Light Damaging Action of all-trans-Retinal and Its Derivatives on Rhodopsin Molecules in the Photoreceptor Membrane
M. Yu. Loginova*, Ye. V. Rostovtseva, T. B. Feldman, and M. A. Ostrovsky
Emanuel' Institute of Biochemical Physics, Russian Academy of Sciences, ul. Kosygina 4, 119334 Moscow, Russia; fax: (495) 137-4101; E-mail: marina.loguinova@mail.ru* To whom correspondence should be addressed.
Received July 13, 2007; Revision received October 8, 2007
We have reproduced the model system containing A2-rhodopsin, NR-PE, A2-PE, and ATR-dimer-PE in order to study photosensitized damage of rhodopsin within photoreceptor membranes of rod outer segments. We have demonstrated that irradiation of such a system with visible light (400-700 nm) distorts the most important functional property of native visual pigment--its ability to regenerate after addition of 11-cis-retinal in the dark. We have also shown that all-trans-retinal bound to membrane phospholipids and rhodopsin has less photosensitizing activity that free all-trans-retinal.
KEY WORDS: visual pigment rhodopsin, all-trans-retinal, photosensitizers, photodamageDOI: 10.1134/S000629790802003X
Abbreviations: ABCR) ATP-binding cassette transporter; A2E) N-bis-retinylidene-ethanolamine; A2-PE) N-bis-retinylidene-phosphatidylethanolamine; A2-rhodopsin) rhodopsin which has each of its three lysine residues modified with two all-trans-retinal molecules; ATR) all-trans-retinal; ATR-dimer-PE) all-trans-retinal-dimer-phosphatidylethanolamine; NR-PE) N-retinylidene-phosphatidylethanolamine; ROS) rod outer segments.
Light absorbed by the visual pigment rhodopsin leads to isomerization of
its chromophore group, 11-cis-retinal, to
all-trans-retinal. During photolysis, after formation of
metarhodopsin II, hydrolysis of the aldimine bond of the Schiff base
leads to release of all-trans-retinal (ATR) from the protein
part of the molecule--opsin [1]. Then it is reduced
to all-trans-retinol by retinol dehydrogenase and transferred
from rod outer segments into retinal pigment epithelial cells by
retinol-binding proteins. But under conditions of abundant illumination
free ATR is likely to accumulate in the photoreceptor membrane and
interact with phosphatidylethanolamine. In this case it can be
transported from the photoreceptor membrane in the form of
retinylidene-phosphatidylethanolamine by ATP-binding cassette
transporter (ABCR) [1-3].
In several degenerative retinal pathologies, e.g. Stargardt's disease, ABCR is defective and ATR is accumulated in the photoreceptor membrane [4-6].
ATR is an effective natural photosensitizer [7-9]. It can induce oxidation of proteins and lipids in the photoreceptor membrane through the mechanism of photosensitized oxidation, thus causing damage to rhodopsin and interphotoreceptor retinol-binding protein [10-14], as well as membrane protein ABCR responsible for transfer of ATR onto the cytoplasmic surface of the disc [15].
It is known that ATR in photoreceptor membrane is able to interact with amino groups of phosphatidylethanolamine leading to formation of N-retinylidene-phosphatidylethanolamine (NR-PE) and N-bis-retinylidene-phosphatidylethanolamine (A2-PE) [16]. These compounds are the precursors of a toxic fluorophore of lipofuscin granules, N-bis-retinylidene-ethanolamine (A2E), that decreases retinal pigment epithelial cell resistance to damage by blue light [16-19]. It is also known that ATR can form nonspecific covalent and noncovalent complexes with opsin/rhodopsin, leading to formation of so-called rhodopsin pseudoproducts [20-24]. Moreover, it has been shown that ATR also interacts with accessible amino groups of rhodopsin located on the hydrophilic cytoplasmic surface of the photoreceptor disk, leading to the formation of a fluorophore, A2-rhodopsin [25], which has each of its three lysine residues in the cytoplasmic domain of opsin connected with two ATR molecules through a pyridine ring. Recently one more product of interaction of phosphatidylethanolamine with two ATR molecules--all-trans-retinal-dimer-phosphatidylethanolamine (ATR-dimer-PE)--was found [26].
Phototoxic properties of products formed via interaction of ATR with rhodopsin and phosphatidylethanolamine in the photoreceptor membrane have been studied insufficiently [27, 28]. There are no data on the influence of these products on one of the main natural properties of rhodopsin--its ability to regenerate in the presence of exogenous 11-cis-retinal. However, complete or impaired rhodopsin regeneration is the most adequate physiological test to reveal damage to the rhodopsin molecule.
MATERIALS AND METHODS
Chemicals. Sucrose, urea, Tris, diethylenetriaminopentaacetic acid (DTPA), p-toluenesulfonyl fluoride, dithiothreitol (DTT), dimethylsulfoxide (DMSO), all-trans-retinal, hexane, ethyl acetate, acetonitrile, water, and trifluoroacetic acid were from Sigma (USA); Hepes was from Serva (Germany). 11-cis-Retinal was kindly provided by Dr. A. A. Khodonov (N. M. Emanuel Institute of Biochemical Physics, Russian Academy of Sciences).
Preparation of suspension of rod outer segments. Rod outer segments (ROS) were obtained from fresh dark-adapted (within 6 h after animal slaughter) retinas of bovine eyes according to the modified method of preparative centrifugation in sucrose density gradient [29].
Preparation of all-trans-retinal modified substances--A2-rhodopsin and products of interaction with phosphatidylethanolamine. A mixture of A2-rhodopsin and A2-PE products was obtained according to the procedure from [25]. To remove membrane-bound proteins, ROS were suspended in 5.0 M urea in 10 mM Tris, pH 7.5, and incubated for 60 min at 4°C. Then ROS were pelleted by centrifugation (100,000g, 60 min). After that, the ROS were resuspended in Hepes buffer (10 mM Hepes, 50 µM DTPA, 100 µM p-toluenesulfonyl fluoride, and 1 mM DTT, pH 7.0). ATR was dissolved in DMSO to final concentration of 7 mM. Rhodopsin and phosphatidylethanolamine were modification via incubation of ROS suspension with 2- and 10-fold molar excess of ATR (i.e. there were two or 10 molecules of ATR per rhodopsin molecule) in the darkness for 3 days at 37şC. ROS suspension incubated with DMSO under the same conditions with no ATR added was used as a control.
Rhodopsin regeneration. Degree of degeneration of rhodopsin native properties was revealed upon estimation of its ability to regenerate after bleaching of a ROS suspension with visible light and subsequent addition of exogenous 11-cis-retinal. In turn, rhodopsin regeneration in the photoreceptor membrane was estimated by restoration of absorption band at 500 nm of the initial spectrum. Rhodopsin regeneration was performed according to [30].
Five-fold molar excess of 11-cis-retinal in ethanol (the final concentration of ethanol in sample was below 2%) was added to rhodopsin bleached with visible light (400-700 nm during different periods of time: 15, 60, and 120 min) (bleaching parameters are described in detail below). After incubation of ROS suspension with 11-cis-retinal for 15 min, absorption spectra were recorded. Then hydroxylamine solution with final concentration of 20 mM was added to ROS suspension. Hydroxylamine was added to remove 11-cis-retinal excess by converting it to retinal-oxime. Then the ROS suspension was bleached with visible light for 5 min. After each step--bleaching and subsequent incubation in the darkness in presence of exogenous 11-cis-retinal--absorption spectra were recorded. Degree of regeneration was estimated according to the equation from [30]:
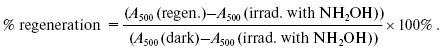
Spectral changes. A UV-1700 spectrophotometer and RF-5301 PC spectrofluorometer (both from Shimadzu, Japan) were used for recording spectra. For spectrophotometric studies, rhodopsin concentration was 0.357 mg/ml. For fluorometric measurements, the sample was 4 times diluted so that its optical density was 0.09.
High performance liquid chromatography (HPLC). Analysis was performed on Knauer chromatography instruments (Germany).
Extraction of retinal isomers from rhodopsin and their analysis by normal-phase HPLC. Retinal isomers were extracted according to [31]. One milliliter of formaldehyde was added to 2 ml of sample, mixed, and incubated for 2 min at 30°C. After addition of 1.5 ml of methylene chloride, the mixture was intensively shaken and incubated for 10 min. Finally, 1.5 ml of hexane was added, shaken, and then centrifuged (680g, 10 min). After centrifugation, the upper layer containing methylene chloride, hexane, and free retinal isomers was collected and concentrated under the vacuum of an water-jet pump. All operations were performed under dim red light. HPLC analysis was performed using IBM Silica column, 5 µm, 250 × 4.5 mm (IBM Instruments Inc, USA), eluent - 5% ethyl acetate in n-hexane, flow rate - 1.5 ml/min, spectrophotometric detection at 365 nm.
Extraction of retinal derivatives from rhodopsin and their analysis by reverse-phase HPLC. Extraction was performed according to the Folch method [32]. Four milliliters of chloroform-methanol mixture (2 : 1 v/v) was added to 1 ml of sample and intensively vortexed (Vortex, Elmi) for 2 min. Then 1 ml of water was added to the sample, stirred, and incubated for 2 min at room temperature. After that sample was centrifuged (680g, 10 min), and the chloroform lower fraction containing fat-soluble substances was collected and concentrated under vacuum of water-jet pump. All operations were performed under dim red light. Retinal derivatives and isomers were analyzed on Diasfer-110-C18 column, 5 µm, 240 × 4 mm (Biochemmack, Russia). Acetonitrile gradient (84-100%) against 0.05% TFA in water (16-0%): 84-90% for 5 min, 88% for 8 min, and 100% for 30 min (flow rate, 1 ml/min) was used for retinal derivatives analysis. Products were detected at 430 nm.
RESULTS
Preparation and identification of rhodopsin and lipids modified with all-trans-retinal in photoreceptor membrane of rod outer segments. Absorption spectra. A2-rhodopsin and A2-PE (rhodopsin and phosphatidylethanolamine modified with all-trans-retinal, respectively) products were prepared according to the method described in [25] without changes.
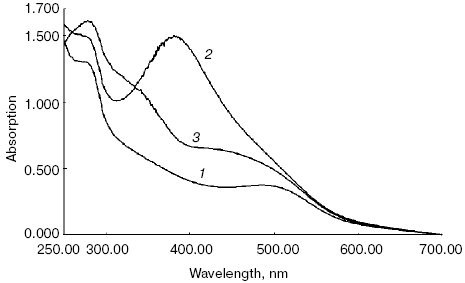
Absorption spectra of this mixture registered in the present work were identical to those presented in the abovementioned paper [25] (Fig. 1). Figure 1 shows that absorption spectrum of the initial dark suspension of ROS had a maximum at 500 nm, which is specific to the absorption spectrum of a native rhodopsin (Fig. 1, spectrum 1). Incubation of ROS suspension for 3 days at 37°C in darkness without exogenous ATR did not lead to any substantial alteration of spectral properties of rhodopsin (spectrum not shown).
Addition of 10-fold molar excess of ATR to the suspension led to the appearance of an absorption band with maximum at 380 nm characteristic of retinal, making the absorption spectrum of the ROS suspension with maximum around 500 nm almost invisible (Fig. 1, spectrum 2, registered immediately after addition of 10-fold excess of ATR).Fig. 1. Absorption spectra of ROS suspension containing rhodopsin: 1) dark ROS suspension (control); 2) dark ROS suspension in presence of 10-fold excess of ATR (spectrum was recorded immediately after addition of ATR excess to the suspension); 3) dark ROS suspension incubated with 10-fold excess of ATR for 3 days in a thermostat at 37°C in the dark.
Subsequent incubation of the ROS suspension in the presence of 10-fold excess of ATR for 3 days at 37°C in the darkness led to significant alteration of its spectral properties (Fig. 1, spectrum 3). It is noticeable that the absorption maximum of ATR at 380 nm present in spectrum 2 disappeared. Instead, two well-distinguished shoulders with maxima at 320-340 and 440-460 nm were observed. This indicates that new products spectrally different from free ATR were formed during the incubation.
Since this absorption spectrum is analogous to that observed in work [25] under the same experimental conditions, it most probably belongs to the same products determined in the mentioned work, namely A2-PE and A2-rhodopsin.
Fluorescence spectra. It is known that numerous products (for example, A2-PE and A2E) modified with all-trans-retinal have characteristic fluorescence [16, 27]. Therefore, since the absorption spectrum of products formed via incubation of ROS in the presence of exogenous ATR has maxima at 340 and 440 nm, we considered it expedient to perform fluorescence analysis exciting them with light of 320 and 460 nm.
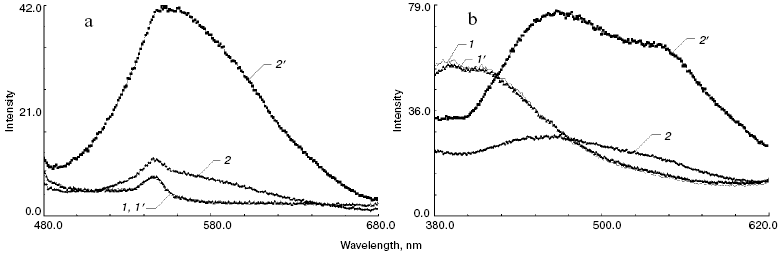
Figure 2 shows fluorescence spectra of a ROS suspension containing dark non-irradiated rhodopsin (before and after incubation without exogenous ATR), as well as ROS suspension before and after incubation with 10-fold excess of ATR for 3 days at 37°C.
It is clearly seen from the fluorescence spectra (Figs. 2a and 2b, spectra 1 and 1´), that the incubation of ROS suspension for 3 days in the thermostat at 37°C in the absence of exogenous ATR did not lead to any alterations of spectral properties of control samples, as was found for the absorption spectra (Fig. 1). This indicates that rhodopsin and its lipid environment in the photoreceptor membrane were stable even during prolonged incubation at rather high temperature.Fig. 2. Fluorescence spectra of ROS suspension containing rhodopsin with excitation at 460 nm (a) and 320 nm (b): 1, 1´) ROS suspension without exogenous ATR used as a control before and after incubation, respectively; 2, 2´) ROS suspension in the presence of 10-fold excess of ATR before and after incubation, respectively, with 10-fold excess of ATR for 3 days in a thermostat at 37°C in the dark.
Treatment of ROS suspension with 10-fold excess of ATR led to a significant alteration of fluorescence spectra both immediately after its addition (Fig. 2b, spectrum 2), and especially after incubation of the sample for 3 days at 37°C (Fig. 2a, spectrum 2´, and Fig. 2b, spectra 2 and 2´). Figure 2 (spectrum 2) shows that the addition of exogenous ATR to ROS suspension (without incubation) did not lead to significant change of fluorescence spectrum when the sample was excited near 460 nm. This indicates that the modification of rhodopsin and phosphatidylethanolamine by all-trans-retinal did not occur yet, and characteristic fluorescence of modified products was absent.
However, sharp increase of fluorescence intensity with maximum around 555 nm (Fig. 2a, spectrum 2´) was observed after incubation of the sample. This indicates the formation of new products upon incubation of the ROS sample with exogenous ATR.
Excitation of samples with 320 nm light demonstrated that non-incubated samples emitted fluorescence caused mainly by tryptophan residues in rhodopsin (Fig. 2b, spectra 1 and 1´).
Addition of exogenous ATR to the ROS suspension (without subsequent incubation) substantially quenched tryptophan fluorescence, and the shape of the spectra was significantly altered (Fig. 2b, spectrum 2). After incubation for 3 days at 37°C a noticeable increase in the fluorescence intensity of the sample was observed (Fig. 2b, spectrum 2´). At the same time, the spectrum shape was significantly altered as compared to the initial one. Fluorescence maxima at 470 and 545 nm were observed.
Considering excitation spectra at 555 nm, the mixture under investigation contains two fluorophores with absorbance maxima near 320 and 460 nm (data not shown).
Therefore, incubation of ROS suspension with 10-fold excess of exogenous ATR led to the formation of new products spectrally different from the initial ROS suspension containing rhodopsin without ATR.
Preparation and identification of products of all-trans-retinal oxidation in the absence of ROS suspension. It is possible to suggest that during prolonged incubation of ROS suspension with exogenous ATR at sufficiently high temperature (37şC) in medium containing oxygen, ATR not only interacts with amino groups of rhodopsin and phosphatidylethanolamine but also undergoes oxidation and degradation. Therefore, experiments were performed to study spectral properties of free ATR upon its incubation under the same conditions, but in the absence of ROS.
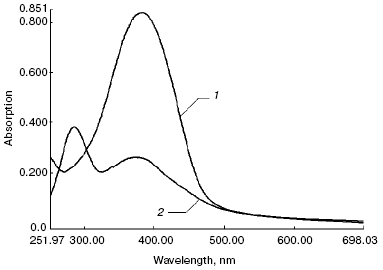
Absorption spectra. Figure 3 shows absorption spectra of free all-trans-retinal (spectrum 1) and products of its oxidation and degradation during incubation for 3 days at 37şC (spectrum 2). ATR initially dissolved in DMSO was administered to the same buffer, and ROS discs were resuspended in Hepes with necessary reagents (as described in “Materials and Methods”), in the same quantity as for the experiments with ROS discs.
The figure shows that products formed during the incubation mainly absorbed in the region 280-380 nm, and their spectral properties were different from those for products of the interaction of ATR with rhodopsin and phospholipid (absorption spectra of these products are shown in Fig. 1, spectrum 3).Fig. 3. Absorption spectra of free ATR (1) and its oxidation and degradation products (2) during the incubation of sample for 3 days at 37°C.
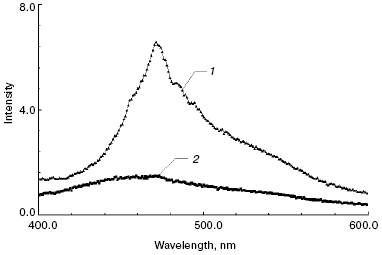
Fluorescence spectra. Fluorescence spectra of free ATR (Fig. 4, spectrum 1) and products of its oxidation and degradation (Fig. 4, spectrum 2) after the incubation were recorded in the same buffer solution used previously for the absorbance spectra. The spectrum of free ATR had a characteristic maximum at 467 nm. Comparison of fluorescence spectra of products of ATR oxidation and degradation (Fig. 4) with those of modified components of the photoreceptor membrane (Fig. 2) in the region 400-600 nm demonstrates that they did not contribute to the fluorescence of the latter in this spectral region.
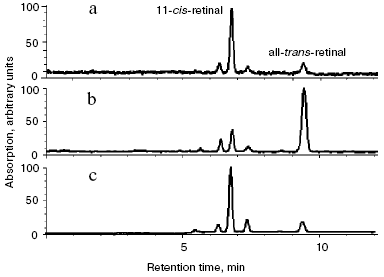
Determination of the nature of modified products. Isomeric composition of retinal and its derivatives in modified rhodopsin and lipids. Figure 5 shows the results of the analysis of retinal isomeric composition by normal-phase HPLC in extracts obtained from ROS suspension.Fig. 4. Fluorescence spectra of free ATR (1) and its oxidation and degradation products (2) during incubation of the sample for 3 days at 37°C. Excitation wavelength 320 nm.
The chromatogram (Fig. 5a) demonstrates that 11-cis- and all-trans-retinal content in the dark-adapted rhodopsin was 90 and 5%, respectively (the remaining 5% represents minor isomeric forms). After addition of 10-fold molar excess of all-trans-retinal to ROS suspension (without incubation) retinal isomer ratio sharply changes: trans-isomer content becomes higher than that of cis-isomer (approximate ratio 10 : 1) (Fig. 5b). After incubation of ROS suspension with 10-fold molar excess of ATR for 3 days at 37°C, isomer ratio again changes significantly and becomes virtually identical to the retinal isomer ratio in the control, i.e. without ATR addition and incubation. Namely, 11-cis- and all-trans-retinal content is equal to 90 and 5%, respectively (the remaining 5% represents minor isomeric forms) (Fig. 5c).Fig. 5. Analysis of retinal isomers by HPLC (normal-phase): a) retinal isomers extracted from dark rhodopsin according to [20] (control); b) retinal isomers extracted from dark rhodopsin with 10-fold excess of exogenous ATR; c) retinal isomers extracted after incubation of ROS suspension containing rhodopsin for 3 days at 37°C in the presence of 10-fold molar excess of ATR. Detection at 365 nm. All chromatograms are normalized.
This suggests that all the exogenous ATR added in 10-fold molar excess to ROS suspension was chemically modified due to its interaction with amino groups of rhodopsin and phosphatidylethanolamine during incubation for 3 days at 37°C.
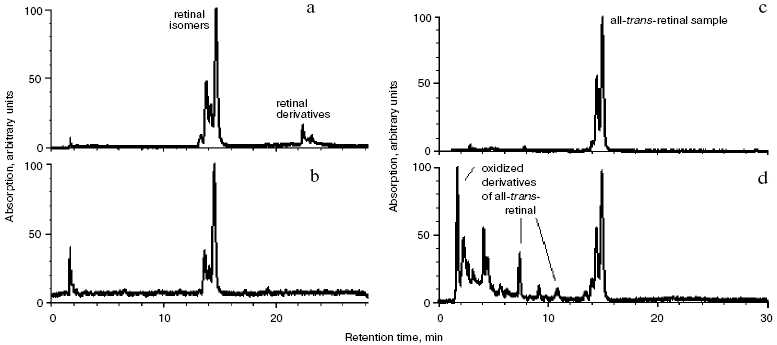
To determine the nature of ATR modification products, analysis of retinal isomers and its derivatives was performed by reverse-phase HPLC (Fig. 6).
HPLC analysis (Fig. 6, a and b) showed that new additional peaks appeared in the chromatogram. Most likely that they represent products of modification of exogenous ATR formed due to its interaction with rhodopsin and phosphatidylethanolamine in the photoreceptor membrane. The chromatograms show that at the beginning (13 min) retinal isomers are eluted (Fig. 6, a and b). These isomers are specific for dark ROS suspension with no exogenous ATR added. Then (23 min), ATR derivatives formed during ATR incubation with ROS suspension (3 days at 37°C) were eluted (Fig. 6a).Fig. 6. Analysis of retinal and its derivatives by HPLC (reverse-phase): a) chloroform-methanol extract of ROS suspension after incubation with 10-fold excess of ATR at 37°C; b) chloroform-methanol extract of ROS suspension; c) ATR (control); d) ATR and products of its oxidation. Detection at 430 nm. All chromatograms are normalized.
Analysis of products of free all-trans-retinal oxidation and degradation. We performed the analysis of products of free ATR oxidation and degradation formed as a result of ATR incubation for 3 days at 37°C (with no ROS suspension added) (Fig. 6, c and d).
The analysis demonstrated that the retention time of all these oxidized more polar products was less than that of the ATR sample (Fig. 6d) (less than 13 min). ATR modification products formed during its incubation with ROS suspension appeared on the chromatogram later, at approximately 23 min (Fig. 6a).
Therefore, based on chromatographic and spectral analysis we concluded that ATR added to ROS suspension in 10-fold molar excess was completely and covalently bound to amino groups of rhodopsin and phosphatidylethanolamine during the incubation of the mixture for 3 days at 37°C [25]. In other words, the mixture of products was formed, containing A2-rhodopsin, A2-PE, and probably, ATR-dimer-PE [26].
Absence of peaks corresponding to products of oxidation and degradation of free ATR on the chromatogram indicates that its interaction with amino groups of rhodopsin and phosphatidylethanolamine is more effective than its oxidation.
Influence of all-trans-retinal and its derivatives on rhodopsin regeneration after its bleaching with visible light. The main aim of the present work was to investigate the native properties of all-trans-retinal-modified rhodopsin, namely, its ability to regenerate after irradiation with visible light.
As already mentioned, regeneration in vitro means the restoration of the initial absorption spectrum of rhodopsin with maximum at 500 nm after its bleaching and subsequent addition of 11-cis-retinal.
Experiments were performed under different irradiation conditions: i) irradiation with soft light in the range 500-700 nm where absorption of ATR covalently attached to rhodopsin (A2-rhodopsin) and phosphatidylethanolamine (A2-PE) is virtually absent; ii) irradiation with hard light (400-700 nm), covering the absorption region of derivatives of all-trans-retinal with rhodopsin and phosphatidylethanolamine from the photoreceptor membrane.
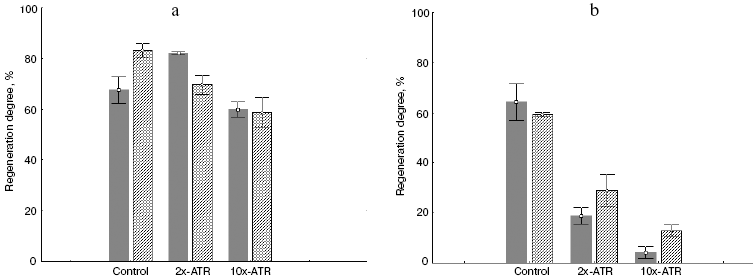
Figure 7 shows histograms representing degree of regeneration of native and rhodopsin modified with all-trans-retinal under different irradiation conditions. During soft irradiation (500-700 nm) photodamage in the presence of both free ATR (sample before incubation) and ATR bound with amino groups of rhodopsin and phosphatidylethanolamine (sample after incubation for 3 days at 37°C) is rather light (Fig. 7a). Rhodopsin regeneration capacity in these samples is almost totally preserved and remains very close to that of native visual pigment.
However, during irradiation with hard light (400-700 nm), i.e. in the absorption region of rhodopsin and phosphatidylethanolamine modified with all-trans-retinal, a different picture was observed. Rhodopsin regeneration degree both before and after incubation (3 days at 37°C) with added ATR dropped sharply (Fig. 7b).Fig. 7. Degree of regeneration for native rhodopsin and modified with all-trans-retinal under different irradiation conditions: a) after irradiation with 500-700 nm light for 120 min; b) after irradiation at 400-700 nm for 120 min. Dark columns, samples in presence of free ATR (before incubation); shaded columns, sample in presence of ATR derivatives (after incubation for 3 days at 37°C in the dark). Control, sample without exogenous ATR; 2x-ATR, samples with 2-fold excess of exogenous ATR; 10x-ATR, samples with 10-fold excess of exogenous ATR.
This indicates that it is in the blue region of the spectrum where derivatives of ATR with rhodopsin and phosphatidylethanolamine absorb light develops pronounced photodamaging activity.
The table shows the resulting data on regeneration of rhodopsin and its all-trans-retinal modification products under different irradiation conditions and at different concentrations of exogenous ATR.
Degree of regeneration for rhodopsin and products of its modification by
exogenous all-trans-retinal after visible light irradiation in
ranges of 500-700 and 400-700 nm for different periods of time
(15, 60, and 120 min) and at different concentrations of exogenous
all-trans-retinal
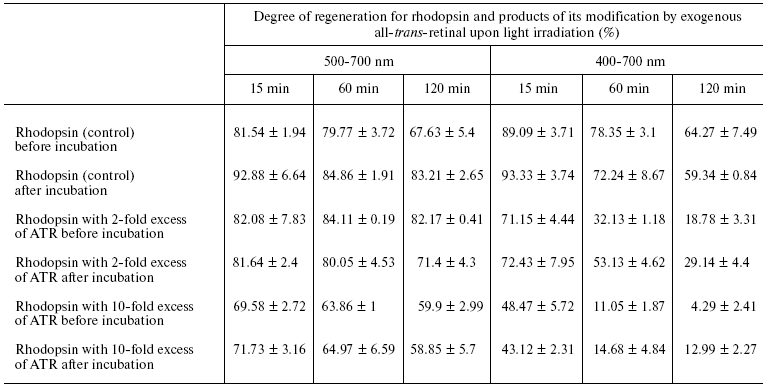
Figure 7 and the table indicate that impairment of the ability of rhodopsin to regenerate depends upon both sample irradiation time and ATR amount added to the ROS suspension. The longer the sample was irradiated and the more ATR was added, to less extent then was rhodopsin able to regenerate.
Blue light induced impairment of the ability of rhodopsin to regenerate was caused both by free ATR (just added to ROS suspension), and ATR bound to rhodopsin and phosphatidylethanolamine (after 3 days incubation at 37°C). Free ATR has noticeably stronger photodamaging effect on the rhodopsin molecule compared to bound retinal in modified products--rhodopsin (A2-rhodopsin) and phospholipid (A2-PE, N-bis-retinylidene-phosphatidylethanolamine; NR-PE, N-retinylidene- phosphatidylethanolamine) [23]. This suggests that photosensitizing ability of free ATR is more pronounced because the oxidation substrates themselves have chaperon-like effect.
DISCUSSION
The data accumulated to date on the mechanisms of light damaging action on retina and pigment epithelium indicates that excessive accumulation of ATR in the photoreceptor membrane of rod external segment can be considered as a risk factor aggravating the development of degenerative retinal diseases. It was shown that not only ATR per se but also products of its interaction with amino groups of phosphatidylethanolamine and rhodopsin [27, 28] are capable of acting as photosensitizers for oxidation of proteins and lipids in the photoreceptor membrane [12-17]. In this regard, it is essentially important to investigate damaging action of light on functional properties of rhodopsin in the photoreceptor membrane, and first of all on its ability to regenerate after irradiation with visible light in the presence of ATR excess. As known, damage and/or impairment of native conformation of the protein part of the rhodopsin molecule (apoprotein opsin) leads to difficulties or total inability of 11-cis-retinal to incorporate into the chromophore center of opsin, i.e. to restore dark conformation after bleaching.
According to the procedure by Fishkin et al. [25], we developed a model system allowing investigation of photosensitized damage of rhodopsin in the photoreceptor membrane of rod outer segment. Products of interaction of the sensitizer, exogenous ATR added in a large excess, with phosphatidylethanolamine and opsin, including substances modified with both two or one ATR molecule via interacting with amino groups of protein and/or lipid, were obtained and identified by spectroscopy and HPLC according to the protocol described.
Irradiation of this mixture of derivatives of all-trans-retinal with soft light in the range 500-700 nm did not lead to any significant difference in rhodopsin regeneration degree compared to the native sample (with no ATR added). Our results indicate that modification of three lysine residues of opsin with all-trans-retinal per se does not lead to deterioration the ability of the visual pigment to regenerate. In other words, modification of these three residues does not create steric hindrance for 11-cis-retinal incorporation into the opsin chromophore center.
Irradiation of native photoreceptor membranes with soft light immediately after the addition of ATR, i.e. not yet modified with retinal, did not impair the ability of rhodopsin to regenerate either (Fig. 7a). The absence of photodamaging effect can be easily explained since neither all-trans-retinal products nor retinal per se absorb a significant amount of light in the range 500-700 nm.
Entirely different results were obtained after irradiating samples with hard light in the range 400-700 nm. Both free ATR and modified products absorb strongly around 400 nm.
Figure 7b clearly shows the sharp drop of regeneration degree both in native samples of the photoreceptor membrane immediately after ATR addition and in samples containing modified products--A2-rhodopsin and A2-PE. These results are in good agreement with modern concepts on the mechanisms of damaging action of short wavelength visible (violet-blue) light on retina photoreceptor cells [4].
It should be noted that free ATR reveals heavier photodamaging influence on rhodopsin ability to regenerate compared to retinal bound to rhodopsin and phospholipid. Indeed, the degree of regeneration of visual pigment where all the exogenous ATR was covalently bound to the substrates--rhodopsin and phospholipid--was 1.5-2 times higher compared to that of samples containing free, non-bound ATR (Fig. 7b and the table). This can probably be explained with a known phenomenon, that free photosensitizer shows more pronounced phototoxic properties than that bound to substrate [28, 33].
It is interesting that during the incubation of photoreceptor membrane all the exogenous ATR was bound to amino groups of phosphatidylethanolamine and rhodopsin. We did not observe oxidized or degraded retinal in the samples of our study. This can indicate that in biological systems the reaction of retinal interaction with amino groups of proteins and lipids is competitive to the oxidation reaction of free retinal. This phenomenon is likely to play an important role in prevention from phototoxic properties of free retinal [33]. For example, when the rate of reduction of retinal to retinol by retinol dehydrogenase is not sufficient, retinal excess is converted to a less toxic form in the photoreceptor membrane. Nevertheless, despite the fact that ATR bound to amino groups of proteins and lipids is less phototoxic compared to free retinal, the effect of photosensitized damage to rhodopsin molecules, namely the impairment of its main physiological property--regeneration ability--is reasonably high (Fig. 7b and the table).
Therefore, the data obtained in the present work in the model system suggest that in vivo excessive accumulation of ATR in the photoreceptor membrane can be considered as a risk factor explaining the aggravating action of light during the development of degenerative retinal diseases, in particular during Stargardt's disease and age-related macular degeneration [34, 35].
This work was supported by Basic Research Programs of the Russian Academy of Sciences (RAS) Presidium “Theoretical and experimental studies of the nature of chemical bonding and mechanisms of key chemical reactions and processes” (Program No. 1 of the Division of Chemistry and the Materials Science of the RAS) and “Physiological mechanisms of the regulation of internal environment and organization of living systems” (Program of OBS RAS), by the Russian Foundation for Basic Research (grant 05-04-49945), and by State contract No. 02.512.11.2113 (2007-2-1.2-04-03-024) in the frame of the Federal Scientific and Technological Program “Research and development along the priority lines of science and engineering of Russia for 2007-2012”.
REFERENCES
1.Saari, J. C. (2000) Invest. Ophthalmol. Vis.
Sci., 41, 337-348.
2.Lamb, T. D., and Pugh, E. N. (2004) Prog. Retin.
Eye Res., 23, 307-380.
3.Weng, J., Mata, N. L., Azarian, S. M., Tzekov, R.
T., Birch, D. G., and Travis, G. H. (1999) Cell, 98,
13-23.
4.Ostrovsky, M. A. (2005) Uspekhi Biol. Khim.,
45, 173-204.
5.Travis, G. H. (1998) Am. J. Hum. Genet.,
62, 503-508.
6.Rozanowska, M., and Sarna, T. (2005) Photochem.
Photobiol., 81, 1305-1330.
7.Harper, W. S, and Gaillard, E. R. (2001)
Photochem. Photobiol., 73, 71-76.
8.Fu, P. P., Cheng, S.-H., Coop, L., Xia, Q., Culp,
S. J., Tolleson, W. H., Wamer, W. G., and Howard, P. C. (2003)
Environ. Carcinogen. Ecotoxicol. Rev., 21, 165-197.
9.Liu, R. S. H., and Hammand, G. S. (2003)
Photochem. Photobiol. Sci., 2, 835-844.
10.Pogogeva, I. D., Fedorovich, I. B., Ostrovsky, M.
A., and Emanuel, N. M. (1981) Biofizika, 26, 398-403.
11.Ostrovsky, M. A., and Fedorovich, I. B (1982)
Fiziol. Cheloveka, 8, 572-577.
12.Ostrovsky, M. A., and Fedorovich, I. B (1994)
Biofizika, 39, 13-25.
13.Ostrovsky, M. A., and Fedorovich, I. B. (1996)
Khim. Fiz., 15, 73-80.
14.Fedorovich, I. B., Semenova, E. M., Grant, K.,
Converse, C. A., and Ostrovsky, M. A. (2000) Curr. Eye Res.,
21, 975-980.
15.Sun, H., and Nathans, J. (2001) J. Biol.
Chem., 276, 11766-11774.
16.Ben-Shabat, S., Parish, C. A., Vollmer, H. R.,
Itagaki, Y., Fishkin, N., Nakanishi, K., and Sparrow, J. R. (2003)
J. Biol. Chem., 277, 7183-7190.
17.Suter, M., Reme, C., Grimm, C., Wenzel, A.,
Jaattela, M., Esser, P., Kociok, N., and Richter, C. (2000) J. Biol.
Chem., 275, 39625-39630.
18.Sparrow, J. R., Nakanishi, K., and Parish, C. A.
(2000) Invest. Ophthalmol. Vis. Sci., 41, 1981-1989.
19.Sparrow, J. R., and Cai, B. (2001) Invest.
Ophthalmol. Vis. Sci., 42, 1356-1362.
20.Bartl, F. J., Ritter, E., and Hofmann, K. K.
(2001) J. Biol. Chem., 276, 30161-30166.
21.Jager, S., Palczewski, K., and Hofmann, K. P.
(1996) Biochemistry, 35, 2901-2908.
22.Sachs, K., Maretzki, D., Meyer, C. K., and
Hofmann, K. P. (2000) J. Biol. Chem., 275, 6189-6194.
23.Buczylko, J., Saari, J. C., Crouch, R. K., and
Palczewski, K. (1996) J. Biol. Chem., 271,
20621-20630.
24.Kolesnikov, A. V., Golobokova, E. Y., and
Govardovskii, V. I. (2003) Vis. Neurosci., 20,
249-265.
25.Fishkin, N., Jang, Y.-P., Itagaki, Y., Sparrow,
J. R., and Nakanishi, K. (2003) Org. Biomol. Chem., 1,
1101-1105.
26.Fishkin, N. E., Sparrow, J. R., Allikmets, R.,
and Nakanishi, K. (2005) PNAS, 102, 7091-7096.
27.Kim, S. R., Nakanishi, K., Itagaki, Y., and
Sparrow, J. (2006) Exp. Eye Res., 82, 828-839.
28.Harper, W. S., and Gaillard, E. R. (2003)
Photochem. Photobiol., 78, 298-305.
29.Papermaster, D. S. (1982) Methods in
Enzymology, Vol. 81, Biomembranes, Pt. H, pp. 48-52.
30.Cusanovich, M. A. (1982) Methods in
Enzymology, Vol. 81, Biomembranes, Pt. H, pp. 443-447.
31.Pepe, I. M., and Schwemer, J. (1987)
Photochem. Photobiol., 45, 679-687.
32.Folch, J., Lees, M., and Stanley, G. H. S. (1957)
J. Biol. Chem., 226, 497-509.
33.Pawlak, A., Wrona, M., Rozanowska, M., Zareba,
M., Lamb, L. E., Roberts, J. E., Simon, J. D., and Sarna, T. (2003)
Photochem. Photobiol., 77, 253-258.
34.Cruickshanks, K. J., Klein, R., Klein, B. E., and
Nondahl, D. M. (2001) Arch. Ophthalmol., 119,
246-250.
35.Wang, J. J., Klein, R., Smith, W., Klein, B.,
Tomany, S., and Mitchell, P. (2003) Ophthalmology, 110,
1960-1967.