Mitochondrial Creatine Kinase: Properties and Function
T. Yu. Lipskaya
Department of Biochemistry, School of Biology, Lomonosov Moscow State University, Moscow, 119899 Russia; fax: (095) 939-3955
Received April 27, 2001; Revision received July 6, 2001
This review describes properties of mitochondrial creatine kinase from heart and skeletal muscle studied in the author's group at the Department of Biochemistry of Moscow State University. The results are compared to the data in the literature. The author's point of view on the physiological role of mitochondrial creatine kinase is presented.
KEY WORDS: mitochondrial creatine kinase, binding to membranes, oligomeric forms, physicochemical properties, physiological role
Abbreviations: CK) creatine kinase; mtCK) mitochondrial creatine kinase; Cr) creatine; CP) creatine phosphate; TAN) translocase of adenine nucleotides; DMSI) dimethyl suberimidate; DMPI) dimethyl pimelimidate; DMAI) dimethyl adipimidate; TSAC) transition-state-analog complex; GA) glutaric aldehyde; PCMB) para-chloromercuribenzoate; DTT) dithiothreitol.
S. E. Severin offered me the opportunity to engage in the study of mtCK
in the middle of the 1970s. This was only ten years after the discovery
of mtCK [1]. Even the first studies indicated that
the enzyme activity was sufficient to provide a high rate of oxidative
phosphorylation in mitochondria of skeletal muscles and of heart [2-4]. Mitochondria were concluded
to be the main place of CP synthesis in the tissues that had the
creatine kinase system. Concurrently an idea appeared on the existence
of intracellular metabolic, or functional, compartments [5] that had no detectable physical boundaries but
maintained complete or partial kinetic isolation [6]. Based on these concepts and on data of kinetics,
Saks et al. [7] supposed that the structural
interaction of mtCK and TAN in mitochondria provided a direct
translocation of ATP from the active site of TAN into the active site
of mtCK and the ADP translocation backward. As a result, the ATP
concentration increases and the concentration of ADP decreases in the
active site of mtCK compared to the environment, and this provides a
high rate of unidirectional synthesis of CP in mitochondria.
Obviously, the results of these first studies and hypotheses were very impressive. The virtual absence of data on the properties of mtCK and on its functional pathways opened a wide field for research. We chose as the object mitochondria of bovine heart and of pigeon pectoral muscle. This choice was reasoned by our wish to have sources of large amounts of mitochondria and of the enzyme. Moreover, the comparison of mtCK properties from heart and from skeletal muscles was promising for assessment of the universality of one or another mtCK property found.
STUDIES ON FORCES RESPONSIBLE FOR BINDING OF mtCK TO
MITOCHONDRIAL MEMBRANES
When we started our work, no general viewpoint existed on the character of the interaction forces between mtCK and membranes. Some data suggested a weak binding of mtCK to membranes by electrostatic interactions and its easy solubility [4, 8]; other data suggested that mtCK should be partially [9] or even completely [10] firmly bound to membranes. A third set of data suggested that mtCK should be an integral protein of the inner membrane [11]. Since mtCK is located on the external surface of the mitochondrial inner membrane [3, 4], we suggested that these discrepancies in the literature data were caused by different degrees of destruction of the outer mitochondrial membrane in the works of different authors.
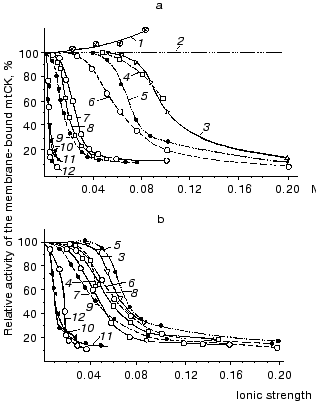
We degraded the mitochondrial outer membrane by hypotonic treatment with water. Figure 1 shows that the solubilizing effect of the substances studied (except adenine nucleotides) occurs at their different molar concentrations (Fig. 1a) but at similar values of ionic strength (Fig. 1b); with increase in the ionic strength, the effects of various substances converge [12]. At the physiological ionic strength of 0.2 only 8-13% of the mtCK activity remains bound to mitochondrial membranes. These experiments suggested that mtCK binds to membranes by forces of electrostatic interaction [12]. Figure 1 also shows that the solubilizing effect of adenine nucleotides is more pronounced, thus, their effect is likely to involve some more specific mechanisms than a simple saline effect. KCl solutions of different ionic strength were similarly effective in solubilization of mtCK from membranes of heart mitochondria and of skeletal muscles [13]. Our conclusion on the electrostatic binding between mtCK and mitochondrial membranes [12] was confirmed by more recent studies on mitochondria from other sources [14-16]. Saks et al. [17] found that not the ionic strength but the ionic composition of the medium was important for solubilization of mtCK. However, Brooks and Suelter [16] and our own further work [18] failed to reproduce these results. Thus, at present it seems that electrostatic binding of mtCK to mitochondrial membranes is a universal property of the enzyme.
We supposed that in the intact mitochondria under conditions of physiological ionic strength and substrate concentrations mtCK could be only partially solubilized into the intermembrane space because the volume accessible for mtCK was at least 100 times less than the volume admissible for mtCK in the experiment [12].Fig. 1. Effects of various substances on the mtCK fraction bound to membranes of bovine heart mitochondria. After hypotonic pretreatment, 9 mg mitochondrial protein was incubated at 0°C for 20 min in 2 ml of one of the below-mentioned media and precipitated by centrifugation. The precipitates were washed with water and finally suspended in 0.25 M sucrose. The mtCK activity was determined in the precipitates and in the supernatants. During the extraction of mtCK with ATP and MgATP solutions, the samples contained 170 µg of oligomycin [12]. 1) Cr; 2) sucrose; 3) imidazole (pH 6.6); 4) Tris-HCl (pH 8.3); 5) KCl; 6) NaCl; 7) potassium phosphate (pH 6.9); 8) CP (pH 7.9); 9) MgCl2; 10) ADP (pH 7.0); 11) MgATP (1 : 1) (pH 7.9); 12) ATP (pH 6.9).
STUDIES ON THE CHEMICAL NATURE OF MITOCHONDRIAL MEMBRANE
COMPONENTS WHICH BIND mtCK
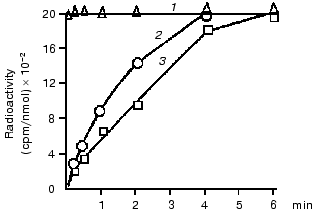
The hypothesis of an interaction of mtCK with TAN in membranes was checked using isotopes, and the hypothesis was supported by some findings [19] and contradicted by others [20]. Therefore, our first experiments were also concerned with the problem of structural coupling of the two proteins. We found that a voluminous inhibitor of TAN, carboxyatractylozide, had no effect on the solubilization of mtCK from mitochondrial membranes [12]. Other authors obtained similar results [16, 21]. In our experiments with isotopes [22], the incubation medium contained unlabeled ATP and Cr and labeled Pi. During oxidative phosphorylation the gamma-phosphate-labeled ATP was from the labeled phosphate. Our findings (Fig. 2) suggested that the labeled ATP was mixed with ATP of the medium before its entrance to the active site of mtCK, i.e., there was no structural coupling between mtCK and TAN. Later we returned to this problem in work performed in the laboratory of S. P. Bessman with a phosphate analyzer providing automated determination of phosphate and radioactivity in each fraction eluting from an ion-exchange column. The result was confirmed with rat heart mitochondria--at ATP concentration in the medium of ~500 µM the ATP which came immediately from TAN during oxidative phosphorylation contributed no more than 10-12% to the synthesis of CP [23].
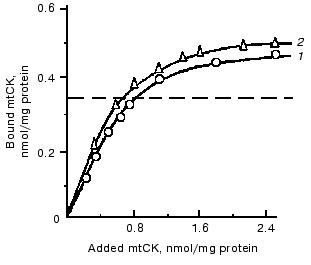
To get additional evidence of the absence of structural coupling between mtCK and TAN, the binding of mtCK to membranes of heart and liver mitochondria was studied [12]. In the experiments, a mtCK-containing extract from heart mitochondria was used with the ionic strength decreased by dialysis. Gel-filtration data showed that the mtCK in the extract was a dimer [12]. Figure 3 shows that the amount of mtCK bound to heart mitoplasts previously deprived of this enzyme is comparable to the enzyme amount bound to liver mitoplasts, although the initial liver mitochondria contained no mtCK and the content of TAN was fourfold lower than in the heart mitochondria [24]. Considering similar phospholipid composition of heart and liver mitochondrial membranes [25, 26], we suggested that mtCK should be bound to membrane phospholipids of the heart mitochondria by electrostatic forces. Similar results were concurrently obtained in the other work [27], but the authors presented no hypotheses on the chemical nature of forces binding mtCK to membranes. In our experiments excess of cytochrome c which could bind non-specifically to mitochondrial membrane phospholipids suppressed binding of mtCK to heart mitochondria membranes [12].Fig. 2. Changes in the specific radioactivity of ATP and CP during CP synthesis coupled with oxidative phosphorylation in bovine heart mitochondria. The samples of 2 ml volume contained 0.25 M sucrose, 10 mM Tris-HCl (pH 7.4), 1 mg/ml BSA, 5 mM potassium phosphate, 17 µCi 32Pi, 5 mM potassium glutamate, 2 mM potassium malate, 0.5 mM ATP, 10 mM Cr, 0.3 mM DTT, and 3.3 mM MgCl2. The reaction was initiated by addition of 1 mg mitochondrial protein. The temperature was 30°C. Adenine nucleotides, Cr, and Pi were separated by two-dimensional thin-layer chromatography on Silufol plates; ATP and CP concentrations were determined at 340 nm using the coupled enzyme system. 1) Pi; 2) ATP; 3) CP [22].
To check the hypothesis of the interaction of mtCK with phospholipids, we studied the binding of mtCK to liposomes prepared from the mixture of phospholipids extracted from heart mitochondria. The binding of mtCK to the liposomes was characterized by a curve with saturation, and the maximum binding was about 10 nmol mtCK dimer per µmol phospholipid phosphorus [28].Fig. 3. Binding of mtCK to mitoplasts of rat liver (1) and of bovine heart (2). After the hypotonic pretreatment, mtCK was extracted from bovine heart mitochondria with 0.2 M NaCl taken so as to be 1 ml per 5 mg protein. The extract was dialyzed against 0.1 M sucrose with 0.25 mM DTT. Washed free from NaCl, the mitoplasts were suspended in 0.1 M sucrose with DTT to the concentration of 10 mg protein/ml. To 1-2 mg protein of the mitoplasts, 1-7 ml of the dialyzed extract was added, and the mixture was incubated for 20 min at 0°C and centrifuged. The precipitates were washed with water, and the mtCK radioactivity was determined in them and in the supernatants. Mitoplasts from rat liver were obtained similarly to mitoplasts from bovine heart. The dotted line shows the mtCK content in the initial heart mitochondria. By the findings of work [12].
Considering the electrostatic interactions of mtCK with mitochondrial membranes and the alkaline character of the enzyme [29], we suggested that positively charged groups on the surface of the mtCK molecule (amino groups of lysine or arginine residues) should interact with negatively charged phosphate groups of mitochondrial membrane phospholipids [12, 28]. To check this hypothesis and to assess the character of functional groups involved in the mtCK binding to mitochondrial membranes, we studied the effect of pH on the binding of mtCK from heart mitochondria to membranes and also the pH stability of this enzyme in solution. It was found that if the ionic strength of buffers was insufficient for solubilization of mtCK (10-20 mM acetate buffer, imidazole, and Tris-HCl buffers), the enzyme binding to mitochondrial membranes was resistant within the pH range from 5 to 9.5 [12], which was comparable to the range of pH stability of mtCK in solution (60 min at 30°C) [28]. These findings indicate that functional groups of mtCK with pK close to neutral pH values (imidazole ring of histidine residue, sulfhydryl group of cysteine residue) are not involved in the mtCK binding to mitochondrial membranes. Thus, these findings do not contradict the concept of the binding of mtCK to phospholipids because they show that the binding occurs with the involvement of functional groups with 10 < pK < 5. Consequently, the acid group might well be phosphate of phospholipid (pK = 1-2 [30]). Similar data on stability to pH changes were obtained for mtCK from human heart [31]. Results inconsistent with our data were obtained in the work of Vial et al. [32]: they found the solubilizing effect of 20 mM phosphate to depend on pH within the range from 6 to 8. However, our special experiments have shown that in this case changes in the mtCK solubilization were produced not by changes in pH but by changes in the ionic strength of phosphate solutions at different pH values [12]. Based on data on mtCK solubilization by organic compounds of mercury, Font et al. [14] concluded that SH groups of mtCK were involved in the binding of the enzyme to mitochondrial membranes. The inaccuracy of this conclusion was shown in more recent studies [15, 21].
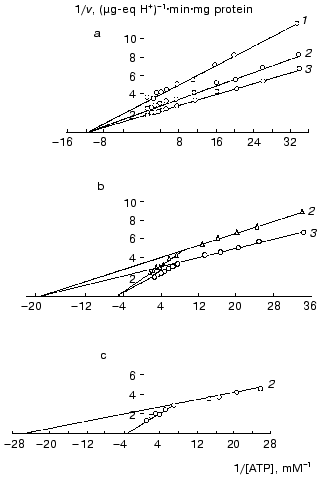
To obtain additional evidence for the binding of mtCK in mitochondria with membrane phospholipids, we studied the effect of binding of mtCK to mitochondrial and liposomal membranes on the creatine kinase reaction kinetics [28, 33-36]. Figure 4a shows that the kinetics of mtCK in mitochondrial extract is described by the Michaelis-Menten equation: KmMgATP = 0.08 mM, KmCr = 7.0 mM, Vf = 0.742 µg-eq H+/min per mg protein of the initial mitochondria [28, 35]. However, two kinetic forms were found for mtCK bound to membranes of mitochondria (Fig. 4b) and of liposomes (Fig. 4c), with similar parameters for both types of membranes. For mtCK in mitochondria: (Km)1MgATP = 0.034, (Km)2MgATP = 0.315 mM; (Km)1,2Cr = 7.0 mM, (Vf)1 = 0.410, and (Vf)2 = 0.381 µg-eq H+/min per mg protein [28, 35]; for mtCK in liposomes: (Km)1MgATP = 0.048, (Km)2MgATP = 0.561 mM; (Vf)1 = 0.387, and (Vf)2 = 0.363 µg-eq H+/min per mg protein of the initial mitochondria [33, 36] (the results were processed as described in [37]). Thus, the membrane-bound forms differ from mtCK in solution in Michaelis constants for MgATP; the maximum rates of both forms are approximately the same, each of them being half of the maximum rate in the extract. The cause of these differences will be discussed later, but it is important to note here that the binding to the phospholipid membrane of liposomes resulted in the same changes in kinetic characteristics of mtCK in the forward reaction as the binding to the protein-phospholipid membrane of mitochondria. The conclusion on the binding of mtCK to phospholipids was more recently supported in several laboratories: mtCK was shown to interact with the acidic phospholipid cardiolipin [21, 38, 39]. Involvement in mtCK binding to membranes was also shown for lysine and arginine but not for histidine and cysteine residues of the enzyme [40].
Fig. 4. Primary double-reciprocal plots for the forward creatine kinase reaction at the following fixed concentrations of Cr (mM): 1) 5; 2) 10; 3) 25. The samples of 4-4.5 ml volume contained 0.25 M sucrose, 1-2.5 mM Tris-HCl (pH 7.4), 10 mM KCl, 3.3 mM MgCl2, 0.25 mM DTT, ATP, and Cr. The temperature was 30°C. During the activity determination in the mitochondria suspensions, the samples also contained oligomycin (2.5-4 µg/ml) and rotenone (10-6 M). In experiments with liposomes, the reaction mixture was supplemented with excess liposomes and the extract from mitochondria was dialyzed against 0.1 M sucrose with 0.25 mM DTT; the mixtures were kept for 5 min, and the reaction was initiated by addition of MgATP. a) Extract from mitochondria [28, 35]; b) mitochondria [28, 35]; c) liposomes [36].
STUDIES ON PHYSICOCHEMICAL PROPERTIES OF OLIGOMERIC FORMS OF mtCK FROM HEART AND FROM SKELETAL MUSCLE
Our previous studies suggested that under physiological conditions mtCK should be only partially bound to membranes; therefore, in the next stage of our work we studied mtCK properties in solution. The enzyme was known to exist in solution as a mixture of at least two oligomeric forms [29, 41]. Our purpose was to elucidate what multiple forms of mtCK can exist in solution and what factors can influence mutual transitions of the forms. Bovine heart mitochondria were isolated by a conventional procedure [42], and we selected conditions for isolation of large amounts of mitochondria from pigeon pectoral muscle [13]. For isolation and purification of mtCK from mitochondria of skeletal muscles and heart a unified procedure was elaborated [13, 43] that allowed us to prepare mtCK with specific activity of about 300 units/mg protein.
Oligomeric composition and quaternary structure of mtCK. The properties of mtCK from skeletal muscles and from heart were compared [13, 43] and were found to be very similar, with merely quantitative differences (Table 1). Table 1 shows that both enzymes in solution have two oligomeric forms: dimer and octamer. We did not find intermediate forms either by gel filtration or by ultracentrifugation in sucrose density gradient, though the resolution of the latter was sufficient for detection of intermediate forms. Other authors later arrived at the same conclusion [44, 45].
Table 1. Quantitative characteristics of
oligomeric forms of mtCK from bovine heart and from pigeon pectoral
muscles
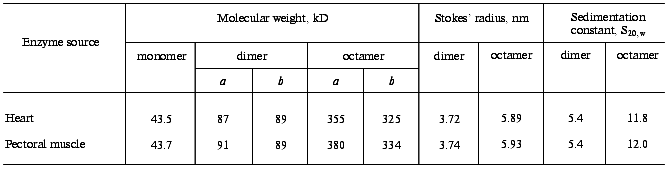
Note: a) by results of gel filtration on Sephacryl S-300;
b) by value of sedimentation constant [13,
43].
For a long time there was no general opinion in the literature about the number of subunits in the high-molecular-weight mtCK oligomer from bovine heart. It was reported to be an octamer [46], a hexamer [47, 48], and also to concurrently exist as both octamer and hexamer [49]. Based on our earlier findings [12] by gel filtration on Sephadex G-200, we believed this oligomer to be a hexamer, but gel filtration on Sephacryl S-300 and ultracentrifugation in a sucrose density gradient (Table 1) suggested that it should be an octamer. To more precisely determine the number of subunits in this high-molecular-weight mtCK oligomer from bovine heart, we cross-linked the subunits in the enzyme molecule with DMSI and then performed electrophoresis in the presence of SDS. The conditions were chosen to provide a sufficient depth of cross-linking. In this case the number of protein bands on electrophoresis corresponded to the number of subunits in the oligomer. In our experiments there were eight protein bands, and an increase in the time of cross-linking resulted in the disappearance of low-molecular-weight bands and in the accumulation of protein in the seventh and eighth lanes [50]. It was shown that all eight lanes were produced due to cross-linking only of intra-oligomeric contacts [50]. The high-molecular-weight mtCK oligomer from other sources was also shown to be an octamer [44, 45]. It was also found that mtCK from chicken brain, which exists in solution as a mixture of dimer and octamer, was different from mtCK from heart and skeletal muscle in some physicochemical properties and was a separate isozyme [51]. Thus, the octameric structure is a specific feature of mtCK.
To elucidate the spatial location of the subunits in the mtCK octamer from bovine heart, we used cross-linking with bifunctional reagents [50]. For the cross-linking bifunctional reagents of a homologous series were used as follows: DMSI, DMPI, DMAI. These experiments lead to the conclusion that the mtCK octamer has a quasi-spherical configuration. This conclusion was supported by data of electron microscopy [50]. The electron microscopy of mtCK from bovine heart was analyzed by Belousova et al. [52] and of mtCK from chicken heart and brain by Schlegel et al. [51] and also by Schnyder et al. [45]. The octamer from every source has a central cavity and forms bilayer structures built from four dimers providing the involvement of each dimer in the formation of both the upper and lower layer. In the octamer from bovine heart the layers are turned relative to each other [50, 52], whereas the mtCK octamer from chicken is cube-like in shape [45, 51], and this is the main difference. X-Ray analysis provided new details about the mtCK structure [53-55]. Thus, it seems that the octameric structure of mtCK from different sources can be somewhat different.
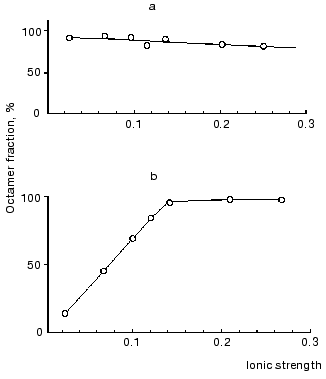
To determine what forces are responsible for the binding of dimers in the octamer, we studied the effect of ionic strength on the distribution of molecular forms. For this, mtCK samples from bovine heart mitochondria and from pigeon pectoral muscles were dialyzed against medium with ionic strength ~0.025, then separate portions of the dialyzed enzymes were supplemented with KCl to provide the required ionic strength; some hours later the samples were ultracentrifuged in a sucrose density gradient. Figure 5 shows that decreasing ionic strength caused a reversible dissociation of the mtCK octamer from pectoral muscles (Fig. 5b) [13] and had no effect on the dissociation of the mtCK octamer from heart (Fig. 5a) [43]. However, mtCK from bovine heart also dissociated at lower concentrations of the protein (~50 µg/ml) and at lower ionic strength of the solution [56]. Elevating the temperature from 5 to 20°C insignificantly displaced the equilibrium towards the octamer [43]. These findings suggested that electrostatic interactions should not play an important role in the association of dimers into octamers because in the case of electrostatic interactions the binding between the subunits should be weakened with an increase in ionic strength and not with a decrease in it [13, 43]. Later, hydrophobic interaction of dimers in the octamer was also established for mtCK from other sources [57].
Fig. 5. Effect of ionic strength on dissociation of the mtCK octamer from bovine heart (a) and from pigeon pectoral muscles (b) [13]. The sucrose gradients (2-10%) contained 25 mM Mes (pH 7.0), 0.25 mM DTT, and KCl in the amount sufficient for the ionic strength values shown on the abscissa. The concentration of the mtCK protein after dialysis (µg/ml): 480 (a), 450 (b). The octamer content in the preparation before the dialysis was taken as 100%. Other details are in the text.
Factors determining the distribution of oligomeric forms. The effects of probably physiologically important factors such as ionic composition of the medium, pH, substrates, and some metabolites were studied.
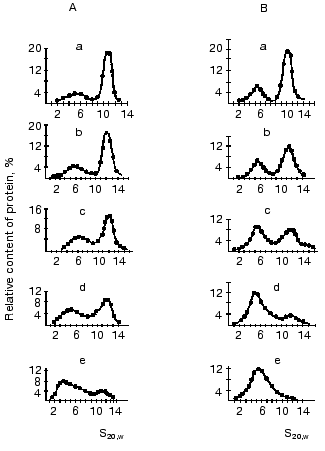
Ultracentrifugation in sucrose density gradient showed that the character of univalent anions (Cl-, NO3-, acetate) and cations (K+ or Na+) failed to affect the distribution of oligomeric forms of mtCK from two sources [13, 43]. However, an increase in pH enhanced the dissociation of octamers. Figure 6 shows that at physiological pH values the enzyme from pigeon pectoral muscles (Fig. 6B) was in solution as the mixture of dimer and octamer, whereas mtCK from bovine heart (Fig. 6A) was present mainly as the octamer. At pH 9.6 mtCK from pectoral muscles completely dissociated to dimers [13], whereas in mtCK from heart muscle at pH values within the range 9.0-9.6 a form appeared with the sedimentation constant of 3.0-3.5 S, which seemed to be a monomer [43]. The decrease in the activity of mtCK from bovine heart at pH values higher than 9.5 observed in studies of the enzyme's stability is suggested to be due to dissociation of the enzyme dimers to monomers.
At pH 8.1 the octamer constitutes about 50% of all mtCK forms from bovine heart [43] and at pH 7.3 of mtCK from pectoral muscles [13]. The comparison of pH-dependence of the octamer dissociation to the finding of nonionic interactions of dimers in the mtCK octamer suggests that SH-groups should play an important role in the region of dimer contacts. Actually, at acidic pH values SH-groups are protonated and do not carry a charge; therefore, nothing prevents the association of dimers into octamers. With increase in pH the dissociation degree of SH-groups increases, and in the region of dimer contacts the negative mercaptide ions appear; this results in the dissociation of octamers. All other functional groups of the protein (carboxy, amino, imidazole groups) that can dissociate within the pH range studied are charged at acidic values of pH; therefore, their involvement in the dimer contacts seems unlikely. A significant effect of the cysteine SH-group on the stability of the interdimer contacts was shown in experiments when the replacement of Trp264 (which is suggested to be a part of the hydrophobic interdimer contact region) by cysteine dramatically increased the dissociation of octamers in the presence of TSAC and the sensitivity of octamers to the presence of salts (cited after [53]). However, the pH-dependence of the dissociation of octamers could reflect conformational changes in the whole octamer molecule under the influence of pH, which would also affect the stability of interdimer contacts.Fig. 6. Effect of pH on the distribution of oligomeric forms of mtCK from bovine heart (A) [43] and from pigeon pectoral muscles (B) [13]. Sucrose gradients (7-20%) contained 0.2 M KCl, 0.25 mM DTT, and either of the following buffers (0.05 M): a, b) Mes (pH 6.0 and 7.0, respectively); c, d) Tris-HCl (pH 8.0 and 9.0, respectively); e) glycine buffer (pH 9.6). The initial concentration of mtCK (µg/ml): 600 (A), 540 (B).
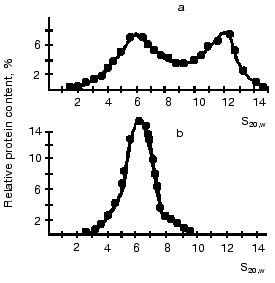
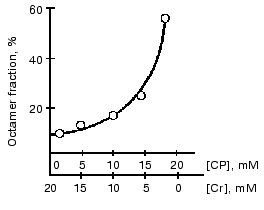
Substrates of the creatine kinase reaction taken separately had no effect on the distribution of oligomeric forms of the enzyme from bovine heart and from pigeon pectoral muscles, whereas an equilibrium mixture of the substrates caused the dissociation of octamers [13, 43] (Fig. 7). The substrate effects were associated with conformational changes in the active site of mtCK during catalysis: the octamers did not dissociate when the medium was deprived of Mg2+, which is a required cofactor of the creatine kinase reaction; however, the dissociation occurred during the generation of TSAC [58] that imitated the transition state of the reaction. TSAC was produced in the presence of Mg2+, of a non-interacting pair of substrates (ADP and Cr), and of certain anions, Cl- or nitrate but not of acetate [13, 43]. At physiological concentrations of adenine nucleotides the octamer dissociation degree depended on the ratio of Cr and CP concentrations and decreased with an increase in the CP concentration (Fig. 8) [43].
Fig. 7. Effect of substrate mixture on the distribution of oligomeric forms of mtCK from pigeon pectoral muscles [13]. Sucrose gradients (7-20%) contained (a and b): 0.12 M KCl, 20 mM Tris-HCl (pH 8.0), 4.5 mM MgCl2, and also (b): 0.1 mM ADP, 3 mM ATP, 10 mM Cr, 3 mM CP. The initial concentration of mtCK was 320 µg/ml.
The substrate effects on the dissociation of octamers depended on pH. At pH 6.0 (compared to pH 7.0) the octamer fraction was increased at any ratio of Cr and CP concentrations [43]. In the case of mtCK from pigeon pectoral muscle the effect was less pronounced than in the case of mtCK from bovine heart and required the presence of Cl- [13]. Pi suppressed the dissociation of octamers in the presence of ATP and of high concentrations of Cr, but this suppression was less pronounced than the suppression by CP [13, 43]. Other regulators of energy metabolism studied by us (AMP, NAD+, NADH, and citrate) at physiological concentrations had no effect on the distribution of oligomeric forms [59]. Soon after our study, works of other laboratories on mtCK from other sources were published [44, 60] that supported our data on pH and substrate effects on the distribution of the oligomeric forms of the enzyme. It seems that the sensitivity of the octamer <--> dimer transition to pH and to the presence of substrates is a general property of all mtCK.Fig. 8. Effect of CP/Cr ratio on the distribution of oligomeric forms of mtCK from bovine heart [43]. Sucrose gradients (2-10%) contained 0.15 M CH3COOK, 50 mM Mes (pH 7.0), 0.25 mM DTT, 10 mM Mg(CH3COO)2, 3 mM ATP, and the CP and Cr concentrations shown on the abscissa. The octamer fraction in the substrate-free medium is taken for 100%. The initial concentration of mtCK was 333 µg/ml.
Kinetic properties of oligomeric forms. When we approached this problem, the kinetic properties of mtCK from various sources in solution were already described by some authors [31, 48, 61-63] and also by us [28, 33-36]. However, the relationship between the kinetic parameters and the oligomeric composition of mtCK remained unclear. To determine kinetic parameters of the octamer and dimer, it was necessary, first, to obtain them in the individual states, and, second, to be sure that even a partial dissociation of the octamer to dimers did not occur during the study of kinetics. For the octamer preparation we used freshly isolated mtCK from bovine heart that at pH 7.4 and about 2 mg/ml protein contained 10-15% dimer by data of ultracentrifugation in a sucrose density gradient. To prepare the dimer, mtCK from bovine heart was preincubated with saturating concentrations of substrates (ADP and Cr in the presence of Cl-) that resulted in the generation of TSAC. In preliminary experiments different concentrations of mtCK were preincubated with this substrate mixture for 18-20 h and then placed onto a sucrose density gradient without the substrates or subjected to gel filtration in an FPLC system with substrate-free buffer as the eluent. The Kdis value was calculated from results of each experiment. The Kdis values obtained by two methods at different concentrations of mtCK were in a good agreement [64]. Using this Kdis value, we obtained the equation for the dependence between the total protein concentration in the mtCK preparation from bovine heart and the dimer content in it. At mtCK protein concentration of 78 µg/ml the dimer fraction was 95%. Based on these calculations, to obtain dimer preparations the mtCK concentration was 75 µg/ml [64]. It was also shown that during the kinetic experiment (~1 min) the octamer did not dissociate under the influence of substrates [64]. This conclusion was supported by data of other authors about the rate of the dissociation of octamer in the presence of substrates [44, 60].
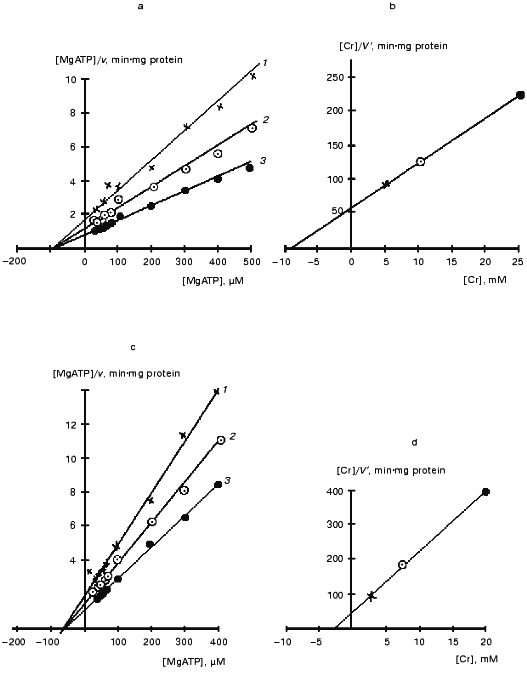
Figure 9 presents kinetic curves obtained in a certain experiment for the mtCK octamer and dimer during the forward reaction. Similar curves were also obtained for the reverse reaction. It should be noted that although the concentration of mtCK protein varied in the initial solutions of dimers and octamers, the incubation medium contained the same amount of the enzyme (~1 µg per 4 ml to start the reaction) during the determination of kinetic parameters. The overall data are presented in Table 2. The kinetics of mtCK octamer and dimer are described by the Michaelis-Menten equation. The catalytic mechanism of the reaction of the octamer and dimer of mtCK from bovine heart was the same as the mechanism found for the cytoplasmic CK [65] and for mtCK from rat heart [7]. Figure 9 (a, c) shows that the kinetic curves intersect at one point on the abscissa. This corresponds to a kinetic mechanism with independent substrate binding when Ks for all substrates are equal to corresponding Km values [7]. Preincubation of the octamer with only ADP or Cr had no effect on the kinetic parameters of the octamer. Equilibrium constants of the creatine kinase reaction (0.016-0.017) calculated using Haldane'sequation are in good agreement with the value of 0.01 found in the equilibrium experiments at pH 7.4 [66].
Table 2. Kinetic parameters of octamer and dimer of mtCK from bovine heart [64]Fig. 9. Kinetic curves for the octamer (a,b) and dimer (c, d) of mtCK from bovine heart [64]. a, c) Primary plots of the [MgATP]/v versus[MgATP] at the following fixed concentrations of Cr (mM): a) 5 (1), 10 (2), 25 (3); c) 3 (1), 7.5 (2), 20 (3); b, d) secondary plots of the [Cr]/V´ dependence on [Cr]. V´ was calculated from the ordinate intercepts of the primary plots (a) and (c), respectively.

*Kapp = Vf·KmMgADP·KmCP/Vr·KmMgATP·KmCr. The results were processed using Student's t-test. Values p <= 0.05 were considered statistically significant. In parentheses, the number of experiments. All results are presented as mean ± SD. Protein was determined at 280 nm (A1%280 = 11.7 [31]).
Isoelectric points of oligomeric forms. Studies preceding our work [29] showed the isoelectric point of mtCK from bovine heart to be within the pH range from 9.2 to 9.6; however, it was not known what oligomeric forms were responsible for these values. Isoelectric points of oligomeric forms were determined by isoelectric focusing in polyacrylamide gel rods. To solve this problem, we used the pH-sensitivity of the equilibrium between dimer and octamer and also the different dissociation degree of the mtCK octamer from heart and skeletal muscles at the low ionic strength characteristic for experiments with isoelectric focusing. Three mixtures of ampholytes were chosen with initial pH values of 6.0, 7.5, and 9.6. After the isoelectric focusing, these mixtures provided pH gradients of 3.0-9.6, 3.9-10.4, and 7.6-10.9, respectively. mtCK was introduced into the ampholyte mixtures prior to the gel polymerization, and at pH 7.5 the dimer and the octamer were expected to be the main oligomeric forms for mtCK from pectoral muscles and from heart, respectively; at pH 9.6 the dimer content was expected to increase in mtCK from heart, and at pH 6.0 the octamer was expected to appear in the enzyme from pectoral muscles. Using this approach, the pI values for the octamer and dimer of mtCK from heart and skeletal muscles were obtained as follows: 8.93, 8.91 and 9.67, 9.56, respectively [56]. Thus, the isoelectric points of both oligomeric forms of mtCK are at alkaline pH values and virtually coincide for the corresponding forms from the two sources. The isoelectric points of oligomeric forms presented in the literature are also at alkaline pH values [67, 68].
To end the section concerning properties of mtCK oligomeric forms in solution it should be emphasized that the effects caused by the exposures studied were reversible. This clearly demonstrates that the dimer and octamer in solution are in the state of dynamic equilibrium. Thus, the dimers generated in the medium with the low ionic strength reassociate as the ionic strength of the solution increases (Fig. 5). The effect of pH is also reversible; the dissociation degree of the octamer during the generation of TSAC and in the absence of substrates depends on the protein concentration and is determined by the equilibrium constant of the dissociation.
IDENTIFICATION OF OLIGOMERIC FORM OF mtCK BOUND TO MITOCHONDRIAL
MEMBRANES AND DETECTION OF DIFFERENCES IN PROPERTIES OF SITES OF ENZYME
LOCATION ON THE MEMBRANES
The isoelectric points of the dimer and octamer suggested that both forms should be bound to membranes. Nevertheless, we allowed for changes in the oligomeric composition of mtCK because of the binding to membranes. Thus, it seemed probable, for example, that the interaction with a membrane would stabilize a certain intermediate form of the enzyme, e.g., a tetramer, or that several octamers on the membrane would combine into higher-molecular-weight aggregates similar to those generated during the precipitation of mtCK [47]. It might further be supposed that the dimer and octamer should be forms instantly produced from mtCK during its solubilization from the membrane. To identify the membrane-bound oligomeric forms of mtCK, we used a “cross-linking” bifunctional reagent, GA, which at neutral pH values could irreversibly cross-link NH2-groups within seconds, and this allowed us to record a particular state of mitochondrial membranes and of the enzyme [69-71]. Moreover, GA can cross-link NH2-groups located at various distances because in the cross-linking monomers, dimers, and trimers of GA are involved. The cross-linking was performed in 0.25 M sucrose when all mtCK was bound to the mitochondrial membranes. We suggested that the cross-linking should stabilize the oligomeric forms that were present on the membranes at that instant, even if they were nonresistant on solubilization in the absence of cross-linking. After the cross-linking, the mitochondria were washed from GA and the membranes were broken by ultrasonication in 0.25 M KCl and then precipitated at 230,000g. The mtCK activities were approximately equally distributed between the precipitate and supernatant, whereas in the absence of cross-linking 95% of the mtCK activity was extracted from the mitochondria. The supernatants were concentrated, and the oligomeric forms in them were analyzed by ultracentrifugation in sucrose density gradient. At pH 7.4 the same oligomeric forms were found in the mitochondrial extract after the cross-linking with GA as in the control extract [72], with the sedimentation constants of 5 and 12 S, i.e., the dimer and the octamer. On placing onto a sucrose density gradient containing the reaction substrates, the control mtCK dissociated to dimers completely, while after the cross-linking the enzyme dissociated only partially. Preincubation at pH 5.5 resulted in a partial transition of the dimer to the octamer, and this association occurred in both the control extract and the extract after the cross-linking with GA without a noticeable difference. If the dimer were present on the membranes during the cross-linking, the cross-links on its surface would make difficult the association of the dimer into the octamers. It is likely that the dimer detectable by sucrose density gradient was generated from the octamer incompletely linked by GA already after the passage of this octamer into the solution during the ultrasonication of mitochondria and the subsequent ultracentrifugation in the density gradient. It might be expected that the cross-linked octamer complexes should be precipitated at 230,000g. However, the mtCK activity distribution in the precipitates after a stepwise centrifugation of the ultrasonicated mitochondria suspension corresponded to the distribution of the marker enzyme of the inner membrane, cytochrome oxidase; consequently, the part of mtCK which had been precipitated was bound to mitochondria [72]. Data on kinetics indicated that the fraction of mtCK non-extractable from mitochondria during the ultrasonication had the same oligomeric composition as the extractable enzyme: both the total fraction and the non-extractable mtCK displayed complicated kinetics (as in Fig. 4b), and the Michaelis constants were coincident [72]. Thus, this part of our work confirmed the presence of octamers on the mitochondrial membranes during the cross-linking. We did not detect any other oligomeric form.
Because in the medium with high ionic strength the equilibrium between the mtCK octamer and dimer is established slowly, we analyzed contents of the mtCK oligomeric forms in the extract from bovine heart mitochondria (without preliminary cross-linking with GA) by rapid HPLC gel chromatography. The freshly prepared extract contained only the octamer of mtCK, but after storage for 22 h a certain amount of the dimer appeared corresponding to the equilibrium distribution of the two forms in solution [72]. In our experiments, the extract from mitochondria contained 80-85% of the mtCK activity; consequently, at least 80-85% of the mtCK in mitochondria isolated in 0.25 M sucrose should be the octamer. The conclusion that the octamer is the main oligomeric form bound to mitochondrial membranes was simultaneously arrived at by French authors [60, 73] who used other approaches and other sources of mitochondria (pig heart and rabbit heart). More recently, similar results were obtained on mitochondria from chicken heart [68].
The increased content of mtCK in the precipitate of GA-pretreated mitochondria was shown to be unrelated with physical barriers preventing the enzyme passage into solution but to be caused by the cross-linking of mtCK to membranes. The cross-linking of mtCK at the same concentration of GA within varied time intervals and also the cross-linking at increasing GA concentrations within a constant time interval suggested that the maximum quantity of the enzyme which could be cross-linked to the membranes was 45-50% of the total mtCK in the mitochondria [74]. The incomplete cross-linking of mtCK with membranes was not caused by different accessibility of individual binding sites of the enzyme for the cross-linking reagent because the hypotonic pretreatment which cause mitochondrial swelling and the unfolding of crista membranes failed to increase the cross-linked fraction of mtCK [74]. Taking into account that under experimental conditions only one oligomeric form of mtCK (octamer) was bound to mitochondrial membranes, that the GA-produced bonds were resistant, and that all binding sites of mtCK were accessible for the cross-linking reagent, we concluded that the incomplete cross-linking of the enzyme with the membranes was due to a difference in the environment of the binding sites [74]. Thus, the binding sites of mtCK can be located in the cristae and on the inner surface membrane [44, 76, 77], which are different in their structure [75].
Why does the binding of octamers to mitochondrial membranes induce subunits that are kinetically equivalent in solution (Figs. 4a and 9a) to lose this property? We know that the mtCK octamer consists of two tetramers. The interaction with the inner membrane is likely to occur through one of them, and this results in the appearance of differences in kinetic features of the subunits bound and not bound to the membrane. This point of view is supported by our findings of similar changes in the enzyme kinetics also during the interactions of mtCK with liposomal membranes [33, 36]. The surface charge of membranes is known to affect the value of Km for the charged substrates of enzymes bound to these membranes [78].
Thus, the octamer is the main oligomeric form of mtCK in freshly isolated mitochondria from various sources. Can the dimer bind to membranes? In our study on the binding of mtCK to mitoplast membranes, we found that both oligomeric forms could bind to the membranes [12]. The number of binding sites was the same for both forms, although the dimer was bound less firmly than the octamer [12]. In our experiments, the dimer also bound to liver mitochondria [12]. More recently, we found that this also took place in the case of mtCK dimer from pigeon pectoral muscles [79]. The literature data are inconsistent. Thus, by data of Marcillat et al. [60], only the octamer could bind to heart mitochondria at neutral and alkaline pH values. Schlegel et al. [68] found that both oligomeric forms and only the octamer could bind at neutral pH values and at pH 8.1, respectively, whereas at pH 8.8 mtCK did not bind to membranes at all. The last finding contradicts data presented in [60] and our finding on stability of the mtCK binding to membrane of bovine heart mitochondria within the pH range from 5.0 to 9.5 [12]. This corresponds to the range of the pH-stability of the enzyme in solution [28] and also to data on the involvement of amino groups of lysine and arginine [40, 55], which have pKa higher than 10, in mtCK binding to membranes. These discrepancies are suggested to be due to different approaches. Thus, Marcillat et al. [60] used a reversible inactivation of mtCK with PCMB solution that was likely to induce changes in the quaternary structure of the dimer. Schlegel et al. [68] obtained mitoplasts using an alkaline phosphate solution (pH 8.8) that could destabilize the inner membrane structure in the absence of the bound enzyme. Schlegel et al. [68] found that at pH 7.0 the octamer fraction was increased in the extract from mitoplasts to which the dimer was preliminary bound. Thus, the overall findings described above suggests that at physiological pH values both mtCK octamer and dimer should bind to mitochondrial membranes and that the equilibrium of these two forms on the membrane surface should be strongly displaced towards the octamer. This is responsible for the predominance of the octamer among oligomeric forms found in mitochondria [60, 68, 72, 73].
Thus, what do we know about the conditions for the functioning of mtCK in mitochondria? Our findings suggest that mtCK should be bound to mitochondrial membranes by electrostatic forces and that in vivo it should be membrane-bound in mitochondria and in the free state in the intermembrane space. Data on the binding of mtCK to membranes of heart and liver mitochondria and similar changes in kinetic properties of the enzyme during its binding to mitochondrial and liposomal membranes indicate that the structural elements of membranes which bind mtCK are phospholipids. Data on the pH-stability of the enzyme binding to mitochondrial membranes and also values of isoelectric points suggest that mtCK should interact with negatively charged groups of the membranes. Our experiments with isotopes indicate that direct nucleotide exchange with TAN is not necessary for functioning of mtCK.
Studies on properties of mtCK from heart and skeletal muscles show that in solution the enzyme exists as a mixture of only two oligomeric forms, dimer and octamer, and that these forms of the enzyme from the two sources have very similar properties. The octamer has a quasi-spherical structure and consists of two tetramers. The equilibrium between the dimer and the octamer can be changed under the influence of physiologically important factors such as pH, the substrates, and Pi. Both oligomeric forms can bind to mitochondrial membranes; the binding to membranes does not produce new oligomeric forms of mtCK but displaces the equilibrium towards the octamer. Mitochondrial membranes have two types of mtCK-biding sites that are different in their environment. The mtCK activity is distributed between these locations at the ratio of 1 : 1. These locations are likely to be on the cristae membranes and on the inner surface membrane. The dimer and octamer are different in their kinetic properties. According to calculations, at low concentrations of ADP characteristic for the rest state of muscle tissue, the catalytic effectiveness of the dimer is higher than that of the octamer. This difference decreases with increasing ADP concentration. The binding to mitochondrial membranes changes the kinetic parameters of the octamer, but these changes only slightly affect its catalytic effectiveness. The decrease in Km for MgATP during oxidative phosphorylation suggests that during this process the mtCK octamer is partially released from the membranes and/or dissociates into dimers in the intermembrane space. In the absence of oxidative phosphorylation, Vmax of mtCK in mitochondria is three times higher than during the oxidative phosphorylation, when both maximal rates are equal [23]. It is known that the oxygen uptake by rat heart at rest is less than 10% of the maximum and at moderate loads it is 25-40% of the maximum (references in [80]). All this suggests that during oxidative phosphorylation in vivo mtCK catalyzes the reaction close to the equilibrium state. This is also supported by findings on transgenic mice deprived of MM-CK [81, 82]. It seems that the existence of two oligomeric forms of mtCK maintains the reaction close to the equilibrium state over a wide range of physiological conditions. Unlike the dimer, the mtCK octamer can interact with the outer membrane of mitochondria [68, 83] and, thus, can be involved in the formation of contact sites between two surface membranes [53] and, possibly, in the regulation of permeability of the outer membrane for metabolites [84, 85]. Thus, we now have much knowledge about the conditions under which mtCK functions in heart and skeletal muscle mitochondria. Nevertheless, there are still no quantitative data on the effect of functional state of mitochondria on the equilibrium between the oligomeric forms and on the equilibrium between the free and membrane-bound enzyme.
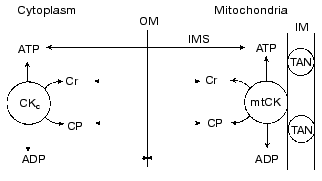
What is the physiological role of mtCK? The physiological role of the creatine kinase system as a whole and of mtCK in particular was considered in our recent review [80]. Based on our above-described experiments and also on the analysis in detail of kinetic, thermodynamic, and structural data which form the foundation for the concept of structural-functional coupling of mtCK and TAN, we arrived at the conclusion that no unidirectional synthesis of CP occurs in mitochondria and that mtCK functions under conditions close to equilibrium. Based on the literature, we concluded that 80-90% of ATP in the heart and skeletal muscle cells should be unbound. We have also used the literature on the limited permeability of the outer membrane of mitochondria for charged substances [86] and on the importance of the resulting concentration gradient for substances which are present in the cells at micromolar concentrations, e.g., for ADP, but not for ATP, Cr, and CP [87]. We arrived at the conclusion that at any instant the concentrations of ATP, Cr, and CP in the intermembrane space of mitochondria should not differ from their concentrations in the cytoplasm. Because the creatine kinase reaction in the cytoplasm of skeletal muscle and heart is close to the equilibrium state over a wide range of conditions, this means that mtCK is unable to provide in the intermembrane space ADP concentration higher than the equilibrium concentration in the cytoplasm. The nearer is mtCK to the equilibrium state, the nearer is the ADP concentration in the intermembrane space to the ADP concentration in the cytoplasm. Thus, we arrived at the conclusion that the specific function of mtCK is responsibility for rapid equilibrating of the ADP concentration, and, consequently, of the ATP/ADP ratio in the intermembrane space with their ratio in the cytoplasm at this instant [80, 88]. Our scheme of mtCK functions is presented in Fig. 10. Because this system is operating without amplification, the activity of mtCK furnishes the oxidative phosphorylation system with undistorted information about the current value of the phosphoryl potential in the cytoplasm. In a bioenergetic model describing the events of the contraction-relaxation cycle in muscle such a feedback mechanism is sufficient for maintenance of energy balance [89]. However, at low concentrations of ADP in muscle cells, the ADP of the intermembrane space can stimulate the respiration of mitochondria much more strongly than it would be stimulated in the presence of the same ADP concentration in the cytoplasm and in the absence of the mtCK activity (because the KmADP value for TAN must be lower than the KmADP value for mitochondria, which is affected by a relative impermeability of the outer membrane). Thus, the appearance of mtCK (and also of other localized CK) is a certain adaptation to the complicated structural organization of cell proteins that is responsible for diffusion limitations inside some protein complexes, and these limitations are significant for substances which are present in the cell at low concentrations.
Fig. 10. Scheme presenting the role of mtCK in elimination of the ADP concentration gradient across the outer membrane (OM) of mitochondria: the direct exchange of nucleotides between mtCK and TAN is absent; the concentrations of ATP, Cr, and CP in the intermembrane space (IMS) are the same as in the cytoplasm (this is shown by two-directional arrows intersecting OM); OM has a limited permeability for ADP (dotted arrows not intersecting OM); creatine kinase reactions in the cytoplasm (CKc) and in mitochondria are close to the equilibrium state (the arrows indicate both directions of the reaction); as a result of the mtCK activity, the ADP concentration in the IMS is equal to its concentration in the cytoplasm. IM) inner membrane [80].
REFERENCES
1.Jacobs, H., Heldt, H. W., and Klingenberg, M.
(1964) Biochem. Biophys. Res. Commun., 16, 516-521.
2.Bessman, S. P., and Fonio, A. (1966) Biochem.
Biophys. Res. Commun., 22, 597-602.
3.Vial, C., Godinot, K., and Gautheron, D. (1972)
Biochimie, 57, 843-852.
4.Jacobus, W. E., and Lehninger, A. (1973) J.
Biol. Chem., 248, 4803-4810.
5.Srere, P. A., and Mosbach, R. (1974) Ann. Rev.
Microbiol., 28, 61-83.
6.Friedrich, P. (1991) J. Theor. Biol.,
152, 115-116.
7.Saks, V. A., Chernousova, G. B., Gukovsky, D. E.,
Smirnov, V. N., and Chazov, E. I. (1975) Eur. J. Biochem.,
57, 273-290.
8.Scholte, H. R. (1973) Biochim. Biophys.
Acta, 305, 413-427.
9.Dmitrenko, N. P., and Bukhanevich, A. M. (1973)
Dokl. Akad. Nauk SSSR, 213, 963-965.
10.Booth, R. F. G., and Clark, J. B. (1978)
Biochem. J., 170, 145-151.
11.Iyengar, M. R., and Iyengar, C. L. (1980)
Biochemistry, 19, 2176-2182.
12.Lipskaya, T. Yu., Temple, V. D., Belousova, L.
V., Molokova, E. V., and Rybina, I. V. (1980) Biokhimiya,
45, 1155-1166.
13.Lipskaya, T. Yu., and Rybina, I. V. (1987)
Biokhimiya, 52, 690-700.
14.Font, B., Goldschmidt, D., Eichenberger, D., and
Gautheron, D. C. (1981) Arch. Biochem. Biophys.,
212, 195-203.
15.Wenger, W. C., Murphy, M. P., Brierley, G. P.,
and Altshuld, R. A. (1985) J. Bioenerg. Biomembr.,
17, 295-303.
16.Brooks, S. P. J., and Suelter, C. H. (1987)
Arch. Biochem. Biophys., 253, 122-132.
17.Saks, V. A., Khuchua, Z. A., Kuznetsov, A. V.,
Veksler, V. I., and Sharov, V. G. (1986) Biochem. Biophys. Res.
Commun., 139, 1262-1271.
18.Smirnova, I. A. (1989) Study on Interaction of
Creatine Kinase with Mitochondrial Membranes: Diploma paper [in
Russian], Moscow State University.
19.Yang, W. C. T., Geiger, P. J., and Bessman, S. P.
(1977) Biochem. Biophys. Res. Commun., 76, 882-887.
20.Altshuld, R. A., and Brierley, G. P. (1977) J.
Mol. Cell. Cardiol., 9, 875-896.
21.Muller, M., Mosor, R., Cheneval, D., and
Carafoli, E. (1985) J. Biol. Chem., 260, 3839-3843.
22.Lipskaya, T. Yu., Temple, V. D., Belousova, L.
V., and Molokova, E. V. (1980) Biokhimiya, 45,
1347-1351.
23.Lipskaya, T. Yu., Geiger, P. J., and Bessman, S.
P. (1995) Biochem. Mol. Med., 55, 81-89.
24.Weidemann, M. J., Erdelt, H., and Klingenberg, M.
(1970) Eur. J. Biochem., 16, 313-335.
25.Guarniery, M., Stechmiller, B., and Lehninger, A.
(1971) J. Biol. Chem., 246, 7526-7532.
26.Krebs, J. J. R., Hauser, H., and Carafoli, E.
(1979) J. Biol. Chem., 254, 5308-5316.
27.Hall, N., and DeLuca, M. (1980) Arch. Biochem.
Biophys., 201, 674-677.
28.Lipskaya, T. Yu., and Molokova, E. V. (1983) in
Biochemistry and Biophysics of Muscles (Severin, S. E., ed.) [in
Russian], Nauka, Moscow, pp. 118-129.
29.Jacobs, H. K. (1974) Fed. Proc.,
33, 1534.
30.Lehninger, A. (1974) Biochemistry [Russian
translation], Mir, Moscow.
31.Blum, H. E., Deus, B., and Gerok, W. (1983) J.
Biochem., 94, 1247-1257.
32.Vial, C., Font, B., Goldschmidt, D., and
Gautheron, D. C. (1979) Biochem. Biophys. Res. Commun.,
88, 1352-1359.
33.Lipskaya, T. Yu., and Molokova, E. V. (1982) in
Special FEBS Meeting on Cell Function and
Differentiation, Athens, Greece, p. 212.
34.Lipskaya, T. Yu., Temple, V. D., Belousova, L.
V., Molokova, E. V., and Rybina, I. V. (1983) Adv. Myocardiol.,
3, 597-611.
35.Lipskaya, T. Yu. (1986) Biol. Nauki,
9, 5-14.
36.Lipskaya, T. Yu. (1989) Sov. Med. Rev. A.
Cardiol., 2, 253-299.
37.Reiner, J. M. (1969) Behaviour of Enzyme
Systems, Van Nostrand Reinhold Co, New York.
38.Newman, R. A., Hacker, M. P., and Fagan, M. A.
(1982) Biochem. Pharmacol., 31, 109-111.
39.Schlame, M., and Augustin, W. (1985) Biomed.
Biochim. Acta, 44, 1083-1088.
40.Cheneval, D., and Carafoli, E. (1988) Eur. J.
Biochem., 171, 1-9.
41.Hall, N., Addis, P., and DeLuca, M. (1977)
Biochem. Biophys. Res. Commun., 76, 950-956.
42.Vallin, I., and Low, H. (1963) Biochim.
Biophys. Acta, 69, 361-374.
43.Lipskaya, T. Yu., Kedishvili, N. Yu., and
Kalenova, M. E. (1985) Biokhimiya, 50, 1571-1581.
44.Schlegel, J., Zurbiggen, B., Wegmann, G., Wyss,
M., Eppenberger, H. M., and Wallimann, T. (1988) J. Biol. Chem.,
263, 16942-16953.
45.Schnyder, T., Engel, A., Lustig, A., and
Wallimann, T. (1988) J. Biol. Chem., 263,
16954-16962.
46.Jacobs, H. K., and Graham, M. (1978) Fed.
Proc., 37, 1574.
47.Farrel, E. C., Baba, N., Brierley, G. P., and
Grumer, H. D. (1972) Lab. Invest., 27, 209-213.
48.Hall, N., Addis, P., and DeLuca, M. (1979)
Biochemistry, 18, 1745-1751.
49.Moskvitina, E. L., and Belousova, L. V. (1985)
Dokl. Akad. Nauk SSSR, 281, 209-213.
50.Lipskaya, T. Yu., Moiseeva, N. S., and Trofimova,
M. E. (1989) Biochem. Int., 18, 1161-1171.
51.Schlegel, J., Wyss, M., Schurch, U., Schnyder,
T., Quest, A., Wegmann, G., Eppenberger, H. M., and Wallimann, T.
(1988) J. Biol. Chem., 263, 16963-16969.
52.Belousova, L. V., Fedosov, S. N., Orlova, E. V.,
and Stel'mashchuk, V. Ya. (1991) Biochem. Int., 24,
51-58.
53.Fritz-Wolf, K., Schnyder, T., Wallimann, T., and
Kabsch, W. (1996) Nature, 381, 341-345.
54.Schlattner, U., Forstner, M., Eder, M.,
Stachowiak, O., Fritz-Wolf, K., and Wallimann, T. (1998) Mol. Cell.
Biochem., 184, 125-140.
55.Stachowiak, O., Schlattner, U., Dolder, M., and
Wallimann, T. (1998) Mol. Cell. Biochem., 184,
141-151.
56.Lipskaya, T. Yu., Borisova, T. A., Trofimova, M.
E., and Kedishvili, N. Yu. (1987) Biokhimiya, 52,
1512-1522.
57.Gross, M., and Wallimann, T. (1995)
Biochemistry, 34, 6660-6667.
58.Milner-White, E. J., and Watts, D. C. (1971)
Biochem. J., 122, 727-740.
59.Lipskaya, T. Yu. (1990) Physicochemical
Properties of Mitochondrial Creatine Kinase and Regulation of Its
Activity: Doctoral dissertation [in Russian], Moscow State
University.
60.Marcillat, O., Goldshmidt, D., Eichenberger, D.,
and Vial, C. (1987) Biochim. Biophys. Acta, 890,
233-241.
61.Saks, V. A., Kupriyanov, V. V., Elizarova, G. V.,
and Jacobs, W. E. (1980) J. Biol. Chem., 225,
755-763.
62.Tanaka, H., Sugiya, H., and Fujita, Y. (1983)
Int. J. Biochem., 15, 297-302.
63.Basson, C. T., Grace, A. M., and Roberts, R.
(1985) Mol. Cell. Biochem., 67, 151-159.
64.Lipskaya, T. Yu., Trofimova, M. E., and Moiseeva,
N. S. (1989) Biochem. Int., 19, 603-613.
65.Morrison, J. F., and James, S. (1965) Biochem.
J., 97, 37-52.
66.Nichei, T., Noda, L., and Morales, M. F. (1961)
J. Biol. Chem., 236, 3203-3209.
67.Quemeneur, E., Marcillat, O., Eichenberger, D.,
and Vial, C. (1989) Biochem. Int., 18, 365-371.
68.Schlegel, J., Wyss, M., Eppenberger, H. M., and
Wallimann, T. (1990) J. Biol. Chem., 265, 9221-9227.
69.Deamer, D. W., Utsumi, K., and Packer (1967)
Arch. Biochem. Biophys., 121, 641-651.
70.Monsan, P., Puro, G., and Morarquil, H. (1975)
Biochimie, 57, 1281-1292.
71.Quiocho, F. A. (1976) Meth. Enzymol.,
44, 546-558.
72.Lipskaya, T. Yu., and Trofimova, M. E. (1989)
Biochem. Int., 18, 1029-1039.
73.Quemeneur, E., Eichenberger, D., Goldshmidt, D.,
Vial, C., Beauregard, G., and Potier, M. (1988) Biochem. Biophys.
Res. Commun., 153, 1310-1314.
74.Lipskaya, T. Yu., and Trofimova, M. E. (1989)
Biochem. Int., 18, 1149-1159.
75.Sjostrand, F. S. (1978) J. Ultrastruct.
Res., 64, 217-245.
76.Baba, N., Kim, S., and Farrell, E. C. (1976)
J. Mol. Cell. Cardiol., 8, 599-617.
77.Biermans, W., Bakker, A., and Jacob, W. (1990)
Biochim. Biophys. Acta, 1018, 225-228.
78.Wojtczak, L., and Nalecz, M. J. (1979) Eur. J.
Biochem., 94, 99-107.
79.Rytikova, N. S. (1996) Mitochondrial Creatine
Kinase: Properties in Solution and in Mitochondria: Candidate's
dissertation [in Russian], Moscow State University.
80.Lipskaya, T. Yu. (2001) Biochemistry
(Moscow), 66, 115-129.
81.Nicolay, K., van Dorsten, F. A., Reese, T.,
Kruiskamp, M. J., Gellerich, J. F., and van Echteld, C. J. A. (1998)
Mol. Cell. Biochem., 184, 195-208.
82.Van Dorsten, F. A., Reese, T., Laudy, M., van
Echteld, C. J. A., Nederhoff, M. G. J., Ton, J., Laterveer, F. D.,
Gellerich, F. N., and Nicolay, K. (1998) in Current and Future
Applications of Magnetic Resonance in Cardiovascular
Disease (Higgins, C. B., Ingwall, J. S., and Pohost, G. M.,
eds.) Futura Publishing Company, Inc., Armonk, New York, pp.
421-445.
83.Schnyder, T., Rojo, M., Furter, R., and
Wallimann, T. (1994) Mol. Cell. Biochem., 133/134,
115-123.
84.Brdiczka, D., Beutner, G., Ruck, A., Dolder, M.,
and Wallimann, T. (1998) Biofactors, 8, 235-242.
85.Beutner, G., Ruck, A., Riede, B., and Brdiczka,
D. (1998) Biochim. Biophys. Acta, 1368, 7-18.
86.Dolder, M., Zeth, K., Tittmann, P., Gross, M.,
Welte, W., and Wallimann, T. (1999) J. Struct. Biol.,
127, 64-71.
87.Gellerich, F. N., Kapischke, M., Kunz, W.,
Neumann, W., Kuznetsov, A., Brdiczka, D., and Nicolay, K. (1994)
Mol. Cell. Biochem., 133/134, 85-104.
88.Lipskaya, T. Yu. (2000) in Proc. Conf. on
Mitochondria, Cells, and Reactive Oxygen Species [in Russian],
Pushchino, June 6-9, 2000, Institute of Cell Biophysics, Russian
Academy of Sciences, pp. 96-97.
89.Kushmerick, M. J. (1998) Comp. Biochem.
Physiol., 120, 109-123.