REVIEW: Natural Catalytic Antibodies (Abzymes) in Normalcy and Pathology
G. A. Nevinsky*, T. G. Kanyshkova, and V. N. Buneva
Novosibirsk Institute of Bioorganic Chemistry, Siberian Division of the Russian Academy of Sciences, ul. Lavrentieva 8, Novosibirsk, 630090 Russia; E-mail: Nevinsky@niboch.nsc.ru* To whom correspondence should be addressed.
Received April 10, 2000; Revision received June 5, 2000
This review summarizes literature data on natural abzymes. Peculiar features of their functioning and substrate specificity are considered in comparison with traditional enzymes. Working hypotheses on the possible biological roles of natural abzymes in autoimmune processes and diseases accompanied by disorders of immune status are analyzed.
KEY WORDS: natural catalytic antibodies, autoimmune diseases
Abbreviations: AD) autoimmune diseases; Ig) immunoglobulins; SLE) systemic lupus erythematosus; TSA) transition state analog; VIP) vasoactive intestinal peptide.
The possibility of the induction of catalytic antibodies (abzymes) was
initially suggested by Pauling in 1948; he noted similar features
between mechanisms of antibody-antigen recognition and the interaction
of a transition state with an enzyme [1]. In 1969,
Jencks proposed the hypothesis that antibodies obtained during
immunization with chemically stable transition-state analogs could
catalyze the corresponding chemical reactions [2].
This was independently confirmed by Lerner's [3]
and Schultz' [4] groups in 1986.
During the thirty-year period of directed synthesis of abzymes, antibodies capable of catalyzing more than 100 various reactions have been generated. Data on induced abzymes have been considered in several reviews [5-17]. Recent development of the technology for induced abzyme production has stimulated not only the rapid development of direct approaches for generation of abzymes with designed properties. It has become possible to improve the properties of abzymes by modifying the antigen-binding site of monoclonal antibodies at the level of the encoding genes using either site-directed mutagenesis or selective chemical modification [18-22]. This has resulted in the generation of abzymes with higher substrate specificity than is found for enzymes. In some cases, rates of chemical reactions catalyzed by abzymes are close to or even higher than those of similar reactions catalyzed by ordinary enzymes. Moreover, there are some abzymes having no natural-enzyme analogs [23]. So, the practical importance for biotechnology and medicine of antibodies with designed catalytic specificity cannot be overestimated. One of the promising directions in this field consists of the production of abzymes catalyzing rapid cleavage of hazardous compounds, including drugs and toxins.
Animals generate such abzymes during directed immunization. Successful manipulations on abzyme generation have stimulated a new round of studies on the structure and functions of Ig in vivo. The discovery by Paul et al. (1989) of natural catalytic antibodies possessing proteolytic activity in blood serum of patients with bronchial asthma [24] opened new perspectives for abzymology.
PECULIARITIES OF THE IMMUNE STATUS OF PATIENTS WITH VARIOUS
AUTOIMMUNE DISEASES
Autoimmune diseases (AD) are characterized by high titer of auto-antibodies (antibodies to endogenous antigens). In the case of bronchial asthma, vasoactive intestinal peptide (VIP), widely distributed in the central and peripheral nervous systems, acts as such an antigen [24]. Autoimmune thyroiditis is characterized by increased concentrations of antibodies to thyroglobulin, the microsomal fraction of thyrocytes, and some other antigens [25].
Multiple sclerosis, a chronic degenerative demyelinating disease of the central nervous system, is a serious medical and social problem. Its etiology remains unclear, and the theory of pathogenesis is mainly focused on the degradation of myelin (the protein-lipid sheath of axons) that stems from autoimmune inflammatory processes [26]. Although the T-cell immune system plays the major role in the pathogenesis of multiple sclerosis, the development of this disease needs normal functioning of the B-cell system. In patients with multiple sclerosis, increased levels of Ig (usually IgG) and free light chains (see [25, 26]) were observed in the brain tissue and cerebrospinal fluid. As in the case of other ADs, poly-specific DNA-binding antibodies interacting with phospholipid have also been recognized in patients with multiple sclerosis [25, 27, 28].
Although some viral diseases like acquired immune deficiency syndrome and viral hepatitis are not related to ADs, they significantly influence the immune status of patients. For example, the development of both viral hepatitis [28] and AIDS [29, 30] is accompanied by autoimmune humoral and cellular reactions; as in the case of AD, tissue-specific and organ-nonspecific antibodies were found in blood of patients with hepatitis.
Increased concentrations of DNA and anti-DNA antibodies were found in blood of patients with various ADs: systemic lupus erythematosus (SLE) [31-36], autoimmune hepatitis, Grave's disease, polymyositis, multiple sclerosis, Sjogren's syndrome [34], and also with lympho-proliferative [35] and some viral diseases (e.g., AIDS [30]). Concentrations of DNA and anti-DNA antibodies were especially high in patients with SLE [31, 34, 35].
The development of AD is characterized by spontaneous generation of primary antibodies to proteins, nucleic acids, and their complexes; then secondary antibodies to the primary ones are generated, etc. [36-38]. The diversity of antibodies often detected in blood of patients with AD can be well-explained using Jerne's anti-idiotypic network [39]. According to this network, primary antibodies (antibodies-1) are denominated as idiotype, and secondary antibodies (antibodies-2) are anti-idiotype, etc. The presence of blood serum antibodies-4 titer has been recognized in experimental animals. Similar antibody generation can be also expected in the case of the viral diseases.
If the active site of an enzyme plays the role of antigen triggering the anti-idiotypic chain, it is logical to suggest that the secondary anti-idiotypic antibodies (antibodies-2) will possess protein structure, part of which represents an “internal image of the active site of the enzyme” and, consequently, these antibodies may possess some properties of this enzyme. So, at present it is believed that natural abzymes can be generated via such an anti-idiotypic chain. Abzyme generation in AD may probably involve other pathways. Some abzymes are antibodies directly generated to hapten (protein, DNA, etc.). This suggests the possibility for abzyme production via the pathway described for antibodies induced by transition-state analogs. In this case, a certain conformer of some endogenous components might play the role of transition-state analog. Autoimmune diseases are accompanied by intensive cell apoptosis. This may result in abnormally high concentrations in the blood of both native and partially destroyed proteins, nucleic acids, and other cell components. This suggests that both pathways of abzyme generation (production of anti-idiotypic antibodies and Ig to substrate itself) can be realized in AD.
NATURAL ABZYMES IN PATIENTS WITH AUTOIMMUNE DISEASES
Natural catalytic antibodies were first found in blood of asthmatic patients [24]. A small fraction of the antibodies was able not only to bind VIP, but also to hydrolyze it. This hydrolytic activity was highly specific with respect to VIP; it was not observed with other peptides and proteins.
However, to be more precise, the first reports on enzymatic activities of natural antibodies appeared earlier, in the middle of the 60s. Slobin [40] demonstrated hydrolysis of p-nitrophenyl acetate catalyzed by Ig. Kulberg et al. [41] found proteolytic activity of highly purified rabbit antibodies. At that time, all the authors believed that although the antibodies were isolated by affinity chromatography using antigen-sorbent, the catalytic activity could be ascribed to some “proteolytic enzymes attached to antibody molecules” [40, 41]. So, until 1986 (when the existence of abzymes was firmly recognized [3, 4]) the central dogma that the main function of antibodies consists in binding and elimination of antigens dominated completely. In 1991, Kit et al. found that homogenous sIgA of human milk possessed protein kinase activity [42]. However, evidence that this catalytic activity is an intrinsic property of these antibodies was presented later.
The demonstration of the intrinsic catalytic activity of natural antibodies is one of the most difficult problems of abzymology. First, catalytic activity of antibodies is often much less than that of ordinary enzymes. Natural antibodies are polyclonal antibodies and therefore they represent a wide spectrum of structurally related Ig with highly variable primary structure (especially in regions responsible for antigen binding). So, Ig may form complexes with various proteins and enzymes, and co-purification of such complexes during antibody purification cannot be ruled out. This stimulated the development of strict criteria (some of them were proposed by Paul et al. [24]), which must be taken into consideration when catalytic activity is attributed to abzymes. Unfortunately, the first reports [40, 41] did not contained such evidence that catalytic activity was an intrinsic property of the antibodies themselves, and the study of Paul et al. [24] is unquestionably considered to be the first convincing evidence for the existence of natural abzymes. Later, criteria by Paul et al. [24] were accepted by other laboratories, and additional criteria accompanied subsequent development of this field.
The second natural catalytic abzymes, DNA-hydrolyzing Ig, were found by Gabibov's group in blood of patients with SLE [43, 44], rheumatoid arthritis, systemic scleroderma [45], and later with AIDS [30]. In patients with systemic lupus erythematosus, our group found RNA-hydrolyzing IgG [46]. In milk of parturient women, we found sIgA-antibodies with protein kinase activity [47] and ATP-hydrolyzing IgG [48]. It was also demonstrated that a small fraction of Ig from blood of patients with autoimmune thyroiditis can specifically cleave thyroglobulin but not other human proteins [25].
We have found Ig possessing phosphatase activity and also RNA-, DNA-, ATP-hydrolyzing IgG and IgM in blood of patients with the following ADs: SLE [46, 49-56], autoimmune thyroiditis [54-57], polyarthritis [54-56], multiple sclerosis [57], and also with viral hepatitis (A, B, C, and D) [58]. Recently Neustroev's group demonstrated the presence of IgG with amylolytic activity in blood of patients with pyelonephritis, rheumatoid arthritis, and some malignant tumors [59]. Our data indicate that antibodies isolated from blood of healthy donors and of patients with influenza, pneumonia, tuberculosis, tonsillitis, duodenum ulcer, and some cancers did not exhibit any catalytic activity [57, 58]. Some catalytic activity was found in antibodies of patients with blood cancers [58]. However, the activity of these abzymes was much less than that of abzymes in AD patients.
CATALYTIC ANTIBODIES OF HUMAN MILK
Due to the absence of evident immunization factors, a hypothesis suggesting the existence of natural abzymes in healthy donors without disorders of immune status did not attract much interest for a long time.
During first months after birth, the immune system of neonates and babies is not completely formed: the mucosal surfaces of the respiratory and gastrointestinal tracts lack antibodies [60, 61]. Antibodies of maternal milk covering mucosal surfaces represent one of the main protective mechanisms of neonates and babies (passive immunity) against bacteria and viruses [60, 61]. Studies on neonate animals revealed that milk IgG can penetrate into their blood from the milk [61]. In this connection, detailed analysis of the possible biological role of antibodies of human milk is especially important. Taking into consideration the special role of maternal milk in the protection of babies against various hazardous environmental factors, we investigated the catalytic properties of antibodies of human milk.
In milk of healthy parturient women, we initially found sIgA catalyzing protein phosphorylation [47, 62, 63]. Casein (and more than 10 other milk proteins) and ATP were substrates for this reaction. At present, this is the only example of natural abzymes catalyzing a bi-substrate reaction.
Later we demonstrated that small sub-fractions of polyclonal IgG and sIgA of human milk catalyzed reactions of hydrolysis of DNA-, RNA- [64-67], ribo- and deoxy-NMP, -NDP, and -NTP [48]; they also exhibited phosphatase activity by catalyzing the cleavage of 5´-terminal phosphate of DNA and RNA [66, 67]. It should be noted that human milk antibodies are usually characterized by higher catalytic activity than that of abzymes isolated from patients with ADs [64-67]. This is consistent with higher amylolytic activity of antibodies from maternal milk than from blood of AD patients [59].
PROBLEMS OF ISOLATION OF HOMOGENOUS PREPARATIONS OF ABZYMES WITH
HIGH CATALYTIC ACTIVITY
Isolation of homogenous preparations of monoclonal abzymes is a relatively simple task that is usually solved by antibody chromatography on sorbents with immobilized haptens. Isolation of homogenous preparations of natural abzymes with high activity is one of the most difficult problems. Total antibody preparations are polyclonal ones containing a large number of Ig characterized by different affinity to various haptens, including proteins and enzymes, etc., which can be tightly-bound to these antibodies. Various abzymes of these polyclonal antibody preparations may have completely different origin, and their interaction with any immobilized substrates may dramatically vary. Thus, at the first stage it is necessary to separate antibodies from all possible antigens, proteins, enzymes, etc. Then an antibody fraction characterized by an increased affinity to potential substrate should be isolated; then the abzyme fraction needs to be separated from antibodies exhibiting affinity for the same ligand but lacking catalytic activity.
Modern methods of abzyme purification can fulfill all the above-described stages except the last one. At present, there are no methods for separation of abzymes and antibodies without catalytic activity. Our data indicate that the abzyme content in fractions of antibodies exhibiting affinity for some ligand (e.g., DNA) may be ~1-5% or even less. So, apparent values of the specific activity of antibodies are greatly underestimated. Purification conditions also significantly influence apparent characteristics of specific activities of antibodies. So, it is relevant to consider the main stages of abzyme purification.
The protocol for purification to homogeneity of human milk sIgA-abzymes with protein kinase activity may serve as a good example of the protocol for abzyme purification including all chromatographic stages of abzyme isolation [47, 62, 63]. Purification of sIgA-abzymes included sequential chromatography of milk protein on protein A-Sepharose, gel-filtration (under condition of “acidic shock”) and ion-exchange chromatography on DEAE-cellulose (separation of antibodies into classes), and affinity chromatography on ATP- and casein-Sepharose (isolation of fractions with increased affinity for these substrates).
Affinity chromatography on sorbents with immobilized anti-IgG-, anti-IgM-, anti-IgA-antibodies, and proteins A or G usually represents the first stage of isolation of abzymes from blood or milk [47, 63]. Under certain chromatographic conditions, the use of these sorbents allows antibody preparations lacking enzymatic contaminants to be obtained. We found that protein A-Sepharose exhibiting (for unknown reasons) higher affinity only to abzymes but not to antibodies without catalytic activity is the optimal sorbent for this stage [47, 56-58, 63, 66, 67].
sIgA-antibodies represent 90-95% of the total pool of human milk antibodies; the content of IgG and IgM does not exceed 1-3% [68, 69]. Anti-IgA-Sepharose quantitatively adsorbs the whole spectrum of milk sIgA-antibodies [47, 48, 63, 66, 67]. Protein A-Sepharose binds only 5-10% of all sIgA (the exact value depends on donors) and ~80-90% of milk IgG and this Ig fraction contains the whole set of abzymes, which we investigated [47, 63, 66, 67]. Adsorption of antibodies on protein A-Sepharose and protein elution by buffer containing Triton X-100 and 0.3-0.5 M NaCl is accompanied by dissociation of even tightly bound complexes of antibodies and proteins. Part of the bound antibodies can be eluted by a weakly acidic buffer (pH 4.6), but these antibodies lack catalytic activity. sIgA-Abzymes are eluted only by acidic buffer (pH 2.6). Thus, the use of protein A-Sepharose instead of anti-IgA-Sepharose produces an antibody preparation enriched in abzymes up to 10-20 times.
A similar situation was observed during purification of IgG- and IgM-abzymes (or their mixture) on protein A-Sepharose. The use of buffer containing Triton X-100 and then weakly acidic buffer (pH 4.6) also resulted in the elution from the sorbent of tightly bound proteins including a small fraction of IgG and/or IgM with lower affinity for protein A. Acidic buffer (pH 2.6) eluted abzyme-enriched fractions representing electrophoretically highly purified antibodies. This approach for antibody purification allows the evaluation of catalytic activity of antibodies from each donor blood after elution from protein A-Sepharose without additional purification [51-59]. The use of ion-exchange chromatography (for antibody separation into classes, IgG, IgM, sIgA) and affinity chromatography on sorbents with one of the immobilized substrates (DNA, RNA, ATP, protein, or lipid) increase activity of antibodies due to the increase of abzyme content in these fractions. After elution of antibodies from protein A-Sepharose by acidic buffer (pH 2.3), the obtained fraction is immediately subjected to gel-filtration under conditions of “acidic shock”. Although these two sequentially run stages usually yield homogenous abzyme preparations, the conditions of “acidic shock” may exert a damaging effect on the abzyme activity. Under such conditions many known enzymes completely loose their catalytic activity. For example, besides IgG- and sIgA-abzymes, human milk contains only one protein kinase of non-immunoglobulin nature; it is completely and irreversibly inactivated under the conditions of acidic shock. At the same time, some enzymes (e.g., human ribonucleases) possessing the same activities as abzymes readily regain catalytic activity after dialysis of fractions obtained after gel-filtration against buffer of neutral pH value. Apparently, all abzymes including antibodies with nuclease activities are characterized by relatively low resistance to “acidic or alkaline shock”.
Using abzymes from blood of patients with multiple sclerosis [57], we found that right after the treatment of antibodies with acidic buffers (chromatography on protein A-Sepharose, gel-filtration) their catalytic activity in the reaction of nucleic acid hydrolysis was significantly reduced. Dialysis and subsequent storage of antibody solutions at 4°C for 1-5 weeks markedly restored their activity.
Such abzyme behavior was also confirmed for antibodies isolated from blood of patients with SLE, autoimmune thyroiditis, and viral hepatitis [55-58]. Such treatment of ordinary RNAses and DNAses insignificantly influenced their activity. In the case of polyclonal antibodies, the recovery of catalytic activity (protein renaturation) of antibodies characterized by distinct specificity in hydrolysis of nucleic acids occurred at various time intervals of the storage of antibody solution after dialysis. Experiments on tRNAPhe hydrolysis revealed that all antibodies exhibited two types of RNAse activity [52-56]. The first type of activity, which was close to RNAse A in specificity, required low salt concentrations, whereas the second type of activity was stimulated by manganese ions. The latter exhibited unique substrate specificity: in contrast to human RNAses, it preferentially hydrolyzed double-stranded sites of tRNA. Immediately after antibody isolation, a low level of the first type (RNAse A like) of activity is detected; its gradual recovery is accompanied by a change of substrate specificity--in addition to breaks at CA and UA, other breaks at other sequences also appeared. In parallel, Mg2+-stimulated (the second type) RNAse activity becomes also detectable. The time of manifestations of various activities varies from preparation to preparation and from one disease to another. In some cases, stable activity of certain specificity appears only after 3-6 weeks of storage of neutral antibody solutions at 4°C. We believe that this phenomenon may be explained by different rates of renaturation of various abzymes constituting polyclonal antibody preparations.
Interestingly, all activities of abzyme preparations gradually increase during storage in neutral buffers. However, full recovery of any catalytic activity cannot be achieved for any type of abzyme.
Thus, harsh isolation methods are required at this stage of abzyme isolation not only to yield homogenous antibody preparation, but also to provide evidence that the catalytic activity is due to Ig. However, these methods do not allow highly active abzyme preparations to be obtained.
Data of abzyme purification on sorbents with immobilized substrates also suggest very high heterogeneity of human polyclonal abzymes. For example, chromatography of homogenous preparations of sIgA-abzymes on ATP-Sepharose using a NaCl concentration gradient results in distribution of the protein material over the whole concentration gradient from 0.05 to 3 M; some portion of the abzymes was eluted from the column only by 3-4 M MgCl2, or by the acidic buffer [47, 62, 63]. All of the resulting fractions catalyzed the phosphorylation reaction of milk proteins. (The only exception was sIgA eluted in the process of application to the column; this represented from 20 to 50% of sIgA in various donors). The increase of salt concentration required for the elution of a certain abzyme fraction correlated with the increase of affinity of these fractions for ATP. Some sIgA fractions differed by substrate specificity (with respect to phosphorylation of various milk proteins).
Subsequent chromatography of sIgA fractions exhibiting affinity for ATP-Sepharose on casein-Sepharose also resulted in the distribution of the protein material over the whole NaCl concentration [47, 62, 63]. This suggests that total preparations of such abzymes are characterized by a wide range of affinity for ATP and casein and variation of these parameters depends on the milk donor.
This separation of polyclonal antibodies into numerous sub-fractions in the process of affinity chromatography on ATP- and casein-Sepharose well reflects the general tendency: abzyme preparations of human milk and blood contain many sub-fractions of antibodies possessing a wide diversity in the affinity for various substrates (DNA, RNA, proteins, nucleotides, etc.). The spectrum of substrate specificity of abzymes and ranges of changes of abzyme affinity for substrates, pH optima, and conditions for manifestation of maximal catalytic activity (concentration of mono- and bivalent metal ions, etc.) strongly depend on type of AD, individual, time-course and stage of the disease, and period of collection of the patient's blood.
EVIDENCE FOR ATTRIBUTING CATALYTIC ACTIVITY TO ANTIBODIES
Demonstration that the catalytic activity represents an intrinsic property of the antibodies themselves is a most difficult task. Paul et al. [24] presented the following evidence for attributing catalytic activity to antibodies: 1) electrophoretic homogeneity of IgG (protein staining with silver); 2) the presence of VIP-hydrolyzing activity in Fab-fragments; 3) complete adsorption of catalytic activity on anti-IgG-Sepharose and elution of IgG from sorbent by buffer with low pH; 4) immunoprecipitation of abzymes with anti-IgG-antibodies accompanied by complete loss of VIP-hydrolyzing activity; 5) the presence of catalytic activity in two of six IgG isolated from blood serum of patients; lack of catalytic activity in antibodies isolated from blood of healthy donors; 6) gel-filtration of antibodies under conditions of pH shock did not cause loss of catalytic activity; 7) Km value (38 nM) for VIP in the hydrolytic reaction suggested high affinity of IgG for substrate compared with antibody-antigen complexes; 8) specificity of cleaving sites of VIP (see below) differed from the specificity of cleavage of this peptide by known proteases.
Similar lines of evidences were used for demonstration that DNAse activity is an intrinsic property of IgG [29, 30, 43, 44].
Studying abzymes from patients with various ADs [46, 49-57] and from human milk [47, 48, 62-67], we have used the whole set of these criteria. However, for precise attribution of the observed activity to antibodies rather than to possible enzymatic contaminants we added the following criteria [47, 48, 51-53, 57, 62-67]: a) stability of catalytic activity of antibodies on gel-filtration under alkaline conditions and in buffers containing isothiocyanate; b) demonstration of catalytic activity in isolated chains of antibodies; c) the existence of differences in the thermostability of catalytic activities of antibodies versus enzymes catalyzing the same reactions; d) effective separation of a mixture of homogenous antibodies from exogenous enzymes catalyzing the same reaction by using methods of abzyme purification from biological tissues; e) selective labeling of oligomeric antibodies and also abzyme subunits (but not any protein impurities) by active substrate analogs; f) detection of catalytic activity in situ in bands of the substrate-containing gel (after SDS-electrophoresis in the Laemmli system) corresponding to IgG and their Fab-fragments or separate chains.
The presence of catalytic activity in monoclonal antibodies obtained using hybridoma technology or gene-engineering methods with the use of DNA-plasmids encoding antibody synthesis is one of the most valuable criteria. However, although hybridoma technology is easily applied for studies of abzymes in mice, it is limited in the case of human abzymes.
After pretreatment of antibodies with alkaline buffers (pH 11-12), and also with neutral buffers containing isothiocyanate, the abzymes retained part of the initial activity of the antibodies during gel-filtration. There were no other protein peaks or analyzing activity associated with non-Ig protein peaks [63]. As mentioned above, abzymes are less resistant to various treatments than enzymes. The latter were more thermostable than abzymes. For example, incubation of IgG- and IgM-abzymes at 55°C and their Fab-fragments at 45°C resulted in almost complete loss of their RNAse-activity, whereas human serum RNAses retained from 50 to 90% activity after incubation at 65°C. RNAse A was completely active after boiling for 5-10 min [51, 53, 54].
Various DNAses and RNAses added to homogeneous antibodies isolated from blood of various patients with ADs or from human milk were readily separated from abzymes irrespectively of conditions used for the antibody purification: affinity chromatography on protein A-Sepharose, gel-filtration in acidic and alkaline buffers, and even filtration of solutions through nitrocelloluse discs permeable for these enzymes (molecular masses 12-40 kD) but not for antibodies (>150 kD) [54].
According to data of SDS-electrophoresis (followed by silver staining), homogenous antibody preparations lacking any detectable enzymatic contaminants were obtained [47, 48, 50-58, 62-67]. However, this method has limited sensitivity, and even in the case of homogenous proteins, possible contamination of antibody preparations with minimal quantities of highly active enzymes cannot be ruled out. We increased the sensitivity of this approach by using affinity modification of antibodies with active 32P-labeled reagents. However, we failed to detect modifications of any (possible) enzymatic contaminants; only abzyme molecules were labeled [47, 48, 63-67], and we detected no 32P-labeled modified proteins other than Ig. This approach reveals structural components of antibodies responsible for substrate binding. Using separation of antibody subunits by SDS-electrophoresis under dissociating conditions (after reduction of disulfide bridges), we found that in most cases the L-chains of natural abzymes were labeled with affinity reagents.
We have developed two approaches for analysis of the catalytic activity of isolated light and heavy chains of antibodies. The affinity chromatography of antibodies on immobilized substrates under denaturing conditions (dithiothreitol, 4-6 M urea) after reduction of disulfide bridges with thiol-containing reagents is the first. Using this approach, we demonstrated that isolated light chains of DNA- and RNA-hydrolyzing IgG-abzymes from human blood and milk not only bind under these conditions with DNA-cellulose but are active after their separation from heavy chains [48, 57, 62, 64-67].
SDS-Electrophoresis in polyacrylamide gel is one of the harshest separation methods used for separation of noncovalent protein complexes. We analyzed the catalytic activity of initial antibody oligomers and their individual subunits (after the reduction of disulfide bonds) using in situ SDS-electrophoresis in gel [48, 51, 54, 57, 64-67]. After the electrophoresis, DNA- or RNA-containing gels were washed to remove SDS, incubated in special buffer for antibody renaturation, and stained with ethidium bromide. The gel region where the cleavage of nucleic acid occurred is seen as a dark spot on the uniform light fluorescent background. The position of protein bands was identified by gel staining with Coomassie R-250. Using this approach, it was demonstrated that oligomeric forms of IgG antibodies (and also their Fab-fragments and separated light chains) isolated from blood of patients with SLE and multiple sclerosis and also from milk of parturient women possessed nucleic acid hydrolyzing activities [47, 54, 57, 63-67]. From our viewpoint, DNA-hydrolyzing activity detected by this in situ method only in the initial IgG preparations and their Fab-fragments under non-dissociating conditions and also in the band corresponding to light chain (in the presence of mercaptoethanol) unquestionably indicate that this catalytic activity is an intrinsic property of Ig [47, 52, 54, 57, 63-67]. In each of these cases, other sites of DNA cleavage were absent.
Isolated light chains of DNA- and RNA-hydrolyzing abzymes also exhibited catalytic activity. In some cases, catalytic activity of the isolated chains was somewhat higher than in the initial oligomeric forms. The efficacy of VIP hydrolysis catalyzed by the isolated IgG light chain was significantly higher than in the case of intact IgG, but site-specificity of cleavage remained the same [70]. This suggests that in some cases active sites of natural abzymes are located on their light chains. Nevertheless, data of analysis of abzymes induced by transition-state analogs suggest that the active site formation involves both light and heavy chains; sometimes antibody heavy chains have greater contribution [16]. Apparently, there are significant variations in the arrangement of active sites of natural abzymes. For example, subunit separation of DNA-hydrolyzing sIgA-antibodies from human milk on DNA-cellulose resulted in binding of only heavy chains with the sorbent [67]. The isolated light chains possessing low affinity for DNA exhibit catalytic activity at high substrate (DNA) concentration. Analysis of catalytic activity of sIgA subunits in gel revealed only light chain activity, whereas reactive oligonucleotide derivatives modify with equal efficacy both light and heavy chains of sIgA-abzymes. So, active site formation involves both types of chains. However, the DNA-binding site of sIgA-nucleases is preferentially located on heavy chains, whereas the active site is located on light chains [67].
The arrangement of the active site of ATP-hydrolyzing human milk IgG is even more complex [48]. Reactive ATP analogs modify both L- and H-chains of these antibodies. However, only initial IgG oligomer (H2L2) and products of its partial reduction (H2L and HL) containing at least one L- and one H-chain of the initial oligomer could catalyze ATP hydrolysis. Free L- and H-chains lack catalytic activity. This suggests that active sites of abzyme-ATPases are located at the “junction” of L- and H-chains and amino acids residues of both antibody chains are involved in catalysis of ATP hydrolysis. X-Ray analysis revealed similar arrangement of hapten-induced monoclonal IgG-abzymes with esterase activity. During complex formation with antibody, the transition-state analog interacted with both light and heavy chains of Ig [71].
Thus, the additional criteria not only provide unquestionable evidence for the presence of catalytic activity as an intrinsic property of antibodies. They also demonstrate significant variations in the organization of the active sites of abzymes. These variations depend on the source of abzymes and their catalytic functions. It is also possible that the organization of active sites of abzymes realizing the same catalytic function will also differ in different donors and even in the same donor, because natural antibodies are polyclonal ones. For example, affinity modification of human milk sIgA-abzymes (possessing protein kinase activity) with reactive ATP analogs revealed that in most cases light chains undergo this modification. At the same time, in several cases of donor samples modification of only the heavy chain or of both chains was observed.
Our data on the determination of the type of abzyme light chains suggest the existence of different variants of immune response in patients with ADs and healthy parturient women. For determination of the type of catalytic light chain of abzymes isolated from blood of patients with ADs and from human milk, we investigated their affinity for the immobilized IgG against light chains of kappa- and lambda-types. All abzymes bound to both sorbents, but the ratio of abzymes containing chains of kappa- and lambda-types depends on type of catalytic activity of the antibodies and their source. A similar situation was observed in the case of IgG- and IgM-abzymes with nuclease activity isolated from patients with multiple sclerosis and SLE: 70-80 and 60-70% antibodies with kappa- and 20-30 or 30-40% antibodies with lambda-chain, respectively [51, 57]. However, in the case of abzymes from blood of patients with SLE only lambda-type light chains possessed DNA-hydrolyzing activity [51]. In bronchial asthma, lambda-type chains were able to catalyze VIP hydrolysis [72]. The majority (95%) of human milk abzymes hydrolyzing nucleic acids contain kappa-type chains [64], whereas the proportion of human milk ATP-hydrolyzing abzymes with this type of light chains was somewhat less (80-90%) [48]. It should be noted that within one preparation abzymes containing kappa-type light chain are usually more effective catalysts of hydrolysis of nucleic acids and ATP than antibodies containing lambda-type light chain [48]. Thus, all these data indicate that all catalytic antibodies are highly polyclonal.
ENZYMATIC PROPERTIES OF NATURAL CATALYTIC ANTIBODIES
Although natural abzymes and corresponding enzymes catalyze the same reactions, their properties significantly differ, especially in substrate specificity and affinity [46-58, 62-67, 70]. All known protein kinases including the single milk protein kinase use only ATP as the nucleotide substrate, whereas milk sIgA-abzymes phosphorylating about 10 milk proteins can use almost all deoxy- or ribonucleoside-5´-triphosphates as the phosphate donor [47, 62, 63]. Abzymes as well as numerous protein kinases phosphorylate serine residues, but at distinct positions of the amino acid sequences [47, 63]. Milk sIgA- and IgG-abzymes with nucleotidase activity exhibit properties of nonspecific phosphatases: they can hydrolyze various deoxy- or ribo-NMP, -NDP, or -NTP; however, in contrast to phosphatases these abzymes do not hydrolyze such typical phosphatase substrates as p-nitrophenyl phosphate and alpha-naphthol phosphate [48].
All natural abzymes are polyclonal antibodies representing a mixture of monoclonal antibodies with various properties, which can be partially separated into sub-fractions using affinity chromatography on immobilized substrates [47, 48, 51, 57, 62-67]. Small sub-fractions of abzymes with various affinities for substrate usually demonstrate distinct kinetic and thermodynamic characteristics. Nevertheless, some abzymes behave like the monoclonal antibodies, i.e., as one enzyme with some constant properties. This group includes DNA-hydrolyzing antibodies (from patients with SLE, human milk) [43-45, 47, 64-67]. These abzymes are rather variable and differ from known eucaryotic DNAses. Nevertheless, it is possible that in each AD case a sub-fraction of abzymes preferentially produced by one clone of immunocompetent cells (or antibodies corresponding to different clones but with close enzymatic characteristics) predominates in the total abzyme preparation.
The activity of RNA-hydrolyzing abzymes also vary in different individuals, but it generally increases in the order: hepatitis <= autoimmune thyroiditis <= polyarthritis < SLE <= multiple sclerosis [51, 54-58]. Using classic RNAse substrates (cCMP, polyU, polyA, and polyC) and also tRNA molecules possessing rigid compact structure (like tRNAPhe), it was found that abzymes have distinct substrate specificity that is individual for each type of AD; the substrate specificity of all abzymes significantly differs from that of human blood RNAses and ribonuclease A [51-56]. The table summarizes the relative rates of various hydrolytic reactions catalyzed by human RNAses and antibodies isolated from blood of patients with various ADs. All IgG- and IgM-abzymes effectively hydrolyzed polyA, whereas RNAse A and related human blood RNAses were almost inactive with this substrate [46, 49-57]. Using the substrate specificity of the hydrolytic reaction catalyzed by antibodies, it is possible to discriminate each AD studied [54, 57]. The ratio of rates of polyA and polyC hydrolytic reactions catalyzed by RNAses and abzymes is especially obvious (table).
Relative specific activities of IgG preparations isolated from blood of
patients with various ADs compared with RNAse A and human plasma
nucleases evaluated by hydrolysis of polyribonucleotides and cCMP
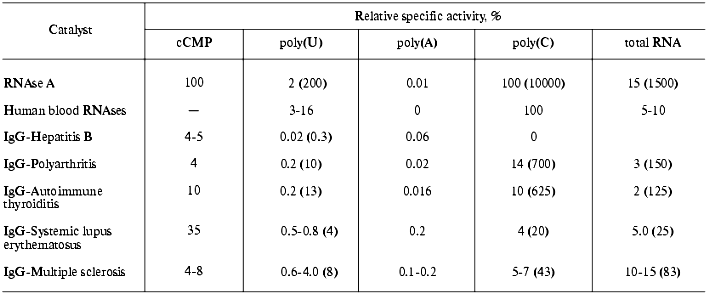
Note: Specific activities were evaluated per mole of protein.
Specific activities of RNAse A catalyzing reactions of polyC and cCMP
hydrolysis were denoted as 100%, and specific activities of each
catalyst during hydrolysis of polyU, polyA, and total RNA were
normalized to RNAse A activity during polyC hydrolysis. Numbers in
brackets indicate reaction rate ratio of hydrolysis of each polymer and
polyA.
Specific activities of RNA-hydrolyzing antibodies are strongly increased in patients during exacerbation of ADs and especially in patients with prolonged chronic duration of these diseases [55-58]. Increase of catalytic activity often correlates with the broadening of substrate specificity of RNA-hydrolyzing antibodies. In the case of patients with ADs and of human milk, there is a correlation between the level of RNA-hydrolyzing antibodies and the increase of other catalytic activities (hydrolysis of DNA, nucleotides, proteins, polysugars) of homogenous antibody preparations that were not fractionated using affinity sorbents. Some abzyme sub-fractions with RNAse activity isolated from blood of patients with ADs were activated by high concentrations of mono- and bivalent metal ions; they hydrolyze substrate under conditions of total inhibition of all known human RNAses [52-55]. Under certain conditions, some sub-fractions of RNA-hydrolyzing antibodies exhibited substrate specificity close to that of RNAses from some bacteria, viruses, and also from cobra and viper snake venom which are highly toxic for humans [52-55]. Large variations of components constituting pools of polyclonal abzymes in various patients can account for different pH behavior of RNAse activity of abzymes. Some antibodies possess one pH optimum (from 7 to 8), others have a few pH optima in the range from 6.0 to 8.5; there are antibodies effectively hydrolyzing RNA at comparable rate within the range of pH from 4.5 to 9.5 [51, 57]. Abzymes are often characterized by one, two, three, or more Km values for substrate; this reflects the efficacy of the interaction of various abzyme sub-fractions with substrate. The range of Km values (1-100 nM) is typical for antibody-antigen interaction; it is a few orders of magnitude less than that of the Km values for all known ribonucleases [50].
We have also analyzed [52-55] possible changes of substrate specificity of abzymes using a representative set of natural RNAses and (transcripts) and varying reaction conditions. This approach reveals not only major but also minor fractions of abzymes; each patient is characterized by a set of abzymes with RNAse activity, and some of them are unique. Taking into consideration all the data [46, 49-58], we came to the conclusion that the formation of highly heterogeneous RNA-hydrolyzing polyclonal IgG and IgM fractions depends on type of disease and individual features of patients. These Ig can contain both kappa- and lambda-type light chains, exhibit maximal activity at various pH values, carry distinct total charge, possess various affinities to substrates, and demonstrate different dependence on mono- and bivalent metal ions. The high diversity of RNAse activities found in some patients cannot be explained by changes in the set of RNAses, because human blood contains just five RNAses, and only one of them differs in substrate specificity from RNAse A [46, 49-58].
In contrast to known viral, procaryotic, and eucaryotic RNAses, some RNA-hydrolyzing antibodies are sensitive to minimal changes of spatial structure of RNA molecules and also to minimal changes in nucleotide sequences, including point base substitutions [52-55]. For example, a single substitution of A for G in the T-loop of tRNALys leads to the development of neuromuscular disorder in man (KMERRF) [52-55]. This mutation cannot be recognized by any structural probing of RNA molecules [52-55]. However, hydrolytic products of natural and mutant tRNA cleaved by abzymes isolated from blood of patients with SLE, autoimmune arthritis, and polyarthritis yield different electrophoretic patterns, allowing quick detection of this disorder [52-55]. Thus, monoclonal antibodies can be considered as a basis for a new generation of enzymes that are promising tools for studies of structural features of RNA molecules.
The comparison of relative efficacy of abzymes with enzymes catalyzing the same reactions is an important problem of abzymology. In many cases, specific activities represent from 0.01 to 5% of the activity of the most effective enzymes hydrolyzing DNA, RNA, proteins, or nucleotides [50, 56-58]. However such abzyme activities cannot be considered as low because not all enzymes are highly active; many of them are characterized by relatively low specific activities (comparable with that of abzymes), and, nevertheless, they accomplish important biological functions. The rate of DNA cleavage catalyzed by antibody is close to that of restriction enzymes (e.g., EcoRI endonucleases widely used in gene engineering) and significantly higher than in DNA methylases [44].
The specific activity of abzymes strongly depends on the patient from which they were isolated and the type of disease. In some cases of SLE, we isolated antibodies hydrolyzing RNA with specific activity that corresponded to 30-40% of the activity of pancreatic RNAse A, one of the most active enzymes [56-58]. The rate of polyU hydrolysis catalyzed by antibody preparations isolated from some patients with multiple sclerosis was 2-4-fold higher than that of RNAse A [57]. Since there are no methods for separation of abzymes from non-catalytic antibodies exhibiting affinity for the same ligands and co-purifying with abzymes, it is clear that maximal rate values determined for antibody-dependent substrate conversion are strongly underestimated. This suggests that in exacerbations of ADs, specific abzyme activities may be quite high, and it is possible that such abzymes can influence the development of the disease.
POSSIBLE BIOLOGICAL ROLE OF NATURAL HUMAN ABZYMES
The data considered above raised many questions both on nature and possible biological role of natural abzymes. So, we investigated possible reasons for abzyme appearance in milk of parturient women.
Abzyme levels are especially high in milk of women who were ill with viral infections or allergic diseases during pregnancy [64, 66, 67]. Immunization of mammals with proteins for 1-3 months (but not more) before delivery results in the appearance of high concentrations of milk antibodies to these proteins [73, 74]. The way of immunization (intravenous or peroral administration of proteins) was not significant. It was also not significant whether specific antibodies (IgG, IgA, and sIgA) can be transported from blood to milk (as IgG) or produced locally by specialized cells of the milk-forming system (sIgA).
We have recently shown that the level of blood abzymes of pregnant women is relatively low; it sharply increases during lactation and this correlates with high titer of human milk abzymes [66, 67]. In other words, pregnant women are characterized by a special type of immune response to all possible antigens entering the body with food or during contact with viruses and bacteria. We believe that pregnant women possess some specific “immune memory” accumulating all “information” about environmental components that may be potentially dangerous for their newborn babies.
The pregnant body is also subjected to internal “hidden autoimmunization”. During the first trimester of pregnancy, a woman's blood (as well as blood of patients with systemic lupus erythematosus) contains elevated concentrations of DNA [67]; the last trimester is characterized by cell apoptosis typical for ADs [75]. The blood of pregnant women also contains small concentrations of embryonal cells [76]. Thus, during pregnancy women are subjected to “external” and “internal” immunization. However, if preparation of the mother's milk is almost evident in the case of antibodies binding antigens, it is possible that such “specific preliminary immunization” does not program the female body for production of antibodies that are able to bind and also cleave components of food, viral particles, and bacterial cells. From our viewpoint, it is reasonable to consider catalytic activities of abzymes as additional enzymatic resources of the body that appear only under special conditions like pregnancy, AD, and other yet unidentified cases.
The possible role of blood abzymes in patients with AD remains unclear. The major question is whether enzymatic properties of abzymes represent function or dysfunction of antibodies. It is equally possible that abzymes represent an additional source for pathological lesions in systemic autoimmune process or, on the contrary, they are involved in elimination of auto-aggressive T-cell clones by apoptosis. It is also possible that in different diseases they may play both positive and negative roles. Paul and his co-workers found that immunization of mice with anti-VIP-abzymes results in induction of bronchial asthma in these animals [77]. They suggest that the dysfunction of the respiratory tract in asthma may stem from protease activity of auto-antibodies [78]. Deficit of VIP in tissues of the respiratory system plays a major role in the pathophysiology of bronchial asthma. The reduced level of this neurotransmitter results in attenuation of the relaxing effect on the respiratory tract and the development of pathology. Thus, direct degradation of VIP by abzymes or competition of such auto-antibodies with cell VIP-receptors significantly reduces functional activity of this neuropeptide in tissues.
DNA-hydrolyzing IgG from blood of patients with SLE and some lymphoproliferative diseases (B-cell lymphosarcoma, T-lymphoma, chronic B-cell leukemia) are cytotoxic [34]. Mechanisms of cytotoxic effect of auto-antibodies and TNF-alpha (tumor necrosis factor) are remarkably similar. (TNF-alpha_is the cytokine inducing cell death via apoptotic mechanisms.) In SLE, the cytotoxic effect of anti-DNA-antibodies was observed at concentrations comparable or even lower than that of cytokines. Thus, cytotoxic function of DNA-hydrolyzing abzymes in AD suggests that catalytic auto-antibodies may realize another pathogenetic mechanism, complement-independent cytotoxicity.
DNA-hydrolyzing Bence-Jones proteins (light chain antibodies) isolated from urine of patients with myeloma are also cytotoxic, but in contrast to ordinary antibodies they are able to penetrate through inner and nuclear membranes and to hydrolyze chromatin DNA [79].
Li et al. consider abzyme-protease as one of the defensive factors of the body in autoimmune thyroiditis [25]. They believe that such elimination of thyroglobulin by its hydrolysis may not only minimize autoimmune response to thyroglobulin, but also prevents formation of immune precipitates in tissues.
Our and literature data suggest that human milk abzymes may protect newborn babies against viral and bacterial infections. DNAses and RNAses are effectively employed in medical practice as external applications against various viral and bacterial infections by cleaning the mucosa [80]. Increased level of ribonucleases in milk correlates with increased resistance of women to breast cancer [81].
Milk antibodies are responsible for passive immunity of newborn babies during the first postnatal months [60, 61]. It is relevant to suggest that these antibodies may not only bind components of pathogenic viruses and bacteria but also hydrolyze their RNA and DNA [48, 64-67]. Taking into consideration our data on significant differences in the substrate specificity of abzymes that appear in blood of patients with ADs, in blood and milk of healthy parturient women it still remains unclear whether abzymes represent an additional enzymatic system functioning under various extreme conditions or side-products of the immune system. Nevertheless, the potential of the human body in generating various abzymes could be quite high, and such antibodies attract much interest as possible important factors of pathogenesis and source of enzymes with new types of substrate specificity.
This work was supported by the Russian Foundation for Basic Research (grant No. 98-04-49719), the Ministry of Education of the Russian Federation “Basic Studies in Chemical Technologies” (grant No. 98-8-3.2-69), and the Siberian Division of the Russian Academy of Sciences (grant for young scientists).
REFERENCES
1.Pauling, L. (1948) Am. Scientist, 36,
51-59.
2.Jencks, W. (1969) Catalysis in Chemistry and
Enzymology, McGraw-Hill, N. Y.
3.Tramontano, A., Janda, K. D., and Lerner, R. A.
(1986) Science, 234, 1566-1570.
4.Pollack, S. J., Jacobs, J. W., and Schultz, P. G.
(1986) Science, 234, 1570-1573.
5.Blackburn, G. M., Kang, A. S., Kingsbury, G. A.,
and Burton, D. R. (1989) Biochem. J., 262, 381-390.
6.Lerner, R. A., and Tramontano, A. (1981) Trends
Biochem. Sci., 12, 427-438.
7.Massey, R. J. (1987) Nature, 328,
457-458.
8.Shultz, P. J., Lerner, R. A., and Benkovic, S. J.
(1990) Chem. Eng. News, 68, 26-40.
9.Shokat, K. M., and Schultz, P. G. (1990) Annu.
Rev. Immunol., 8, 335-363.
10.Lerner, R. A., Benkovic, S. J., and Shultz, P. J.
(1991) Science, 252, 659-667.
11.Benkovic, S. J. (1992) Annu. Rev.
Biochem., 61, 29-54.
12.Hilvert, D. (1992) Pyre Appl. Chem.,
64, 1103-1113.
13.Stewart, J. D., and Benkovic, S. J. (1993)
Chem. Soc. Rev., 22, 213-219.
14.Suzuki, H. (1994) J. Biochem., 115,
623-628.
15.Schultz, P. G., and Lerner, R. A. (1995)
Science, 269, 1835-1842.
16.Schourov, D. V. (1997) Mol. Biol.
(Moscow), 32, 5-15.
17.Nevinsky, G. A., Semenov, D. V., and Buneva, V.
N. (2000) Biochemistry (Moscow), 65, 1233-1244.
18.Sastry, L., Mubaraki, M., Janda, K. D., Benkovic,
S. J., and Lerner, R. A. (1991) Ciba Found. Symp.,
159, 145-151.
19.Guo, J., Huang, W., Zhou, G. W., Fletterick, R.
J., and Scanlan, T. S. (1995) Proc. Natl. Acad. Sci. USA,
92, 1694-1698.
20.Miller, G. P., Posner, B. A., and Benkovic, S. J.
(1997) Bioorg. Med. Chem., 5, 581-590.
21.Roberts, V. A., Stewart, J., Benkovic, S. J., and
Getzoff, E. D. (1994) J. Mol. Biol., 235, 1098-1116.
22.Stewart, J. D., Krebs, J. F., Siuzdak, G.,
Berdis, A. J., Smithrud, D. B., and Benkovic, S. J. (1994) Proc.
Natl. Acad. Sci. USA, 91, 7404-7409.
23.Na, J., Houk, K. N., and Hilvert, D. (1996) J.
Am. Chem. Soc., 118, 6462-6471.
24.Paul, S., Volle, D. J., Beach, C. M., Johnson, D.
R., Powell, M. J., and Massey, R. J. (1989) Science, 244,
1158-1162.
25.Li, L., Paul, S., Tyutyulkova, S., Kazatchkine,
M. D., and Kaveri, S. (1995) J. Immunol., 154,
3328-3332.
26.Boiko, A. N., and Favorova, O. O. (1995) Mol.
Biol. (Moscow), 29, 727-749.
27.Shoenfeld, Y., Ben-Ehuda, O., Messinger, J.,
Bentwithc, Z., Rauch, J., Isenberg, D. I., and Gado, N. (1988)
Immunol. Lett., 17, 285-291.
28.Sugiama, Y., and Yamamota, T. (1996)
Tohoku J. Exp. Med., 178, 203-215.
29.Bigazzi, P. E. (1983) Mechanisms of
Immunology (Cohen, S., Wozd, P. F., and McKlaskie, R. T., eds.)
[Russian translation], Meditsina, Moscow, pp. 181-206.
30.Gololobov, G. V., Mikhalp, S. V., Starov, A. V.,
Kolesnikov, A. V., and Gabibov, A. G. (1994) App. Biochem.
Biotechnol., 47, 305-315.
31.Gabibov, A. G., Gololobov, G. V., Makarevich, O.
I., Schourov, D. V., Chernova, E. A., and Yadav, R. P. (1994) Appl.
Biochem. Biotechnol., 47, 293-303.
32.Gilbert, D., Brard, F., Jovelin, F., and Tron, F.
(1996) J. Autoimmun., 9, 247-257.
33.Krause, I., Blank, M., and Shoenfeld, Y.
(1998) J. Biol. Regul. Homeost. Agents, 12, 49-52.
34.Shoenfeld, Y., Teplizki, H. A., Mendlovic, S.,
Blank, M., Mozzes, E., and Isenberg, D. A. (1989) Clin. Immunol.
Immunopathol., 51, 313-325.
35.Kozyr, A. V., Kolesnikv, A. V., Aleksandrova, E.
S., Sashchenko, L. P., Gnuchev, N. V., Favorov, P. V., Kotelnikov, M.
A., Iakhnina, E. I., Astsaturov, I. A., Prokaeva, T. V., Alekberova, Z.
S., Suchkov, S. V., and Gabibov, A. G. (1998) Appl. Biochem.
Biotechnol., 75, 45-61.
36.Raptis, L., and Menard, H. A. (1980) J. Clin.
Invest., 66, 1391-1399.
37.Reimer, C., Raska, I., Tan, E. M., and Sheer, U.
(1987) Virchows Arch., 54, 131-136.
38.Earnshaw, W. C., and Rothfield, N. (1985)
Chromosoma, 91, 313-320.
39.Jerne, N. K. (1974) Ann. Immunol.,
125, 373-398.
40.Slobin, I. I. (1966) Biochemistry,
5, 2836-2841.
41.Kulberg, A. Ya., Docheva, Yu. V., Tarhanova, I.
A., and Spivak, V. A. (1969) Biokhimiya, 34,
1178-1183.
42.Kit, Yu. Ya., Kim, A. A., and Sidirov, V. N.
(1991) Biomed. Sci., 2, 201-204.
43.Shuster, A. M., Gololobov, G. V., Kvashuk, O. A.,
Bogomolova, A. E., Smirnov, I. V., and Gabibov, A. G. (1992)
Science, 256, 665-667.
44.Gololobov, G. V., Chernova, E. A., Schourov, D.
V., Smirnov, I. V., Kudelina, I. A., and Gabibov, A. G. (1995) Proc.
Natl. Acad. Sci. USA, 92, 254-257.
45.Bronstein, I. B., Shuster, A. M., Gololobov, G.
V., Gromova, I. I., Kvashuk, O. A., Belostotskaya, K. M., Alekberova,
Z. S., Prokaeva, T. V., and Gabibov, A. G. (1992) FEBS Lett.,
314, 259-263.
46.Buneva, V. N., Andrievskaya, O. A., Romannikova,
I. V., Gololobov, G. V., Yadav, R. P., Yamkovoi, V. I., and Nevinsky,
G. A. (1994) Mol. Biol. (Moscow), 28, 738-741.
47.Kit, Yu. Ya., Semenov, D. V., and Nevinsky, G. A.
(1995) Mol. Biol. (Moscow), 29, 893-906.
48.Semenov, D. V., Kanyshkova, T. G., Kit, Yu. Ya.,
Khlimankov, D. Yu., Akimdzhanov, A. M., Gorbunov, D. A., Buneva, V. N.,
and Nevinsky, G. A. (1998) Biochemistry (Moscow), 63,
935-943.
49.Schourov, D. V., Makarevich, O. I., Lopaeva, O.
A., Buneva, V. N., Nevinsky, G. A., and Gabibov, A. G. (1994) Dokl.
Ros. Akad. Nauk, 337, 407-410.
50.Andrievskaya, O. A., Kanyshkova, T. G., Yamkovoi,
V. I., Buneva, V. N., and Nevinsky, G. A. (1997) Dokl. Ros. Akad.
Nauk, 355, 401-403.
51.Andrievskaya, O. A., Buneva, V. N., Zabara, V.
G., Naumov, V. A., Yamkovoi, V. I., and Nevinsky, G. A. (1998) Mol.
Biol. (Moscow), 32, 908-915.
52.Vlassov, A. V., Andrievskaya, O. A., Kanyshkova,
T. G., Baranovsky, A. G., Naumov, V. A., Breusov, A. A., Giege, R.,
Buneva, V. N., and Nevinsky, G. A. (1997) Biochemistry (Moscow),
62, 474-479.
53.Vlassov, A. V., Helm, M., Naumov, V. A., Breusov,
A. A., Buneva, V. N., Florentz, C., Giege, R., and Nevinsky, G. A.
(1999) Mol. Biol. (Moscow), 33, 866-872.
54.Vlassov, A., Florentz, C., Helm, M., Naumov, V.,
Buneva, V., Nevinsky, G., and Giege, R. (1998) Nucleic Acids
Res., 26, 5243-5250.
55.Vlassov, A. V., Helm, M., Florentz, C., Naumov,
V. A., Breusov, A. A., Buneva, V. N., Giege, R., and Nevinsky, G. A.
(1999) Russ. J. Immunol., 4, 25-32.
56.Vlassov, A. V., Baranovsky, A. G., Kanyshkova, T.
G., Prince, A. V., Zabara, V. G., Naumov, V. A., Breusov, A. A., Giege,
R., Buneva, V. N., and Nevinsky, G. A. (1998) Mol. Biol.
(Moscow), 32, 559-569.
57.Baranovsky, A. G., Kanyshkova, T. G.,
Mogilnitsky, A. S., Naumov, V. A., Buneva, V. N., Gusev, E. I., Boiko,
A. N., Zargarova, T. A., Favorova, O. O., and Nevinsky, G. A. (1998)
Biochemistry (Moscow), 63, 1239-1248.
58.Baranovsky, A. G., Matyushin, V. G., Vlassov, A.
V., Zabara, V. G., Naumov, V. A., Buneva, V. N., and Nevinsky, G. A.
(1997) Biochemistry (Moscow), 62, 1358-1366.
59.Savel'ev, A. N., Eneyskaya, E. V., Shabalin, K.
A., Filatov, M. V., and Neustroev, K. N. (1999) Protein Peptide
Lett., 6, 179-184.
60.Hanson, L. A., Carlsson, B., and Cruz, J. R.
(1979) in Immunology of Breast Milk (Ogra, P. L., and Dayton, D.
H., eds.) Raven Press., N. Y., pp. 145-157.
61.Brambell, F. W. (1970) in The
Transmission of Passive Immunity from Mother to Young (Neuberger,
A., and Tatum, E. L., eds.) North-Holland, Amsterdam-London.
62.Kit, Yu. Ya., Semenov, D. V., and Nevinsky, G. A.
(1996) Biochem. Mol. Biol. Int., 39, 521-527.
63.Nevinsky, G. A., Kit, Yu. Ya., Semenov, D. V.,
and Buneva, V. N. (1998) Appl. Biochem. Biotechnol., 75,
77-91.
64.Kanyshkova, T. G., Semenov, D. V., Vlassov, A.
V., Khlimankov, D. Yu., Baranovsky, A. G., Shipitsyn, M. V., Yamkovoi,
V. I., Buneva, V. N., and Nevinsky, G. A. (1997) Mol. Biol.
(Moscow), 31, 1088-1096.
65.Kanyshkova, T. G., Semenov, D. V., Khlimankov, D.
Yu., Buneva, V. N., and Nevinsky, G. A. (1997) FEBS Lett.,
416, 23-27.
66.Buneva, V. N., Kanyshkova, T. G., Vlassov, A. V.,
Semenov, D. V., Khlimankov, D. Yu., Breusova, L. R., and Nevinsky, G.
A. (1998) Appl. Biochem. Biotechnol., 75, 63-76.
67.Nevinsky, G. A., Kanyshkova, T. G., Semenov, D.
V., Vlassov, A. V., Gal'vita, A. V., and Buneva, V. N. (2000) Appl.
Biochem. Biotechnol., 83, 115-129.
68.Balint, J. R., and Ikeda, Jr. Y. (1981)
Immunol. Commun., 10, 533-540.
69.Xanthou, M. (1998) Biol. Neonate,
74, 121-133.
70.Mei, S., Mody, B., Eklund, S. H., and Paul, S.
(1991) J. Biol. Chem., 266, 15571-15574.
71.Golinelly-Pimpaneu, B., Gigant, B., Bizebard, T.,
Nazava, J., Saludjian, P., Zemel, R., Tawfik, D. S., Eshhar, Z., Green,
B. S., and Knossow, M. (1991) Curr. Biol. Struct., 2,
175-180.
72.Sun, M., Gao, Q. S., Li, L., and Paul, S. (1994)
J. Biol. Chem., 269, 734-738.
73.Fey, H. R., Burtler, R., and Marti, F. (1973)
Vox Sang., 25, 245-253.
74.Mestecky, J., and McGhee, J. R. (1987) Adv.
Immunol., 40, 153-245.
75.Mikhailov, V. M., Linde, V. A., Rosanov, Yu. M.,
Tarova, N. A., Susloparov, L. A., and Konysheva, E. A. (1992)
Tsitologiya, 34, 67-73.
76.Lo, D. Y., Tein, M. S., Lau, T. K., Haines, C.
J., Leung, T. N., Poon, P. M., Wainscoat, J. S., Johnson, P. J., Chang,
A. M., and Hjelm, N. M. (1998) Am. J. Hum. Genet., 62,
768-775.
77.Paul, S. (1998) Appl. Biochem.
Biotechnol., 75, 13-24.
78.Paul, S. (1994) Appl. Biochem.
Biotechnol., 47, 241-255.
79.Sinohara, H., and Matsura, K. (1998) Proc.
“Catalytic and Super Antibodies”, University of New
Delhi, New Delhi, p. 36.
80.Glukhov, B. M., Jerusalemsky, R. P., and
Salganik, R. I. (1968) Zh. Nevrol. Psikhiatr., 10,
361-368.
81.Ramaswamy, H., Swamy, Ch. V. B., and Das, M. R.
(1993) J. Biol. Chem., 268, 4181-4187.