peptide, respectively.
Table 3. Kinetic characteristics of
inhibition by unlabeled ACTH (13-24), somatostatin, and ACTH (4-10) of
the specific binding of 10 nM [3H]immunocortin to
mouse thymocytes

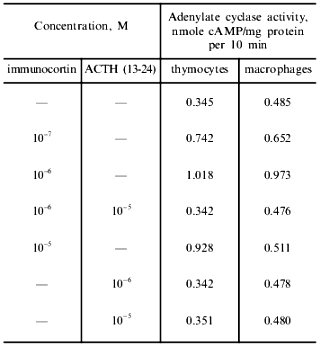
Effect of immunocortin and ACTH (13-24) on adenylate cyclase activity and intracellular cAMP content. The results presented in Table 4 indicate that immunocortin at concentrations in the range 10-7-10-5 M activated the adenylate cyclase of plasma membranes of mouse thymocytes and peritoneal macrophages. ACTH (13-24) studied in parallel had no effect on the enzyme activity, but in both cases blocked the activating effect of immunocortin in the concentration ratio immunocortin/ACTH (13-24) 1:10.
Table 4. Effect of immunocortin and ACTH
(13-24) on adenylate cyclase activity of mouse thymocyte and peritoneal
macrophage plasma membranes
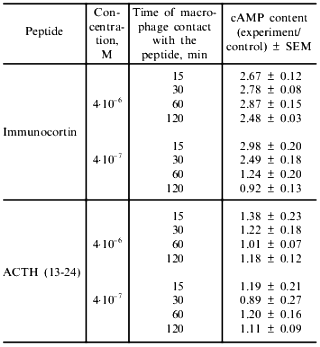
In the presence of immunocortin (4·10-7-4·10-6 M) an increase in intracellular cAMP content of macrophages was observed. The results shown in Table 5 indicate that within 15 min after the peptide (4·10-7 M) made contact with macrophages, their intracellular cAMP concentration increased approximately threefold, and 2 h later it returned to the control level. ACTH (13-24) examined in parallel did not significantly effect this parameter (Table 5).
Table 5. Effect of immunocortin and ACTH
(13-24) on intracellular cAMP content of mouse peritoneal
macrophages
DISCUSSION
The system of immune regulation together with the endocrine and central nervous systems form a unified network for bioregulation in animals. At present a growing body of evidence favors the hypothesis that along with endocrine hormone and tissue hormone (kinin) bioregulation systems, there is a third system based on cellular hormones (tetins) which are oligopeptides--the products of limited proteolysis of protein in situ. Immunoglobulins, interferons, interleukins, growth factors, and other biologically active proteins are the precursors of tetins. The following compounds are tetin precursors: tuftsin (TKPR), peptides of the rigin family (GQPR, GRPK, GKPR) and eskarin family (SKAK, SKAR, SKTK), immunopoietins (RPDVY, EPWEY, RPEVH), the C-terminal fragments of immunoglobulins (RAVSVNPGK, KSLSLSPGK), tyrosine-containing pentapeptide fragments of interferons (RITLY family), and many other (usually polar) fragments of immunoactive proteins [14].
Immunocortin can also be assigned to cellular hormone group. The results of the present study show that immunocortin possesses prominent immunosuppressive properties, manifesting themselves by inhibition of mitogen-induced thymocyte proliferation, decline in spontaneous motility of mouse peritoneal macrophages, and reduction of their bactericidal activity in relation to the virulent strain S. typhimurium 415. We found previously that immunocortin exhibits neurotropic activity and the ability for high-affinity binding to ACTH receptors on mouse brain synaptic membranes (the Ki of [125I]ACTH (13-24) specific binding was 0.9 nM) [3, 6]. Thus, immunocortin possesses both immunosuppressive and neurotropic activities. Such "versatility" may result from the same or very similar peptide action mechanism in the immune and the central nervous systems. The results presented in this paper support this idea.
In parallel with immunocortin, the synthetic ACTH (13-24) peptide (H-Val-Gly-Lys-Lys-Arg-Arg-Pro-Val-Lys-Val-OH), corresponding to the ACTH “address” segment responsible for hormone--receptor interaction [15], was studied. The results indicate that this peptide is capable of high-affinity binding to immunocompetent cell receptors (Table 3), but it is not able to activate adenylate cyclase of these cells and to modify their activity. ACTH (13-24) was also found to bind with high affinity to brain synaptic membranes (Kd of [125I]ACTH (13-24)--receptor complex was 2.2 nM) without modifying their adenylate cyclase activity [3]. In the present study we have shown that ACTH (13-24) competes with [3H]immunocortin for binding to the receptor sites on thymocytes and macrophages (Ki 1.3 and 1.9 nM, respectively) (Table 3). It is notable that the kinetic characteristics of the interaction of immunocortin and ACTH (13-24) with receptors on thymocytes, macrophages, and synaptic membranes are similar to those of ACTH binding to receptors on mouse splenocyte plasma membranes [16] as well as to cloned human [17] and mouse [18] receptors.
Thus, kinetic analysis of the competition between immunocortin and the ACTH “address” fragment, ACTH (13-24), has revealed that the receptors on immunocompetent cells and synaptic membranes to which immunocortin binds are the receptors for ACTH.
It is known that binding of ACTH to the receptor results in the activation of adenylate cyclase [19, 20]. The results presented in this paper show that immunocortin activates adenylate cyclase of thymocyte and macrophage plasma membranes with a consequent increase in cAMP content of these cells (Tables 4 and 5).
Thus, the interaction of immunocortin with mouse thymocytes, macrophages, and brain synaptic membranes includes the following main steps: binding to the ACTH receptor, activation of adenylate cyclase, elevation of the intracellular cAMP content. This is followed by the standard reaction cascade resulting in a biological response of the target cell.
Immunocortin has not been investigated previously. We were the first to describe its properties and mechanism of action. At present we have no information about the formation and metabolism of the peptide, but taking into account that processing of immunoglobulins in antigen--antibody complexes usually happens in phagocytes, it is quite probable that similar to other immunoactive peptides of immunoglobulin origin [14], immunocortin can also be formed in macrophages.
It is well known that the most effective processing of immunoglobulin Fc subunits occurs in the acid environment inside phagolysosomes [21, 22]. Peptides formed during catalysis with acid hydrolases can enter the milieu surrounding a zone of inflammation as the result of secretion or phagocyte disruption, and this may serve as a kind of “SOS” signal that recruits immune cells for antigen elimination. Fab-fragments under acid conditions have a structure that is more resistant to proteolysis [23]. However, at pH values close to neutral the cooperativity of their variable parts (VH) diminishes, and they are processed by neutral proteases. As this takes place, VH1 domains can contribute to the formation of the peptides with ACTH-like sequences.
Because the cleavage of IgG F(ab)2 fragments in phagolysosomes is usually insignificant and the VH1 subgroup comprises only a part of the total antibodies, immunocortin can reach biologically active concentrations only at reasonably high antibody titers, those near the peak of the immune response. Based on this, it seems likely that the role of the peptide is to provide a feedback signal for regulation of the immune response.
This work was supported by grants of the Russian Foundation for Basic Research (No. 97-04-49227) and the International Science and Technology Center (project No. 91).
REFERENCES
1.Julliard, J. H., Shibasaki, T., Ling, N., and
Guillamin, R. (1980) Science,208, 183-185.
2.Houck, J. C., Kimball, C., Chang, C., Pedigo, N.
W., and Yamamura, H. I. (1980) Science, 207, 78-79.
3.Mitin, Y. V., Navolotskaya, E. V., Vasilenko, R.
N., Abramov, V. M., and Zav'yalov, V. P. (1993) Int. J. Peptide
Protein Res., 41, 517-521.
4.Zav'yalov, V. P., Navolotskaya, E. V., Abramov, V.
M., Galaktionov, V. G., Isaev, I. S., Kaurov, O. A., Kozhich, A. T.,
Maiorov, V. A., Prusakov, A. N., Vasilenko, R. N., and Volodina, E. Yu.
(1991) FEBS Lett., 278, 187-189.
5.Uchitel, I. Ya. (1978) Macrophages in Immunity
[in Russian], Meditsina, Moscow, pp. 168-180.
6.Navolotskaya, E. V. (1994) Structure-Functional
Studies of Human alpha2-Interferon, Interleukin-2,
and Immunoglobulin G Using Synthetic Peptides: Doctoral
dissertation [in Russian], Institute of Immunology, Moscow.
7.Fahlbusch, B., and Dornberger, G. (1979) Acta
Biol. Med. Germ., 38, 1453-1460.
8.Naldini, L., Cirillo, D., Moody, T. W., Comoglio,
P. M., Schlessinger, J., and Kris, R. (1990) Biochemistry,
29, 5153-5160.
9.Lowry, O. H., Rosebrough, N. J., Farr, A. L., and
Randall, R. J. (1951) J. Biol. Chem., 193,
265-275.
10.Chang, K.-J., Jacobs, S., and Cuatrecasas, P.
(1975) Biochim. Biophys. Acta, 406, 294-303.
11.Chang, Y. C., and Prusoff, W. H. (1973)
Biochem. Pharmacol., 22, 3099-3108.
12.Saltarelli, D., Fisher, S., and Gacon, G. (1985)
Biochem. Biophys. Res. Commun., 127, 318-325.
13.Podoprigora, G. I., and Abakumov, O. Yu. (1976)
Byul. Eksp. Biol. Med., 82, 953-956.
14.Chipens, G. I., Veretennikiva, N. I., Vegner, R.
E., Gnilomedova, L. E., and Rosental, G. F. (1990) Structural Bases
for Peptide and Protein Immunomodulator Action [in Russian],
Znanie, Riga.
15.Kapas, S., Canmas, F. M., Hinson, J. P., and
Clark, A. J. (1996) Endocrinology, 137, 3291-3294.
16.Johnson, H. M., Smith, E. M., Torres, E. A., and
Blalock, J. E. (1982) Proc. Natl. Acad. Sci. USA,
79, 4171-4174.
17.Weber, A., Kapas, S., Hinson, J., Grant, D. B.,
Grossman, A., and Clark, A. J. (1993) Biochem. Biophys. Res.
Commun., 197, 172-178.
18.Canmas, F. M., Kapas, S., Barker, S., and Clark,
A. J. (1995) Biochem. Biophys. Res. Commun., 212,
912-918.
19.Buckley, D. I., and Ramachandran, J. (1981)
Proc. Natl. Acad. Sci. USA, 78, 7431-7435.
20.Naville, D., Barjhoux, L., Jaillard, C., Saez,
J., Durand, P., and Begeot, M. (1997) Mol. Cell. Endocrinol.,
129, 83-90.
21.Abramov, V. M., Arkhangelskaya, Z. A., and
Zav'yalov, V. P. (1983) Biochim. Biophys. Acta,
742, 295-302.
22.Tischenko, V. M., Zav'yalov, V. P., Medgyesi, G.
A., Potekhin, S. A., and Privalov, P. L. (1982) Eur. J.
Biochem., 126, 517-521.
23.Schneider, I. I., de Dwe, C., and Trowet, D.
(1981) J. Cell. Biol., 88, 380-387.