Features of Mitochondrial Energetics in Living Unicellular Eukaryote Tetrahymena pyriformis. A Model for Study of Mammalian Intracellular Adaptation
E. A. Prikhodko1,2, I. V. Brailovskaya3, S. M. Korotkov3, and E. N. Mokhova1*
1Belozersky Institute of Physico-Chemical Biology and 2Faculty of Bioengineering and Bioinformatics, Lomonosov Moscow State University, 119992 Moscow, Russia; fax: (495) 939-3181; E-mail: mokhova@genebee.msu.su3Sechenov Institute of Evolutionary Physiology and Biochemistry, Russian Academy of Sciences, 194223 St. Petersburg, Russia; fax: (812) 552-7931
* To whom correspondence should be addressed.
Received October 26, 2008; Revision received December 22, 2008
Tetrahymena pyriformis is used in diverse studies as a non-mammalian alternative due to their resemblance in many main metabolic cycles. However, such basic features of mitochondrial energetics as Δψ (electrical potential difference across the inner mitochondrial membrane) or maximal stimulation of respiration by uncouplers with different mechanisms of uncoupling, such as DNP (2,4-dinitrophenol) and FCCP (p-trifluoromethoxycarbonylcyanide phenylhydrazone), have not been studied in living ciliates. Tetrahymena pyriformis GL cells during stationary growth phase after incubation under selected conditions were used in this study. Maximal stimulation of cellular respiration by FCCP was about six-fold, thus the proton motive force was high. The DNP uncoupling effect was significantly lower. This suggests low activity of the ATP/ADP-antiporter, which performs not only exchange of intramitochondrial ATP to extramitochondrial ADP, but also helps in the uncoupling process. It participates by a similar mechanism in electrophoretic transport from matrix to cytosol of ATP4– and DNP anion, but not FCCP anion. Thus, in contrast with mammalian mitochondria, T. pyriformis mitochondria cannot rapidly supply the cytosol with ATP; possibly the cells need high intramitochondrial ATP. The difference between DNP and FCCP is hypothetically explained by low Δψ value and/or an increase in concentration of long-chain acyl-CoAs, inhibitors of the ATP/ADP-antiporter. The first suggestion is confirmed by absence of mitochondria with bright fluorescence in T. pyriformis stained with the Δψ-sensitive probe MitoTracker Red. These data suggest that T. pyriformis cells are useful as a model for study of mitochondrial role in adaptation at the intracellular level.
KEY WORDS: ATP/ADP-antiporter, membrane potential, mitochondria, Tetrahymena pyriformis, uncouplers, DNP, intracellular adaptationDOI: 10.1134/S0006297909040038
Abbreviations: BSA, bovine serum albumin; DNP, 2,4-dinitrophenol; FCCP, p-trifluoromethoxycarbonylcyanide phenylhydrazone; Δψ, transmembrane difference of electrical potential across the inner mitochondrial membrane; ΔpH, difference in hydrogen ion concentrations between the two sides of the inner mitochondrial membrane.
The unicellular eukaryote Tetrahymena pyriformis resembles
mammalian cells in a number of the main metabolic cycles [1, 2] and manifests similar or
higher sensitivity to diverse biologically active substances, including
some hormones [1-4]. Due to
resemblance to mammalian cells in both these and other properties,
T. pyriformis and some other protozoa are used instead of
mammalian cells in fundamental and applied studies.
The ciliate T. pyriformis can easily adapt to different environmental conditions, for instance, to a considerable decrease in oxygen concentration [1]. Tetrahymena pyriformis cells have ability to grow to high concentrations both in simple media containing organic substances and in synthetic media comprising only inorganic components [5].
Mitochondria isolated from T. pyriformis cells resemble those from rat liver in their cytochrome c content and in such characteristics of efficiency of oxidative phosphorylation as P/O ratio and other properties ([6] and references within).
An integral characteristic of mitochondrial energetics is the proton motive force (difference in the electrochemical potential of hydrogen ions across the inner mitochondrial membrane). It is composed of electrical component, Δψ, and concentration component, ΔpH [7]. In mammalian mitochondria most of the proton motive force is stored in the form of Δψ ([8] and references within). High Δψ value is a prerequisite for normal mammalian cell functioning. One of the most important functions of mammalian mitochondria is the rapid supply of cells with ATP synthesized during oxidative phosphorylation. The main function of the ATP/ADP-antiporter is electrophoretic exchange of intramitochondrial ATP4– for extramitochondrial ADP3–. Because this reaction is highly dependent upon Δψ [9, 10], the exchange can proceed at a rate close to that of ATP synthesis only at high Δψ value.
In addition to the main function, the ATP/ADP-antiporter participates in the uncoupling process of such uncouplers as long-chain fatty acids and 2,4-dinitrophenol (DNP) ([8, 11], see also reviews [12, 13] and references within). The molecular mechanism of this process is explained by the hypothesis called “fatty acid circuit” [14]. According to this hypothesis, the ATP/ADP-antiporter uses similar mechanisms for electrophoretic transport of ATP4– and fatty acid anion from matrix through the inner mitochondrial membrane. So, mediated by the ATP/ADP-antiporter long-chain fatty acid- or DNP-induced uncoupling can proceed only at high values of Δψ. It is important that some powerful uncouplers, such as p-trifluoromethoxycarbonylcyanide phenylhydrazone (FCCP), increase proton conductance of the inner mitochondrial membrane by a different molecular mechanism that is not mediated by the ATP/ADP-antiporter [15-17].
It should be added that Δψ value has not been even approximately estimated in living (moving) T. pyriformis cells and other ciliates.
MATERIALS AND METHODS
Tetrahymena pyriformis GL culture in stationary phase of growth was used in all experiments. Conditions of cell cultivation, their preparation for experiments, and methods have been described earlier [18]. During some series of experiments, the following modifications have been introduced. The cells were cultivated in suspension for 2 days in the liver broth buffered with 7 mM phosphate (pH 6.5). The cells taken during stationary phase of growth were washed free of the culture medium with saline medium composed of 30 mM KCl, 10 mM NaCl, 6 mM KH2PO4, and 5 mM Hepes (pH 7.4) and diluted with the same medium to concentration (0.7-1.5)·106 cells/ml and incubated in this medium without oxidation substrates for 1 day. Then the cells were diluted with the same incubation medium to concentration (0.3-0.5)·106 cells/ml of medium, and 2 ml of the suspension was poured in each from a number of small (4 cm in diameter) plastic Petri dishes. The cells in Petri dishes for most experiments were incubated with mixing during 5-20 min for aeration before measurement of respiration. In some experiments, the Petri dishes with cells were placed in a common moist chamber for a more prolonged incubation.
The cell suspension was transferred to a polarographic cell just before recording of oxygen consumption as described earlier [18]. The experiments were performed at 20-26°C. The oxygen consumption was recorded at 25°C.
In some experiments, a Petri dish with cells was placed under a microscope for observation of cellular movement and behavior before and just after registration of endogenous respiration. A Nikon Eclipse E200 microscope with phase-contrast system and long-focus objective lens was used. To demonstrate tracks of cellular movement, photos of the cell suspension were taken with a Cannon A640 digital camera.
Fixation of ciliates, their staining by MitoTracker Red, and analysis of images of fluorescence cells were performed as described earlier [18].
In some experiments, fluorescence in living cells was also observed visually under a Univar fluorescence microscope (Reichert, Austria). An Axiovert 200 M fluorescence microscope (Carl Zeiss, Germany) was used in some experiments as well.
Mitochondria were isolated from T. pyriformis cell suspension as described in [6] with some modifications; the cells were disintegrated with a French press. Kinetics of changes in Δψ was recorded by the safranin method [19] with an Aminco DW-2000 spectrophotometer in the dual wavelength mode. The difference in light absorption between 555 and 523 nm was recorded. The mitochondrial incubation medium contained 250 mM sucrose, 1 mM EGTA, 10 mM KCl, 3 mM KH2PO4, 5 mM Mops-KOH (pH 7.4), 0.1 mg/ml BSA (bovine serum albumin), and 10 µM safranin O. Oxygen consumption was recorded in the same incubation medium with additions of 5 mM succinate and 2 mM glutamate but without safranin O.
Oleic acid, oligomycin, fatty acid-free BSA, Hepes, Mops, and carboxyatractylate were from Sigma (USA); safranin O and KH2PO4 were from Serva (Germany); DNP was from Merck (Germany); FCCP and nigericin was from Fluka (Switzerland); formaldehyde solution and yeast extract were from MP Biomedicals (Germany); KCl was from Analar (USA); MitoTracker Red was from Molecular Probes (USA).
RESULTS
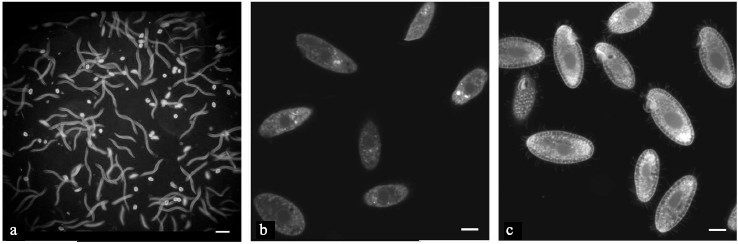
It should be emphasized that for all experiments the cells were taken in stationary growth phase and were prepared for registration of respiration under selected conditions as described above (see “Materials and Methods”). To ensure that the oxygen recording does not damage the infusorian, the movement of T. pyriformis cells was observed. Figure 1a demonstrates that the cells were moving at different rates. The tracks of cells with a low motility could be seen on the image as dots, while the faster moving cells have tracks of various lengths depending upon the movement velocity. In most experiments, we used visual observation only since strong light for making photos affects the movement of cells. It is known that some ciliates have a photoreceptor system and avoid illuminated places ([20] and references within).
A microscopic observation of the cells shortly after measurement of the initial respiration confirmed that the cells completely recuperated and exhibited a similar behavior as in a control assay. Some of the ciliates were close to the bottom of the Petri dish where they gathered around some food-like particles. A small portion of the cells was swimming either up or down. Some of the ciliates were swimming in the uppermost layer; it looked as if the ciliates needed to make contact with air from time to time after being in a hypoxic state. The absence of gross changes in shape, movement, and behavior strongly indicates that under our experimental conditions the metabolic state of mitochondria was assessed in living T. pyriformis cells. Such experiments also illustrate the high resistance of these ciliates to intervals of hypoxia.Fig. 1. Images of living (a, b) and formaldehyde-fixed (c) T. pyriformis cells: a) movement of cells, exposure was 1 sec. Scale bar 100 µm; b) living cells were stained with MitoTracker Red (2 µM, 10 min) and the image was taken immediately after several cells had ceased movement; c) cells were stained with MitoTracker Red (2 µM, 10 min) after formaldehyde fixation. b, c) Scale bar 10 µm.
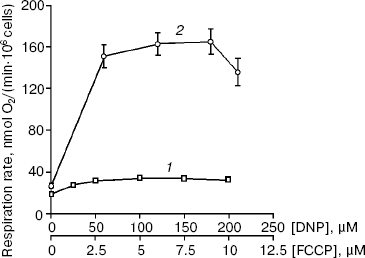
The maximal stimulation of endogenous cellular respiration by DNP was close to two (1.88 ± 0.06, n = 16) while that achieved by oleic acid was a little lower (1.60 ± 0.11, n = 10). Under the same conditions, FCCP produced a maximal stimulation of respiration close to six, about three times stronger than that with DNP (Fig. 2). The maximal stimulation of cell respiration by FCCP was obviously indicative of the high value of the mitochondrial proton motive force in these cells.
The great differences were subsequently reproduced in all experiments and appeared to be independent of the cultivation conditions and periods of food deprivation before experiments (from 3 h to 2 days). The same difference between these uncouplers was observed during recording of cellular respiration in assays with 20-30 mM pyruvate, which was added to the polarographic vessel before the T. pyriformis cells.
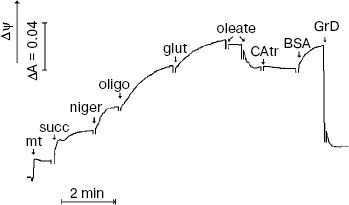
A limited number of mitochondria isolated from T. pyriformis cells were used mainly for recording of Δψ kinetics (Fig. 3). Oligomycin significantly increased Δψ in most experiments. Nigericin was added in about 18 assays and was found to increase Δψ in all of them when added at low concentrations. Oleic acid (2-5 µM) produced a clear decrease in Δψ in the presence of oligomycin. However, in these experiments a powerful specific inhibitor of the ATP/ADP-antiporter, carboxyatractylate, did not produce a stable increase in Δψ when added after oleic acid (Fig. 3). However, during experiments with rat liver mitochondria carboxyatractylate always increased Δψ under similar conditions [11].Fig. 2. Effects of DNP (1) and FCCP (2) on respiration of T. pyriformis cells. Before recording respiration, the cells were incubated for 1 day in medium containing 30 mM KCl, 10 mM NaCl, 6 mM KH2PO4, and 5 mM Hepes, pH 7.4. The respiration rates are mean values (from 10 to 15 experiments) ± S. E.
Recording mitochondrial respiration (see “Materials and Methods” for details) showed that maximal stimulation of respiration by DNP was about two when the incubation medium was supplemented with succinate (5 mM) and glutamate (2 mM). Thus, the maximal uncoupling effect of DNP was about equal in experiments with T. pyriformis cells and with mitochondria isolated from them.Fig. 3. Coupling effects of nigericin and oligomycin and uncoupling effect of oleic acid in mitochondria isolated from T. pyriformis cells. Incubation medium contained 250 mM sucrose, BSA (0.1 mg/ml), 1 mM EGTA, 10 mM KCl, 3 mM KH2PO4, 5 mM Mops-KOH (pH 7.4), and 10 µM safranin O. Additions: mt, mitochondria (0.3 mg protein/ml); succ, 5 mM succinate; niger, 25 nM nigericin; oligo, oligomycin (5 µg/ml); glut, 2 mM glutamate; oleate, oleic acid (2.5 µM × 2); CAtr, 1 µM carboxyatractylate, BSA, BSA (0.1 mg/ml); GrD, gramicidin D (1 µg/ml).
Such large difference between DNP and FCCP in maximal stimulation of respiration (see Fig. 2) was not observed earlier in experiments with cells or mitochondria isolated from mammalians tissues or other eukaryotes. Usually maximal respiration rates were about the same in the presence of the two uncouplers if measurements were performed under generally accepted conditions of control experiments. For example, DNP and FCCP produced strong and about the same maximal stimulation of respiration in experiments with thymus lymphocytes [21], salmon eggs [22], and rat liver mitochondria [11]. It should be noted that such studies have not been carried out earlier on moving unicellular organisms.
These results encouraged the beginning of a new series of more detailed experiments with Δψ-dependent fluorescence probes. As always, the cells were taken in a stationary growth phase. According to the data of electronic microscopy, during a logarithmic growth phase of T. pyriformis cells mitochondria were mainly concentrated near the cellular membrane [23]. During stationary growth phase the number of mitochondria increased, and they were found in different parts of the cells. Many lipid droplets were seen in the cytosol during this stationary phase of growth [23].
Tetrahymena pyriformis cells were stained with MitoTracker Red for observation of fluorescence in living cells. As a rule, this Δψ-dependent fluorescent probe is bound with mitochondria after transport into them. On many occasions, several structures with brighter fluorescence of rounded shape and different dimensions were observed within the cells. They might be mitochondrial aggregates induced by photodynamic damage.
However, in all experiments fluorescence of cellular structures including mitochondrial-like organelles was weak (Fig. 1b). A similar picture was observed in all cases: in cells moving at different rates and in those just after the cessation of movement, which were used for monitoring images of living cells (Fig. 1b).
It should be noted that under the same experimental conditions, including the use of the same fluorescence microscope, bright contrast fluorescence was observed in mitochondria of HeLa cells and other cultured cells stained with MitoTracker Red [24]. As earlier [18], many mitochondrial-like organelles with bright fluorescence were clearly seen in fixed cells (Fig. 1c), where the lipophilic cation should be concentrated in hydrophobic areas.
DISCUSSION
The uncoupling effects of DNP and FCCP are due to their participation in transport of protons across the hydrophobic barrier of the inner mitochondrial membrane. This results in partial dissipation of the proton motive force and in stimulation of respiration rate ([7, 8, 25-27] and references within). The most interesting and unanticipated result of this study is the large three-fold difference between FCCP and DNP in maximal stimulation of cellular respiration (Fig. 2).
It is important that the same difference between FCCP and DNP was also found during measurement of the influence of uncouplers on the conductance of artificial planar phospholipid membranes (Fig. 5 from [11]). Evidently, the artificial membranes do not contain the ATP/ADP-antiporter and other mitochondrial anion carriers. Figure 5 from [11] shows as well that under the same experimental conditions FCCP and DNP produced equal maximal stimulation of respiration of rat liver mitochondria. Together these data suggest that the weak uncoupling effect of DNP in T. pyriformis (Fig. 2) reflects a low activity of the ATP/ADP-antiporter and, possibly, of other anion carriers similar to those participating in long-chain fatty acid-induced uncoupling in liver mitochondria [28] and/or others which are specific for T. pyriformis mitochondria.
Earlier data were obtained which demonstrated that a large decrease in Δψ produced different effects on the uncoupling induced by FCCP and palmitic acid (which has DNP-type mechanism of uncoupling) [27]. The proton conductance in rat liver mitochondria was measured by the “acid pulse” method [26]. For strong decrease in Δψ, the incubation medium and the mitochondrial suspension were bubbled with argon. As a result, the palmitic acid-induced conductance was decreased by about three-fold while the FCCP-induced conductance was decreased by only 14% [27].
The assumption about low Δψ in T. pyriformis under our experimental conditions seems to be a likely explanation of all the data. This hypothesis predicts both low fluorescence of mitochondria in living T. pyriformis cells stained with MitoTracker Red (Fig. 1b) and a sharp difference between DNP and FCCP in maximal stimulation of cellular respiration in these cells (Fig. 2).
In the case of low Δψ, most of the proton motive force in mitochondria should be stored in the form of ΔpH; the ΔpH value should be high as FCCP strongly stimulated respiration of these cells (Fig. 2). If this is true, considering low Δψ and high ΔpH, the energetics of mitochondria in living T. pyriformis cells more closely resembles that of chloroplasts or some bacteria than mammalian mitochondria ([8, 29] and references within). Additional evidence in support of the hypothesis is that low Δψ and high ΔpH give great advantages to ciliates for free living in natural water reservoirs where they should exist under occasional conditions of hypoxia, fasting, and other similar unfavorable factors. One of the advantages of the proton motive force storage mainly in the form of ΔpH is the greater buffer capacity of the mitochondrial membrane energy as compared with its storage as Δψ [8, 29]. High ΔpH accelerates the transport of some oxidative substrates in mitochondria.
Some decrease in Δψ should decrease the exchange of intramitochondrial ATP for extramitochondrial ADP through the inner mitochondrial membrane via the ATP/ADP-antiporter [9, 10]. However, for T. pyriformis cells a decrease in this exchange could be useful since it should prevent the dangerous exhaustion of the intramitochondrial ATP pool. It would be interesting to determine whether this intramitochondrial ATP is used for other diverse mitochondrial functions [8, 29], including participation of mitochondria in nuclear degradation in apoptotic processes [30], or it is necessary to preserve mitochondrial structures or high intramitochondrial ATP is necessary for support of the cell state under the experimental conditions.
Changes in other Δψ-dependent functions should be determined by features of their dependence on Δψ and a degree of the Δψ dissipation.
There is another mechanism that can decrease the ATP/ADP-antiporter activity in the described experiments. Fats become the preferential oxidative substrates during fasting, hibernation, and some other states. This results in increase in long-chain acyl-CoA concentrations; they inhibit the ATP/ADP-antiporter activity [31, 32]. A rise in fatty droplets was observed in T. pyriformis cells during the stationary growth phase [23].
It is impossible to exclude one more explanation. Mitochondria of ciliates could have an isoform of the ATP/ADP-antiporter with low activity. The answer to this question can be obtained from experiments with mitochondria isolated from infusorians.
It is very important to clarify whether the results described in this paper are reproduced in experiments with T. pyriformis cells taken during exponential phase of growth and with other protozoa characterized by high mobility.
The great advantage of using ciliates and some other protozoans as models for studying the role of mitochondria in intracellular adaptation should be emphasized. Ciliates show that some environmental factor is unfavorable for them by demonstrative change in shape and in movement [5, 33, 34]. Titration of respiration by DNP and FCCP gives valuable information about the state of mitochondrial energetics. The minimal concentration of DNP that induced the maximal uncoupling (Fig. 2) is probably dependent on the concentration of anion carriers participating in the uncoupling under the experimental conditions. It is possible and reasonable to measure the maximal stimulation of respiration by DNP and FCCP and other mitochondrial energy coupling properties in parallels experiments with living cells and mitochondria isolated from them. Analysis of the study results and published data lead to the suggestion that this model is suitable for study of mitochondrial energetics during hibernation and similar states.
Investigations of the ATP/ADP-antiporter properties in experiment with isolated from T. pyriformis mitochondria is one of the approaches for studying basic features of mitochondrial energetics in cells, which are used as an alternative to mammalian cells in diverse studies.
However, it is more important but difficult to clarify whether these ancient unicellular organisms contain supramolecular protein complexes located in the contact sites between inner and outer mitochondrial membrane; they include the ATP/ADP-antiporter and other proteins which regulate cellular energetics ([35] and references within). A number of weighty arguments led to the hypothesis that these complexes “govern” not only energetics but the fate of cells [35]. Such studies with ciliates and other protozoans might give information concerning the origin or ancient prototypes of these structures.
The authors express their heartfelt thanks to D. B. Zorov, V. P. Skulachev, and A. A. Starkov for helpful advice and constructive criticism.
This work was supported by the Russian Foundation for Basic Research (grant 05-04-49700).
REFERENCES
1.Levy, M. R. (1973) in Biology of Tetrahymena
(Elliott, A. M., ed.) Dowden, Hutchinson and Ross, Inc, Stroudsburg,
Pennsylvania, pp. 227-257.
2.Hutner, S. H., Baker, H., Frank, O., and Cox, D.
(1973) in Biology of Tetrahymena (Elliott, A. M., ed.) Dowden,
Hutchinson and Ross, Inc, Stroudsburg, Pennsylvania, pp. 411-433.
3.Kohidai, L., Vakkuri, O., Keresztesi, M.,
Leppaluoto, J., and Csaba, G. (2002) Cell Biochem. Funct.,
20, 269-272.
4.Csaba, G., and Pallinger, E. (2008) Cell
Biochem. Funct., 26, 303-308.
5.Sauvant, M. P., Pepin, D., and Piccini, E. (1999)
Chemosphere, 38, 1631-1669.
6.Kilpatrick, L., and Erecinska, M. (1977)
Biochim. Biophys. Acta, 460, 346-363.
7.Mitchell, P. (1961) Nature, 191,
144-148.
8.Skulachev, V. P. (1988) Membrane
Bioenergetics, Springer-Verlag, Berlin.
9.Kramer, R., and Klingenberg, M. (1980)
Biochemistry, 19, 556-560.
10.Vignais, P. V., Block, M. R., Boulay, F.,
Brandolin, V., and Lauquin, G. J. M. (1985) in Structure and
Properties of Cell Membrane (Bengha, V., ed.) Vol. 2, CRC Press,
Paris, pp. 139-179.
11.Andreyev, A. Yu., Bondareva, T. O., Dedukhova, V.
I., Mokhova, E. N., Skulachev, V. P., Tsofina, L. M., Volkov, N. I.,
and Vygodina, T. V. (1989) Eur. J. Biochem., 182,
585-592.
12.Skulachev, V. P. (1998) Biochim. Biophys.
Acta, 1363, 100-124.
13.Mokhova, E. N., and Khailova, L. S. (2005)
Biochemistry (Moscow), 70, 159-163.
14.Skulachev, V. P. (1991) FEBS Lett.,
294, 158-162.
15.Starkov, A. A., Dedukhova, V. I., and Skulachev,
V. P. (1994) FEBS Lett., 355, 305-308.
16.Starkov, A. A., Bloch, D. A., Chernyak, B. V.,
Dedukhova, V. I., Mansurova, S. E., Severina, I. I., Simonyan, R. A.,
Vygodina, T. V., and Skulachev, V. P. (1997) Biochim. Biophys.
Acta, 1318, 159-172.
17.Starkov, A. A. (2006) Chem. Biol.
Interact., 161, 57-68.
18.Brailovskaya, I. V., Kudryavtseva, T. A.,
Larionov, V. N., Prikhodko, E. A., and Mokhova, E. N. (2007) Doklady
Biokhim. Biofiz., 413, 72-75.
19.Akerman, K. E., and Wikstrom, M. K. (1976)
FEBS Lett., 68, 191-197.
20.Sobierajska, K., Fabczak, H., and Fabczak, S.
(2006) J. Photochem. Photobiol. B, Biol., 83,
163-171.
21.Markova, O. V., Mokhova, E. N., and Tarakanova,
A. N. (1990) J. Bioenerg. Biomembr., 22, 51-59.
22.Holcomb, M., Cloud, J. G., Woolsey, J., and
Ingermann, R. L. (2004) Comp. Biochem. Physiol., Part A. Mol.
Integr. Physiol., 138, 349-354.
23.Elliott, A. M., and Bak, I. J. (1964) J. Cell
Biol., 20, 113-129.
24.Chernyak, B. V., Izyumov, D. S., Lyamzaev, K. G.,
Pashkovskaya, A. A., Pletjushkina, O. Y., Antonenko, Y. N., Sakharov,
D. V., Wirtz, K. W., and Skulachev, V. P. (2006) Biochim. Biophys.
Acta, 1757, 525-534.
25.Liberman, E. A., Mokhova, E. N., Skulachev, V.
P., and Topaly, V. P. (1968) Biofizika, 13, 188-193.
26.Mitchell, P., and Moyle, J. (1967) Biochem.
J., 104, 588-600.
27.Brustovetsky, N. N., Dedukhova, V. I., Egorova,
M. V., Mokhova, E. N., and Skulachev, V. P. (1991) FEBS Lett.,
295, 51-54.
28.Samartsev, V. N., Smirnov, A. V., Zeldi, I. P.,
Markova, O. V., Mokhova, E. N., and Skulachev, V. P. (1997) Biochim.
Biophys. Acta, 1319, 251-257.
29.Skulachev, V. P. (2003) in Selected Topics in
the History of Biochemistry: Personal Recollections VII (Comprehensive
Biochemistry) (Semenza, G., and Turner, A. J., eds.) Vol. 42,
Elsevier Science B. V., pp. 319-410.
30.Kobayashi, T., and Endoh, H. (2005) FEBS
J., 272, 5378-5387.
31.Lerner, E., Shug, A. L., Elson, C., and Shrago,
E. (1972) J. Biol. Chem., 247, 1513-1519.
32.Panov, A. V., Konstantinov, Y. M., and
Lyakhovich, V. V. (1975) J. Bioenerg., 7, 75-85.
33.Dias, N., Mortara, R. A., and Lima, N. (2003)
Toxicol. in vitro, 17, 357-366.
34.Prlina, I. S., Gabova, A. V., Raikov, I. B., and
Tairbekov, M. G. (1989) Tsitologiya, 31, 829-838.
35.Brdiczka, D., Zorov, D. B., and Sheu, S. S.
(2006) Biochim. Biophys. Acta, 1762, 148-163.