REVIEW: Non-photochemical Quenching of Fluorescence in Cyanobacteria
N. V. Karapetyan
Bach Institute of Biochemistry, Russian Academy of Sciences, Leninsky pr. 33, 119071 Moscow, Russia; fax: (495) 954-2732; E-mail: nkarap@inbi.ras.ru
Received May 7, 2007
The pathways of energy dissipation of excessive absorbed energy in cyanobacteria in comparison with that in higher plants are discussed. Two mechanisms of non-photochemical quenching in cyanobacteria are described. In one case this quenching occurs as light-induced decrease of the fluorescence yield of long-wavelength chlorophylls of the photosystem I trimers induced by inactive reaction centers: P700 cation-radical or P700 in triplet state. In the other case, non-photochemical quenching in cyanobacteria takes place with contribution of water-soluble protein OCP (containing 3´-hydroxyechinenone) that induces reversible quenching of allophycocyanin fluorescence in phycobilisomes. The possible evolutionary pathways of the involvement of carotenoid-binding proteins in non-photochemical quenching are discussed comparing the cyanobacterial OCP and plant PsbS protein.
KEY WORDS: allophycocyanin, cyanobacteria, 3´-hydroxyechinenone, light-harvesting complex LHCII, non-photochemical quenching, orange carotenoid-binding protein, phycobilisomeDOI: 10.1134/S0006297907100100
Abbreviations: APC) allophycocyanin; ELIP) early light-inducible protein (formed in plants under stress conditions); HLIP) high light-inducible protein (formed in cyanobacteria under stress conditions); IsiA) iron stress-induced protein A (formed in cyanobacteria at iron depletion); LHCII) light-harvesting complex of photosystem II; OCP) orange carotenoid-binding protein; P700 (P700+)) primary electron donor of photosystem I reaction center in reduced (oxidized) state; 3P700) P700 in triplet state; PSI) photosystem I; PSII) photosystem II.
STRATEGY OF PROTECTION OF OXYGENIC PHOTOSYNTHESIS AGAINST INTENSE
LIGHT
Photosynthetic reactions occur with optimal efficiency under illumination with low light intensity when the energy of quanta absorbed by chlorophylls is almost completely utilized in photosynthesis. Energy of singlet excited molecules of antenna chlorophylls migrates to chlorophylls of the reaction centers of photosystem I and II (PSI, PSII) causing charge separation (photochemical quenching, qP). Excessive absorbed excitation energy is emitted by singlet excited chlorophyll molecules as fluorescence (qF). When reaction centers are inactive, the absorbed energy is transferred into heat (qE, or non-photochemical quenching) or used to form the triplet state of antenna chlorophylls [1, 2]. Interaction of chlorophyll triplets with oxygen generates singlet oxygen that causes destruction of the photosynthetic apparatus [3].
Core antenna of all photosynthetic organisms is organized in the same way and consists of chlorophylls and carotenoids. Membrane bound carotenoids in the form of chlorophyll-carotenoid-protein complexes participate both in light-harvesting and in protection of complexes against intense light that stimulates generation of active species of oxygen and triplet state chlorophylls [4, 5]; membrane-bound carotenoids also play a structural role [6, 7].
Since under illumination with high light intensity not all absorbed energy can be used for photosynthetic CO2 assimilation, the excess energy leads to photooxidative destruction of the photosynthetic apparatus induced by active oxygen species [3, 8]. In the course of evolution, the photosynthetic organisms created several strategies to dissipate the excessive absorbed energy to stabilize the photosynthetic apparatus. Unbalance of absorbed energy and electron transport under high light illumination causes structural transformations that optimize photosynthesis. But if the transients are not enough to promote the utilization efficiency of absorbed quanta energy, dissipation processes of excessive absorbed energy are involved. Therefore, photosynthesis is optimal under the balance of activity and stability of the photosynthetic apparatus that is extremely important for the final photosynthetic productivity.
There are several protection pathways of the photosynthetic apparatus against intense light:
- optimization of photosynthetic electron transport due to fast reversible transients of the photosynthetic apparatus (so-called “state transitions”) that result in redistribution of excitation between photosystems [9-11], or due to regulation of the PSI/PSII relationship in course of long-term adaptation of the photosynthetic organisms to light quality [12, 13];
- non-photochemical quenching of the excitation energy via light-harvesting complex of photosystem II (LHCII) [14-17];
- dissipation of energy absorbed by PSI due to formation of triplet state of antenna chlorophylls [18];
- dissipation of excitation energy due to cation radicals of PSI and PSII of cyanobacteria [19, 20] and higher plants [21, 22];
- biosynthesis of stress proteins ELIP (early light-inducible protein) in higher plants [23, 24] or HLIP (high light-inducible protein) in cyanobacteria [25, 26] that can serve for binding of additionally formed chlorophyll molecules under high light illumination;
- de novo biosynthesis of destroyed complexes like D1 protein of the PSII reaction center [27]; biosynthesis of damaged PsaA/PsaB of PSI complex is very slow [28].
Thus dissipation of the excessive absorbed energy takes place with contribution of pigments of light-harvesting complexes and core antenna chlorophylls that interact with primary electron donor of both photosystems in closed (inactive) state. In this review, the mechanisms of recently discovered in cyanobacteria non-photochemical quenching, especially the carotenoid triggered quenching of phycobilisome fluorescence [29-31], will be discussed in comparison with more fully studied mechanisms of non-photochemical quenching in higher plants with contribution LHCII [1, 2, 14-17].
DISSIPATION OF EXCESS EXCITATION IN HIGHER PLANTS
Light-harvesting complex LHCII, the most widespread membrane protein on the Earth [32], serves not only for absorption and transfer of excitation energy to PSII antenna; dissipation of energy not utilized in photosynthesis in higher plants occurs with contribution of the same complex. LHCII is organized in vivo as trimers [33-35] that are able to form aggregates; LHCII trimers are packed into a heptamer in the thylakoids of higher plants [36]. Aggregation of LHCII in vitro is accompanied by structural changes as well as by strong decrease of fluorescence yield and by red shift of absorption and fluorescence bands [37]. Much lower fluorescence life-time of LHCII trimer aggregates in comparison with separate trimers is indicative of efficient quenching in aggregates. It was shown that fluorescence life-time of isolated LHCII trimers is of about 4.3 nsec, while it is much lower for aggregates--only 110 psec [38]. Dissipation of energy in LHCII aggregates may occur with contribution of the peripherally localized chlorophylls and carotenoids. Violaxanthin causes deaggregation, while zeaxanthin causes aggregation of LHCII complexes [37].
The localization of the molecules of carotenoid and chlorophylls a and b in LHCII complex is established. According to the first atomic model, each monomeric complex consists of 12 chlorophyll and two lutein molecules [34]. More correct LHCII structural models developed by analysis with 2.72 ? [35] and 2.5 ? resolution [39] have shown that each LHCII monomer includes eight chlorophyll a and six chlorophyll b molecules, four molecules of carotenoids (two luteins, one 9´-cis-neoxanthin, and one violaxanthin localized on the interface of monomers within a trimer and therefore is weakly bound to complex), and two lipid molecules.
LHCII complexes have diverse functions. They absorb light quanta and transfer excitation to PSII reaction center, contribute in redistribution of excitation energy between PSI and PSII in course of state transitions, and are involved in stacking of thylakoid membranes of chloroplast grana. Finally, LHCII is involved in dissipation of excess absorbed energy protecting the photosynthetic apparatus against destruction. Contribution in non-photochemical quenching requires the ability of LHCII for structural and functional mobility that allows the regulation of the energetic state of the photosynthetic apparatus in response to environmental signal (intense light). Non-photochemical quenching is accompanied by a decrease of the fluorescence yield of PSII reversible in the dark. Illumination of higher plants by intense light causes an acidification of thylakoid lumen triggering the de-epoxidation of violaxanthin into zeaxanthin. Zeaxanthin is characterized by higher amount of conjugated pi-bonds and by lower first singlet state level (S1) and therefore is the most efficient energy acceptor [40, 41]. Note that low pH of thylakoid lumen promotes the protonation of one or more PSII proteins involved in qE [42, 43].
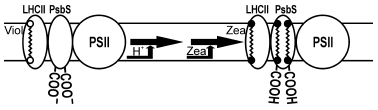
Non-photochemical quenching is a complex process involving various proteins. PsbS protein bound to LHCII is one of the most important polypeptides involved in thermal dissipation [2, 16, 43-45]. In addition to low pH of lumen and zeaxanthin, non-photochemical quenching requires also PsbS protein [44], which is sensitive to low pH (Fig. 1). PsbS protein lacks chlorophylls and xanthophylls, but in protonated form is able to bind exogenous zeaxanthin [43, 46]. It is suggested that zeaxanthin-bound PsbS protein may be the site of non-photochemical quenching. PsbS protein formally belongs to the family of light-harvesting proteins but participates only in energy dissipation.
Orientation of pigments and conformation of proteins in LHCII trimers determine not only efficient light harvesting and high protein stability but also the ability to control energy dissipation. It is suggested that transformation of violaxanthin into zeaxanthin favors the energy dissipation via different mechanisms dependent on conditions. Conformation changes in LHCII cause the interaction of chlorophylls and carotenoids, transfer of electron from chlorophyll to zeaxanthin, and non-radiative transformation of chlorophyll-zeaxanthin complex into the ground state [47]. Non-radiative transition of chlorophylls into the ground state may occur also in course of intermolecular charge transfer when two chlorophyll molecules become closer as a result of conformational changes of protein caused by violaxanthin de-epoxidation [39, 48].Fig. 1. Scheme of non-photochemical quenching in plants with participation of LHCII (with permission of Dr. K. K. Niyogi). Illumination by high light intensity decreases the lumen pH below 6.0, which triggers de-epoxidation of violaxanthin (Viol) into zeaxanthin (Zea); protonated PsbS binds zeaxanthin forming a qE site.
PIGMENT APPARATUS OF CYANOBACTERIA
The photosynthetic electron transport chains in higher plants and cyanobacteria are organized similarly in principle, but the pigment apparatus of cyanobacteria reveals significant differences that determine the specifics of their pathways of energy dissipation.
All chlorophylls in cyanobacteria are localized only in core antenna of PSI and PSII; the chlorophyll/P700 ratio in PSI is about 100, while the chlorophyll/P680 ratio in PSII is only 40 [49]. The PSI core antenna of cyanobacteria, in contrast to core antenna of PSI of higher plants, is enriched with long-wavelength chlorophylls that absorb in the further red spectral range than P700. The long-wavelength chlorophylls are able not only to transfer excitation uphill to active P700 [50, 51] but also to contribute in dissipation of excess absorbed energy when PSI reaction centers are accumulated in oxidized or triplet state, i.e. kept in closed state [19, 20, 52].
Since cyanobacteria lack LHCII (internal antenna), phycobilisomes, non-membrane water-soluble complexes, function as the main light-harvesting complexes (external antenna). Phycobilisomes consist of core cylinders containing allophycocyanin (APC), and lateral cylinders (or rods) containing phycocyanin and phycoerythrin [53-56]. Pigments of lateral rods transfer the excitation energy via APC to terminal acceptor of energy (also APC) that transfers the excitation to antenna chlorophylls of PSII and PSI localized within the membrane [49, 57, 58]. Phycobilisomes can diffuse laterally on the surface of thylakoid membranes [59]. Phycobilisomes are bound with membranes through lipids even when they are energetically uncoupled from photosystems [60].
Core antennae of both photosystems contain many carotenoid molecules, mainly beta-carotenes [61, 62]. None of 22 molecules of beta-carotenes in the monomeric complex of PSI of the cyanobacterium Thermosynechococcus elongatus is localized in close vicinity of P700. Therefore, P700 is active even when the biosynthesis of carotenoids is completely inhibited [63]. According to a structural model, the beta-carotenes are localized close to the long-wavelength chlorophylls [61] and may serve for photoprotection [64].
Because of lacking the violaxanthin cycle [65], cyanobacteria are incapable of non-photochemical quenching that is characteristic for higher plants. As it will be shown, carotenoids, involved in energy dissipation in cyanobacteria, quench the APC fluorescence under illumination of cells by blue-green light [29, 66, 67]. Water-soluble protein OCP (orange carotenoid-binding protein) containing 3´-hydroxyechinenone is involved in non-photochemical quenching of cyanobacteria [30, 68].
NON-PHOTOCHEMICAL QUENCHING OF LONG-WAVELENGTH CHLOROPHYLLS
FLUORESCENCE IN CYANOBACTERIA BY INACTIVE REACTION CENTERS
Under high light illumination, the primary electron donors of the reaction centers of PSI and PSII are accumulated in closed state as cation radical or triplet state dependent on redox state of the acceptor side cofactors. Here the situation with PSI of the cyanobacterium Arthrospira platensis, whose core antenna contains the most red-shifted (longest-wavelength) form of chlorophyll absorbing at 740 nm and emitting at 760 nm at 77K will be mainly discussed [69, 70]. Fluorescence yield of this red chlorophyll form strongly depends on P700 redox state, allowing the monitoring of the fate of energy absorbed by PSI antenna [19, 71]. It was shown that P700+ (oxidized P700) at 77K is a very efficient quencher of fluorescence of red chlorophyll at 760 nm [50]. The value of the overlap integral of the fluorescence band at 760 nm (77K) and absorption band of P700+ is more than two orders of magnitude higher than that of the fluorescence band of the same chlorophyll and the absorption band of P700 [64, 72]. Though the longest-wavelength form of chlorophyll (absorbing at 740 nm and emitting at 760 nm) is localized in PSI core antenna at a distance of 32-40 ? from P700 [52, 73], the high value of overlap integral is the main reason for P700+ being an efficient quencher.
Since these values of the overlap integral are similar at physiological temperature because of wide fluorescence bands of antenna chlorophylls, P700 and P700+ may be equally efficient quenchers of excitation, which may explain the absence of variable fluorescence of PSI. Note that fluorescence yield of long-wavelength chlorophylls at 735 nm (77K) of PSI of higher plants is not dependent on the P700 redox state because the chlorophylls are located on heterodimer Lhca1/Lhca4, the light-harvesting complex of plant PSI [33], and are positioned at a distance of about 65 ? from P700 [73], making their interaction difficult.
When the photochemistry in PSI reaction centers is blocked under oxidative conditions and P700 is accumulated in its oxidized state, the absorbed excitation energy leads to formation of triplet state of antenna chlorophylls registered as light-induced decrease of absorption at 830 nm [18]. The time course of that decay at 77K with half-times 10 and 80 nsec coincides exactly with the kinetics of appearance of 532 nm signal ascribed to formation of triplet state of beta-carotene resulting from direct triplet-triplet exchange.
P700 in triplet state (3P700) of PSI trimers of the cyanobacterium A. platensis quenches efficiently the fluorescence at 760 nm emitted by the longest-wavelength chlorophyll form [52]. When the cofactors of the PSI acceptor side (A1, FX, FA/FB) are preliminarily reduced and electron transport from AO to A1 is blocked, recombination of separated charges leads to formation of 3P700 with half-times of 0.75 and 5.2 msec at 5K. The decay kinetics of 3P700 coincide exactly with the kinetics of light-induced decrease of 760 nm fluorescence, confirming that energy absorbed by long-wavelength chlorophyll as terminal energy acceptor is quenched by 3P700 [52]. Since in both situations the absorbed energy cannot be utilized by reaction centers, the observed quenching of PSI antenna fluorescence can be considered as non-photochemical quenching.
The preferable existence of PSI complexes in the form of trimers in cyanobacterial thylakoids [19, 71] is the basis for an additional pathway of energy dissipation. When P700 in one of the monomer complexes within a PSI trimer of the cyanobacterium A. platensis is oxidized, there is dissipation of energy as a result of interaction of antenna chlorophylls via peripherally located longest-wavelength chlorophyll emitting at 760 nm [52, 74, 75]. Thus aggregation of pigment-protein complexes into trimers stimulates dissipation of excessive absorbed energy, protecting PSI from photodestruction. Note that data concerning the quenching of fluorescence of the long-wavelength chlorophyll by P700+ and 3P700 are registered at 77K and therefore may be considered only as possible pathways of non-photochemical quenching in cyanobacteria. It is quite possible that at physiological temperatures both P700 and P700+ are characterized by lower efficiency of quenching. The role of 3P700 in energy dissipation under physiological conditions is not yet clear.
CAROTENOID TRIGGERED NON-PHOTOCHEMICAL QUENCHING OF PHYCOBILISOME
FLUORESCENCE IN CYANOBACTERIA
The response of cyanobacteria on the change of light intensity and quality depends on the duration of illumination. Short-term illumination leads to changes in state of the photosynthetic apparatus that optimize electron transport [11]; the mechanisms of redistribution of the excitation in cyanobacteria lacking LHCII differ from that of plants. Long-term illumination leads to regulation of biosynthesis of PSI and PSII complexes [12] as well as to biosynthesis of HLIP and ELIP stress proteins that serve to bind the additionally formed chlorophylls [23-26].
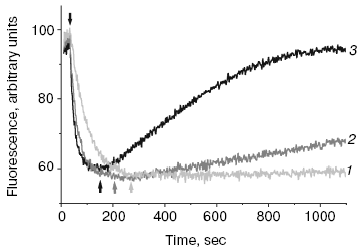
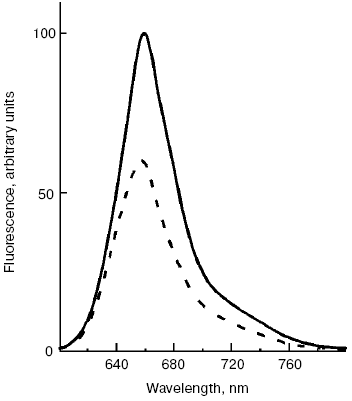
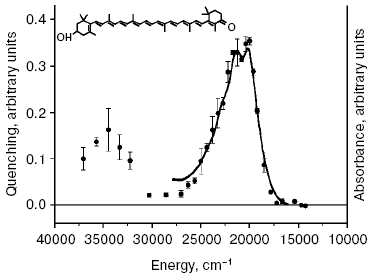
A new pathway of energy dissipation was recently discovered in cyanobacteria that rather belongs to prompt response to high light illumination [29, 76]. Illumination of PSII-less mutant cells of the cyanobacterium Synechocystis sp. PCC 6803 by intense blue-green light (>200 µE·m-2·sec-1, 450-500 nm) resulted in fluorescence decrease, slowly reversible in the dark (Fig. 2); the higher was temperature the faster was reversibility. This mutant was chosen to exclude possible interference in the found phenomenon of PSII variable fluorescence; to exclude the variable fluorescence of PSII of wild strain cells diuron was added [66, 67]. The spectrum of fluorescence quenching (i.e. difference between emission spectra measured before and after blue light illumination) peaked at 660 nm emitted by phycobilisomes (Fig. 3). Action spectrum of light-induced fluorescence quenching was characterized by three bands in the spectral region 450-500 nm and a band at 290 nm (Fig. 4) that have been attributed to myxol glycoside as a sensitizer responsible for quenching of phycobilisome fluorescence [29]. The band at 290 nm indicates that the trans-form of carotenoid was involved in quenching. However, initially it was unclear how carotenoids that usually are localized within the photosynthetic membrane can interact with non-membrane localized phycobilisomes. Investigation of the origin and location of carotenoid that triggers blue light-induced fluorescence quenching of water-soluble phycobilisomes, as well as an investigation of interaction of carotenoids with partner in phycobilisome showed the new mechanisms of energy dissipation in cyanobacteria.
Fig. 2. Time-course of blue light-induced quenching of phycobilisome fluorescence and dark recovery of quenching in PSII-less mutant of the cyanobacterium Synechocystis sp. PCC 6803 at 15 (1), 25 (2), and 35°C (3); measured with PAM-101; arrows v and ^, 500-nm actinic light on and off, correspondingly.
Fig. 3. Fluorescence emission spectra of PSII-less mutant of the cyanobacterium Synechocystis sp. PCC 6803 registered before (solid line) and after (dashed line) illumination of cells by intense 500-nm light, 15°C.
There is some prehistory of the new non-photochemical quenching in cyanobacteria. Since studies of blue light-induced fluorescence quenching have been carried out using a PAM-101 pulse fluorometer (Walz, Germany), the registered fluorescence quenching with wave-length more that 700 nm was ascribed to changes of the fluorescence yield of chlorophylls [76-79]. Note that in contrast to non-photochemical quenching in plants, blue light-induced quenching of phycobilisome fluorescence is not dependent on pH of the lumen and on uncouplers [77], and it is not found on illumination of cyanobacterial cells under red light absorbed by chlorophylls [29]. But the spectrum of fluorescence quenching was characterized by an emission band at 660 nm attributed to phycobilisomes, not to chlorophylls [29].Fig. 4. Action spectrum of quenching of phycobilisome fluorescence (black circles) measured for cells of PSII-less mutant of the cyanobacterium Synechocystis sp. PCC 6803 [29] and the absorption spectrum of isolated OCP (continuous line), with permission of Dr. T. Polivka. Inset, structure of 3´-hydroxyechinenone.
The identity of the action spectrum of phycobilisome fluorescence quenching with absorption spectrum (Fig. 4) of 3´-hydroxyechinenone of OCP indicates that this water-soluble carotenoid-binding protein localized outside of membranes induces the fluorescence quenching of phycobilisomes under blue light illumination [30]. OCP found in 1997 [80] is a 35-kD two-domain protein that contains one molecule of 3´-hydroxyechinenone and functions in cells as a dimer [81-84]. OCP isolated from the cyanobacterium Synechocystis sp. PCC 6803 contained the glycoside derivative of 3´-hydroxyechinenone [80, 82]. Thus the measured action spectrum of fluorescence quenching of phycobilisomes [29] revealed the function of a special family of water-soluble carotenoid-binding proteins (lacking chlorophylls) of photosynthetic organisms.
Carotenoid-protein complexes containing chlorophylls have been found in cells of the dinoflagellate Amphidinium carterae [85]: each peridinin-protein monomer complex consisted of eight molecules of peridinin and two chlorophyll molecules; energy absorbed by peridinins was able to migrate to chlorophylls. The isolated complex is functionally active in vivo as a trimer. Two chlorophyll molecules are isoenergetic at room temperature and weekly interact but strongly coupled to peridinin molecules [86]. The properties of carotenoid-binding proteins that contain chlorophylls as well as the chromoplast carotenoid-binding proteins without chlorophyll bound with lipoprotein structures [87] are not considered in this review.
MECHANISMS OF NON-PHOTOCHEMICAL QUENCHING OF PHYCOBILISOME
FLUORESCENCE IN CYANOBACTERIA
Thus carotenoids are responsible for blue light-induced fluorescence quenching of phycobilisomes in cyanobacteria. Because that quenching was more prominent in cyanobacteria grown under iron depletion, it was suggested that IsiA (iron stress-induced protein A) complex may serve as a quencher [77]. “Free” phycobilisomes could interact with chlorophyll containing IsiA complex that may replace LHCII, which is lacking in cyanobacteria [79]. IsiA complex consists of 16-18 subunits that circle the PSI trimers; each subunit contains 16 chlorophyll molecules and several carotenoids [88]. It was suggested that IsiA complex bound with PSI can function for additional light harvesting [89] or as a quencher of excess absorbed energy [90]; IsiA bound with PSII is mainly involved in discharging of excess energy [91].
However, it was shown later that blue light absorbed by carotenoid quenched phycobilisome fluorescence in the wild strain of Synechocystis sp. as in the IsiA-less mutant. This means that the IsiA complex is not necessary for blue light-induced quenching of phycobilisome fluorescence [30, 66-68]. The blue light-induced quenching was not found in an OCP-less mutant of Synechocystis sp. [30] as well as in apcE- mutant [30, 66, 67] with destroyed APC phycobilisome core [92]. Thus OCP and APC are the main participants of blue light induced fluorescence quenching of phycobilisomes in cyanobacteria. Light absorbed by carotenoid induces the quenching not only of phycobilisome fluorescence at 660 nm, but also fluorescence at 679 nm of chlorophylls that are in energetic equilibration with APC [67].
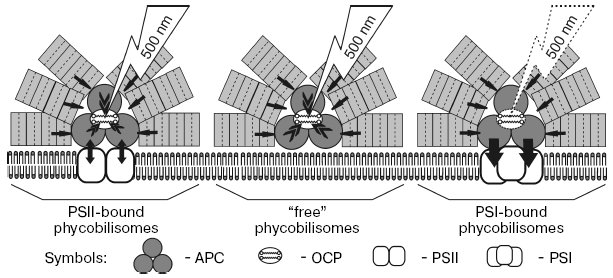
Based on the presented results and on data of time-resolved fluorescence spectroscopy of phycobilisomes and photosystems of cyanobacteria [31, 93-95], we proposed a scheme of carotenoid induced quenching of phycobilisome fluorescence in the cyanobacterium Synechocystis sp. (Fig. 5). The possible participation in quenching of three types of phycobilisomes coupled with PSI, with PSII, and with membrane lipids (“free” phycobilisomes) is discussed. The time of energy transfer from phycocyanin to APC was found equal to 20-40 psec, while time of energy migration from APC to PSII is 90-170 psec [93, 95]. When PSII reaction centers are open, the life-time of the PSII excited state is less than the time of energy migration from APC to PSII, and energy absorbed by phycobilisomes migrates to PSII chlorophylls [93, 95]. But if PSII reaction centers are closed, the life-time of the excited states of chlorophylls and reverse energy migration from chlorophylls to APC is about 1500-1700 psec [93, 94], and therefore part of the excitation energy could migrate from chlorophylls to APC (arrows APC-PSII). Measurements at high light illumination for wild strain of Synechocystis sp. in the presence of diuron, when PSII reaction centers are closed, allow registration of the fluorescence quenching both of phycobilisomes and chlorophylls [67]. If the phycobilisome is energetically coupled to PSI trimer, energy absorbed by phycobilisomes quickly (20-40 psec) migrates to PSI antenna chlorophylls independent from the P700 redox state (arrows from APC to PSI). If phycobilisomes are bound with membrane and energetically uncoupled with both photosystems, OCP quenches the phycobilisome fluorescence. Thus only fluorescence of phycobilisomes uncoupled from PSI, i.e. phycobilisomes energetically coupled with inactive PSII or bound with membrane, is quenched. The effect of high glycerol concentrations on the time-course and fluorescence emission spectra of PSII-less mutant strain of the cyanobacterium Synechocystis sp. have indicated that fluorescence of phycobilisomes bound with membrane can be quenched by blue light illumination [96]. Increasing protein hydration by addition of glycerol, which uncouples phycobilisomes from the membrane and damages their structure, suppresses blue light induced fluorescence quenching.
The temperature dependence of the rate of blue light induced fluorescence quenching and the calculated values of activation enthalpy indicate that light absorbed by carotenoid causes conformational changes of protein and may induce protein-protein interaction like folding of water-soluble proteins [96]. This is confirmed by the nonlinear dependence of the rate constant of fluorescence quenching of PSII-deficient Synechocystis sp. mutant and very high value of activation enthalpy of quenching (48 kJ/mol at 25°C and 86.5 kJ/mol at 5°C).Fig. 5. Scheme of 500-nm (absorbed by OCP) actinic light-induced non-photochemical quenching of phycobilisome fluorescence in the cyanobacterium Synechocystis sp. PCC 6803. Two molecules of carotenoid indicate that OCP functions as a dimer. Black arrows show migration of energy absorbed by phycobilisomes to OCP. Carotenoid triggered quenching of APC fluorescence under illumination with 500-nm light takes place in phycobilisomes energetically coupled with PSII dimer (reaction centers closed) and in “free” phycobilisomes bound with membrane. Note that OCP binding to phycobilisomes energetically coupled with PSI trimers is unclear.
The activation enthalpy calculated for the initial quenching rate of LHCII trimers and monomers [97] was only 6 kJ/mol, which is indicative of the absence of protein conformational changes at 15-35°C (confirmed by circular dichroism data). Low values of activation enthalpy in this temperature range are ascribed to local changes in orientation or position of chlorophylls and carotenoids within the LHCII subunit. The quenching rate at temperatures higher than 40°C increases, which is accompanied by one order of magnitude increase of activation enthalpy (70-90 kJ/mol), indicating significant changes in LHCII protein conformation.
The molecular mechanisms of fluorescence quenching of phycobilisomes is still unclear; it is quite possible that carotenoid of water-soluble OCP may serve as a quencher. Absorption of blue light may cause trans-cis isomerization of carotenoid leading to changes in protein conformation. Change in OCP conformation regulates the binding of quenching protein with phycobilisome or makes easier the interaction of OCP with phycobilisome core. In this case, the energy is transferred from S1 excited state of APC to carotenoid and dissipated [31, 96].
Thus, non-photochemical quenching as a general property of all oxygenic photosynthetic organisms protects the photosynthetic apparatus against destruction due to the ability to dissipate excessive absorbed energy of quanta. Fluorescence quenching of the longest-wavelength chlorophylls of PSI trimers by P700 cation radical and by P700 triplet state is considered as a type of non-photochemical quenching in cyanobacteria. The quenching mechanisms of excess absorbed energy with participation of carotenoids are principally similar in evolutionarily different photosynthetic organisms. OCP of cyanobacteria like PsbS protein of plants involved in non-photochemical quenching lack chlorophylls but bind carotenoid: 3´-hydroxyechinenone (OCP) or zeaxanthin (PsbS in protonated form). The water-soluble carotenoid-binding protein OCP interacting with water-soluble extra-membrane light-harvesting complex (phycobilisomes) was replaced in the course of evolution by membrane bound PsbS protein because of lack of extra-membrane phycobilisomes and the appearance of membrane bound light-harvesting complex LHCII. Both carotenoid-binding proteins function as a dimer, indicating the necessity of conformational changes of proteins in the course of non-photochemical quenching.
This work was supported by the Russian Academy of Sciences, program “Molecular and Cell Biology”, and the Russian Foundation for Basic Research, grant 05-04-48526a.
REFERENCES
1.Mueller, P., Li, X.-P., and Niyogi, K. K. (2001)
Plant Physiol., 125, 1558-1566.
2.Niyogi, K. K., Li, X.-P., Rosenberg, V., and Jung,
H.-S. (2004) J. Exp. Bot., 56, 375-382.
3.Krieger-Liszkay, A. (2005) J. Exp. Bot.,
56, 337-346.
4.Cogdell, R. J., and Frank, H. A. (1987) Biochim.
Biophys. Acta, 895, 63-79.
5.Young, A. J., and Frank, H. A. (1996) J.
Photochem. Photobiol., 36B, 3-15.
6.Moskalenko, A. A., and Karapetyan, N. V. (1996)
Z. Naturforsch., 51C, 763-771.
7.Havaux, M. (1998) Trends Plant Sci.,
3, 147-151.
8.Barber, J., and Andersson, B. (1992) Trends
Biochem. Sci., 17, 61-66.
9.Biggins, J., and Bruce, D. (1989) Photosynth.
Res., 20, 1-34.
10.Allen, J. F. (1992) Biochim. Biophys.
Acta, 1098, 275-335.
11.Mullineaux, C. W., and Emplyn-Jones, D. (2005)
J. Exp. Bot., 56, 389-393.
12.Fujita, J. (1997) Photosynth. Res.,
53, 83-93.
13.Bolychevtseva, Y. V., Mazhorova, L. E.,
Terekhova, I. V., Egorova, E. A., Shugaev, A. G., Rakhimberdieva, M.
G., and Karapetyan, N. V. (2003) Prikl. Biokhim. Mikrobiol.,
39, 503-508.
14.Demmig-Adams, B. (1990) Biochim. Biophys.
Acta, 1020, 1-24.
15.Horton, P., Ruban, A., and Walters, R. G. (1996)
Ann. Rev. Plant Physiol. Plant Mol. Biol., 47,
655-684.
16.Niyogi, K. K. (1999) Ann. Rev. Plant Physiol.
Plant Mol. Biol., 50, 333-359.
17.Horton, P., and Ruban, A. V. (2005) J. Exp.
Bot., 56, 365-373.
18.Schlodder, E., Paul, A., and Cetin, M. (2001) in
Proc. of 12th Int. Congr. on Photosynthesis, CSIRO; Publishing:
Melbourne, Australia, www.publishcsiro.au/ps2001; contribution
S6-015.
19.Karapetyan, N. V., Holzwarth, A. R., and Roegner,
M. (1999) FEBS Lett., 460, 395-400.
20.Karapetyan, N. V., Schlodder, E., van Grondelle,
R., and Dekker, J. P. (2006) in Photosystem I: The Light-Driven
Plastocyanin:Ferredoxin Oxidoreductase (Golbeck, J. H., ed.) Vol.
24, Springer, Dordrecht, pp. 177-192.
21.Bukhov, N. G., Heber, U., Wiese, C., and
Shuvalov, V. A. (2001) Planta, 212, 749-758.
22.Bukhov, N. G., Rajagopal, S., and Carpentier, R.
(2002) Photosynth. Res., 74, 295-302.
23.Grimm, B., Kruse, E., and Kloppstech, K. (1989)
Plant Mol. Biol., 13, 583-593.
24.Yurina, N. P., Pogulskaya, E. N., and Karapetyan,
N. V. (2006) Biochemistry (Moscow), 71, 430-436.
25.Dolganov, N. A. M., Bhaya, D., and Grossman, A.
R. (1995) Proc. Natl. Acad. Sci. USA, 92, 636-640.
26.Funk, C., and Vermaas, W. F. J. (1999)
Biochemistry, 38, 9397-9404.
27.Demmig-Adams, B., Adams, III, W. W., and Mattoo,
A. K. (eds.) (2006) Photoprotection, Photoinhibition,
Gene Regulation, and Environment, in Series: Advances in
Photosynthesis and Respiration, Springer, Dordrecht, Vol. 21.
28.Sonoike, K. (2006) in Photosystem I: The
Light-Driven Plastocyanin:Ferredoxin Oxidoreductase (Golbeck, J.
H., ed.) Vol. 24, Springer, Dordrecht, pp. 657-668.
29.Rakhimberdieva, M. G., Stadnichuk, I. N.,
Elanskaya, I. V., and Karapetyan, N. V. (2004) FEBS Lett.,
574, 85-88.
30.Wilson, A., Ajlani, G., Verbavatz, J.-M., Vass,
I., Kerfeld, C. A., and Kirilovsky, D. (2006) Plant Cell,
18, 992-1007.
31.Scott, M., McCollum, C., Vasiliev, S., Croizer,
C., Espie, G. S., Krol, M., Huner, N. P. A., and Bruce, D. (2006)
Biochemistry, 45, 8952-8958.
32.Peter, G. F., and Thornber, J. P. (1991) J.
Biol. Chem., 266, 16745-16754.
33.Jansson, S. (1994) Biochim. Biophys. Acta,
1184, 1-19.
34.Kuehlbrandt, W., Wang, D. N., and Fujiyoshi, Y.
(1994) Nature, 367, 614-621.
35.Liu, Z., Yan, H., Wang, K., Kuang, T., Zhang, J.,
Gui, L., An, X., and Chang, W. (2004) Nature, 428,
287-292.
36.Dekker, J. P., van Roon, H., and Boekema, E. J.
(1999) FEBS Lett., 449, 211-214.
37.Ruban, A. V., Phillip, D., Young, A. J., and
Horton, P. (1997) Biochemistry, 36, 7855-7859.
38.Mullineaux, C. W., Pascal, A. A., Horton, P., and
Holzwarth, A. R. (1993) Biochim. Biophys. Acta, 1141,
23-28.
39.Standfuss, J., van Scheltinga, L. M., and
Kuelbrandt, W. (2005) EMBO J., 24, 919-928.
40.Frank, H. A., Bautista, J. A., Josue, J. S., and
Young, A. J. (2000) Biochemistry, 39, 2831-2837.
41.Holt, N. E. F., Fleming, G. R., and Niyogi, K. K.
(2004) Biochemistry, 43, 8281-8289.
42.Horton, P., and Ruban, A. V. (1992)
Photosynth. Res., 34, 375-385.
43.Aspinal-O'Dea, M., Wentworth, M., Pascal, A. A.,
Robert, B., Ruban, A., and Horton, P. (2002) Proc. Natl. Acad. Sci.
USA, 99, 16331-16335.
44.Li, X.-P., Bjorkman, O., Shih, C., Grossman, A.
R., Rosenquist, M., Jansson, S., and Niyogi, K. K. (2000)
Nature, 403, 391-395.
45.Minagawa, J., and Takahassi, Y. (2004)
Photosynth. Res., 82, 241-263.
46.Dominici, P., Cafarri, S., Armenante, F., Ceoldo,
S., Crimi, M., and Bassi, R. (2002) J. Biol. Chem., 272,
2750-2758.
47.Horton, P., Ruban, A. V., and Wentworth, M.
(2000) Phil. Trans. R. Soc. London, 355B, 1361-1370.
48.Pascal, A. A., Liu, Z., Broess, K., van Oort, B.,
van Amerongen, H., Wang, C., Horton, P., Robert, B., Chang, W., and
Ruban, A. (2005) Nature, 436, 134-137.
49.Rakhimberdieva, M. G., Boichenko, V. A.,
Karapetyan, N. V., and Stadnichuk, I. N. (2001) Biochemistry,
40, 15780-15788.
50.Shubin, V. V., Bezsmertnaya, I. N., and
Karapetyan, N. V. (1995) J. Photochem. Photobiol., 30B,
153-160.
51.Gobets, B., van Stokkum, I. H. M., Roegner, M.,
Kruip, J., Schlodder, E., Karapetyan, N. V., Dekker, J. P., and van
Grondelle, R. (2001) Biophys. J., 81, 407-424.
52.Schlodder, E., Cetin, M., Byrdin, M., Terekhova,
I. N., and Karapetyan, N. V. (2005) Biochim. Biophys. Acta,
1706, 53-67.
53.Gantt, E. (1994) in Molecular Biology of
Cyanobacteria (Bryant, D. A., ed.) Kluwer Academic Publisher,
Dordrecht, pp. 119-138.
54.Bald, D., Kruip, J., and Roegner, M. (1996)
Photosynth. Res., 49, 103-118.
55.Roegner, M., Boekema, E., and Barber, J. (1996)
Trends Biochem. Sci., 21, 44-49.
56.MacColl, R. (1998) J. Struct. Biol.,
124, 311-334.
57.Mullineaux, C. W. (1992) Biochim. Biophys.
Acta, 1100, 285-292.
58.Mullineaux, C. W. (2001) Mol. Microbiol.,
41, 965-971.
59.Mullineaux, C. W., Tobin, M. J., and Jones, G. R.
(1997) Nature, 390, 421-424.
60.Sarcina, M., Tobin, M. J., and Mullineaux, C. W.
(2001) J. Biol. Chem., 276, 46830-46834.
61.Jordan, P., Fromme, P., Klukas, O., Witt, H. T.,
Saenger, W., and Krauss, N. (2001) Nature, 411,
909-917.
62.Zouni, A., Witt, H. T., Kern, J., Fromme, P.,
Krauss, N., Saenger, W., and Orth, P. (2001) Nature, 409,
739-743.
63.Bolychevtseva, Y. V., Rakhimberdieva, M. G.,
Karapetyan, N. V., Popov, V. I., Moskalenko, A. A., and Kuznetsova, N.
Y. (1995) J. Photochem. Photobiol., 27B, 153-160.
64.Karapetyan, N. V. (2004) Biofizika,
49, 212-226.
65.Hirschenberg, J., and Chamowitz, D. (1994) in
Molecular Biology of Cyanobacteria (Bryant, D. A., ed.) Kluwer
Academic Publisher, Dordrecht, pp. 119-138.
66.Rakhimberdieva, M. G., Elanskaya, I. V., Vavilin,
D. V., Vermaas, W. F. J., and Karapetyan, N. V. (2006) Biochim.
Biophys. Acta, 1757, Suppl. 1, 286-287.
67.Rakhimberdieva, M. G., Vavilin, D. V., Vermaas,
W. F. J., Elanskaya, I. V., and Karapetyan, N. V. (2007) Biochim.
Biophys. Acta, 1767, 732-741.
68.Wilson, A., Boulay, C., Wilde, A., Kerfeld, C.
A., and Kirilovsky, D. (2007) Plant Cell, 19,
656-672.
69.Shubin, V. V., Murthy, S. D. S., Karapetyan, N.
V., and Mohanty, P. (1991) Biochim. Biophys. Acta, 1060,
28-36.
70.Karapetyan, N. V., Dorra, D., Schweitzer, G.,
Bezsmertnaya, I. N., and Holzwarth, A. R. (1997) Biochemistry,
36, 13830-13837.
71.Shubin, V. V., Tsuprun, V. L., Bezsmertnaya, I.
N., and Karapetyan, N. V. (1993) FEBS Lett., 334,
79-82.
72.Karapetyan, N. V. (1998) Membr. Cell
Biol., 12, 571-584.
73.Dorra, D., Fromme, P., Karapetyan, N. V., and
Holzwarth, A. R. (1998) in Proc. of the XIth Int. Photosynthesis
Congress (Garab, G., ed.) Vol. 1, pp. 587-590.
74.Karapetyan, N. V., Shubin, V. V., and Strasser,
R. J. (1999) Photosynth. Res., 61, 291-301.
75.Karapetyan, N. V. (2004) Biochemistry
(Moscow), 69, 1299-1304.
76.El-Bissati, K., Delphin, E., Murata, N., and
Kirilovsky, D. (2000) Biochim. Biophys. Acta, 1457,
229-242.
77.Cadoret, J.-C., Demouliere, R., Lavand, J., van
Gorkom, H., Houmard, J., and Etienne, A.-L. (2004) Biochim. Biophys.
Acta, 1659, 100-104.
78.Bailey, S., Mann, N., Robinson, C., and Scanlan,
D. J. (2005) FEBS Lett., 579, 275-280.
79.Joshua, S., Bailey, S., Mann, N. H., and
Mullineaux, C. W. (2005) Plant Physiol., 138,
1577-1585.
80.Wu, Y. P., and Krogmann, D. W. (1997) Biochim.
Biophys. Acta, 1322, 1-7.
81.Kerfeld, C. A., Sawaya, M. R., Brahmandam, V.,
Cascio, D., Ho, K. K., Trevithick-Sutton, C. C., Krogmann, D. W., and
Yeates, T. O. (2003) Structure, 11, 56-65.
82.Kerfeld, C. A. (2004) Photosynth. Res.,
81, 215-225.
83.Kerfeld, C. A. (2004) Arch. Biochem.
Biophys., 430, 2-9.
84.Polivka, T., Kerfeld, C. A., Pascher, T., and
Sundstroem, V. (2005) Biochemistry, 44, 3994-4003.
85.Hofmann, E., Wrench, P. M., Sharples, F. P.,
Hiller, R. G., Welte, W., and Diederichs, K. (1996) Science,
272, 1788-1791.
86.Kleima, F. J., Hofmann, E., Gobets, B., van
Stokkum, I. H. M., van Grondelle, R., Diederichs, K., and van
Amerongen, H. (2000) Biochemistry, 39, 5184-5195.
87.Vishnevetsky, M., Ovadis, M., and Vainstein, A.
(1999) Trends Plant Sci., 44, 232-235.
88.Barber, J., Nield, J., and Duncan, J. (2006) in
Photosystem I: The Light-Driven Plastocyanin:Ferredoxin
Oxidoreductase (Golbeck, J. H., ed.) Vol. 24, Springer, Dordrecht,
pp. 99-117.
89.Boekema, E. J., Hifney, A., Yakushevska, A. E.,
Piotrowski, M., Keegstra, W., Berry, S., Michel, K. P., Pistorius, E.
K., and Kruip, J. (2001) Nature, 412, 745-748.
90.Ihalainen, J. A., D'Haene, S., Yeremenko, N., van
Roon, H., Arteni, A. A., Boekema, E. J., van Grondelle, R., Matthijs,
H. C. P., and Dekker, J. P. (2005) Biochemistry, 44,
10846-10853.
91.Sandstroem, S., Park, Y.-I., Oequist, G., and
Gustafsson, P. (2001) Photochem. Photobiol., 74,
431-437.
92.Shen, G., Boussiba, S., and Vermaas, W. F. J.
(1993) Plant Cell, 5, 1853-1863.
93.Mullineaux, C. W., and Holzwarth, A. R. (1991)
Biochim. Biophys. Acta, 1098, 68-78.
94.Bittersmann, E., and Vermaas, W. F. J. (1991)
Biochim. Biophys. Acta, 1098, 105-116.
95.Vavilin, D. V., Hu, S., Lin, S., and Vermaas, W.
F. J. (2003) Biochemistry, 42, 1731, 1746.
96.Rakhimberdieva, M. G., Bolychevtseva, Y. V.,
Elanskaya, I. V., and Karapetyan, N. V. (2007) FEBS Lett.,
581, 2429-2433.
97.Wentworth, M., Ruban, A. V., and Horton, P.
(2003) J. Biol. Chem., 278, 21845-21850.