REVIEW: Pathways of Formation of Pigment Forms at the Terminal Photobiochemical Stage of Chlorophyll Biosynthesis
O. B. Belyaeva* and F. F. Litvin
Biological Faculty, Lomonosov Moscow State University, 119991 Moscow, Russia; E-mail: olgabelyaeva@mail.ru* To whom correspondence should be addressed.
Received May 13, 2009; Revision received May 22, 2009
The pathways of transformation of the chromophore of pigment–protein complexes have been studied at the terminal light-dependent stage of chlorophyll biosynthesis in plant leaves. The overall scheme of the sequence of photochemical and dark reactions of the pigment chromophore initiated by the reaction of photochemical hydration of a molecule of the precursor (protochlorophyllide) is presented. Schemes of the transformations of the components of the photoactive protochlorophyllide-oxidoreductase complex are discussed. Data are presented of features of the process at different stages of the formation of the pigment apparatus of plants.
KEY WORDS: chlorophyll, chlorophyll biosynthesis, protochlorophyllide, protochlorophyllide-oxidoreductase, photosystemsDOI: 10.1134/S0006297909130070
Abbreviations: Chl, chlorophyll; Chld, chlorophyllide; PChld, protochlorophyllide; Pheo, pheophytin; PLB, prolamellar bodies; POR, protochlorophyllide-oxidoreductase; PS I(II), photosystem I(II); RC, reaction center.
The synthesis of chlorophyll (Chl) a in the absence of light is
inhibited at the stage of the formation and accumulation of the direct
precursor of Chl, protochlorophyllide (PChld), in the etiolated leaves.
This pigment differs from Chl by the absence of a phytol substituent
and presence of a double (not single) bond in pyrrole ring D of the
macrocycle. The termination of the process goes as a fast
photoreaction. This terminal stage can be observed on exposure to light
by the changes in the absorption and fluorescence emission spectra of
the etiolated leaves and the appearance of the reaction product,
chlorophyllide (Chld) a in the extract of the pigments.
Thus, from the chemical viewpoint, the terminal stage of Chl biosynthesis is based on the photochemical reaction of the selective hydration of the double bond in the tetra-pyrrole macrocycle and appearance of the chlorine structure, principally different from the precursor in spectral and chemical properties, which allowed it to play the role of the main photosynthetic pigment. Later, at this terminal stage phytol addition takes place, i.e. chlorophyllide is transformed into chlorophyll.
At the beginning of the studies, the phototransformations of the precursor were regarded as a simple photochemical reaction similar to the photoreactions going in the pigment solution. However, already in the first experiments, under illumination of the etiolated leaves some spectral bands quickly changing each other were observed. With the development of spectroscopic techniques, and, in particular, low-temperature fluorescence spectroscopy, due to the efforts of several laboratories it was possible to clarify the complicated picture of intertransformation of pigment–protein complexes of the precursor and chlorophyllide and obtain important information concerning the branching chain of the dark and photochemical reactions responsible for the observed spectral effects (Fig. 1).
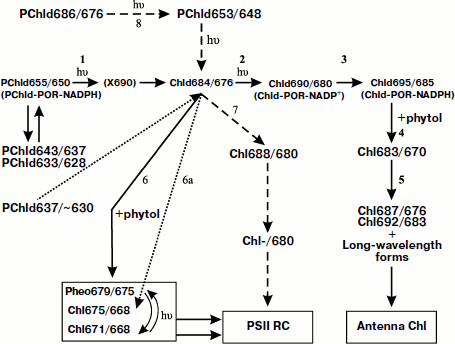
In the recent decades, the progress of the studies of the terminal stage of Chl biosynthesis is connected with discovery of protochlorophyllide-oxidoreductase (POR), a “photoenzyme” catalyzing the photoreaction of PChld transformation to Chld. As for the act of photochemical hydration, it takes place in a special complex including the precursor, photoenzyme, and donor of hydrogen, NADPH.Fig. 1. General scheme of pigment chromophore transformations at the light stage of chlorophyll biosynthesis in plant leaves [3-25, 41-48, 56-58]. PChld, protochlorophyllide; Chld, chlorophyllide; Chl, chlorophyll. The figures indicate the positions of the fluorescence maxima (first index) and absorption maxima (second index) of the pigment forms. See text for the interpretations of reactions 1-8. Dashed arrows show the pathway of formation of the pigment Chl-/680 (pigment P-680) from the long-wavelength form of protochlorophyllide. The dotted arrows show the pathway of transformation of the short-wavelength protochlorophyllide in embryo leaves. X690 is a non-fluorescent intermediate [80-89].
Modeling of the natural process in artificial ternary complexes provided new information about the mechanism of the photoreaction and started a deeper study of the physical nature of the primary photoprocesses and the structure of the complex. At the same time, comparison of the processes observed in the model systems and in whole cells brought us to the conclusion that the natural process is much more complicated, not only in the number of intermediate pigment forms involved in it, but also in variability of the resulting products.
In the course of the investigations, it became clear that the terminal stages of chlorophyll biosynthesis should not be regarded only as the termination of the chemical synthesis of the pigment, but also as the start of the pathway of the transformation of pigment–protein complexes of the precursor into the pigment–protein complexes forming the structural basis of the photosynthesis apparatus. It became possible to observe the moment of incorporation of chlorophyll forms formed from the precursor into the two photochemical systems of photosynthesis. Thus, the study of the terminal stage of chlorophyll biosynthesis acquires more general character as a study of the biogenesis of photoactive pigment structures of photosynthesis.
An “ontogenetic” approach to the studies of the pigment apparatus of photosynthesis seems rather promising for understanding of the evolution of photosynthesis, the ways that Nature has come to the discovery of the unique, extremely efficient, and ecologically pure method of accumulation and conversion of solar energy. Perhaps the results of such studies will be useful in the future also for the solution of the problem of auto-renewal of the elements of artificial systems of solar energy accumulation.
This survey is mainly devoted to description and generalization of results concerning the scheme of the pathways of the main components in the active complex consisting of the chromophore of pigment–protein complexes, POR, and hydrogen donor at the terminal photo-dependent stage of chlorophyll biosynthesis in plant leaves.
GENERAL SCHEME OF THE SEQUENCE OF CHLOROPHYLL PHOTOBIOSYNTHESIS
REACTIONS in vivo
Due to the work of many scientists, the general sequence and mechanisms of the reactions involved in the final light-dependent stage of Chl formation from its precursor, PChld, has gradually been becoming clear (see reviews [1, 2]). The results of these studies can be summarized in a scheme (Fig. 1) that seems now the best-grounded and most complete. The scheme presents a branching chain of transformations of pigment–protein complexes leading to the formation of several native chlorophyll forms: the main pigment mass of the light-harvesting antenna and of minor but functionally significant pigment forms included in the two photochemical systems of photosynthesis.
The linear reaction chain (reactions 1-5 in the scheme) includes two consecutive photochemical reactions (1 and 2 in the scheme) [3-11] and further dark processes [3-13]. Judging by changes of the character of the circular dichroism spectra for homogenates from etiolated leaves exposed to illumination, it can be supposed that in the first photoreaction (1) one of the molecules of protochlorophyllide dimer is involved, due to which a complex is formed of two weakly bound molecules: protochlorophyllide and chlorophyllide. In the second photoreaction, phototransformation of the second protochlorophyllide molecule occurs [7, 8].
The temperature-dependent long-wavelength spectral shift Chld690/680 → Chld695/685 (reaction 3 in the scheme) occurs without the participation of light [3, 4, 12]. It depends on the reconstruction of the reduced form of hydrogen donor NADPH in the pigment–protein complex (the reduction of NADP+ formed in the course of the primary reaction) [11, 14-17]. As follows from the studies of energy migration, it was suggested that the bathochromic shift of the spectral bands in this reaction might be connected with a decrease in intermolecular distances in the aggregated active complex [18]. The considered stage of the process probably depends on two factors: alteration of the structure of the aggregated complex and reconstruction of NADPH under condition that the structure of POR complex and chromophore–chromophore interaction depend on the state of NADPH (oxidized or reduced).
Reaction 4 in Fig. 1 is manifested as a short-wavelength shift of the spectral bands of the pigment Chld695/685 → Chl683/670 connected with the name of the Japanese researcher K. Shibata—the “Shibata shift” [19, 20]. However, the first to observe this spectral effect under illumination of etiolated leaves were Russian scientists Monteverde and Ljubimenko [21]. The Shibata shift is observed both in whole etiolated leaves and in their homogenates and in the preparations of pigment–protein complexes [22-25]. At this stage in plant leaves etherification of a chlorophyllide molecule occurs with formation of chlorophyll [22, 23, 26-28].
A group of experimental facts indicates that the short-wavelength Shibata shift is connected with disaggregation of the pigment. The evidence for this is an increase in Chl fluorescence yield [29-31], disappearance of the double signal of circular dichroism [8, 32, 33], and disturbance of energy migration from protochlorophyllide to chlorophyll [30, 31, 34]. In estimating the initial distance between the protochlorophyllide molecules in the active complex of the order of 25 Å, Thorn advanced the proposition that the disturbance of energy migration might be indicative of chlorophyll and non-converted protochlorophyll moving apart from each other by approximately 10 Å at this stage [31]. Results of studies using fractionation of prolamellar bodies (PLB) with the method of isoelectric focusing led to the conclusion that the Shibata shift causes not the destruction of the complex but its disaggregation with transformation of large POR aggregates in smaller ones [35]. The disaggregation of POR might, as well, lead to disaggregation of the pigment. Perhaps it is loosening of the complex that promotes the etherification—the addition of the hydrophobic alcohol, phytol, to the pigment molecule.
The terminal link of the linear reaction chain (reaction 5 in Fig. 1) is exhibited by the long-wavelength spectral shift Chl683/670 → Chl687/676; Chl692/683. This stage is not connected with the action of light; it begins with a decrease in the quantum yield of the chlorophyll fluorescence from Chl683/670, which was formed as a result of the Shibata shift. This terminal stage corresponds to the final integration of chlorophyll into different pigment–protein complexes of thylakoid membrane and to the formation of the chlorophyll spectral forms typical of a mature green leaf [36].
The linear reaction chain apparently leads to the synthesis of chlorophyll a of light-harvesting antenna (the main mass of the pigment); this is indicated by the formation of energy connection (electron excitation energy transfer) of chlorophyll a with carotenoids and chlorophyll b [37, 38].
Chlorophyll Biosynthesis Chain Branching. Biosynthesis of Pheophytin a—a Component of the Reaction Center (RC) of Photosystem II (PS II)
The intermediate Chld684/676 formed as a result of the first photoreaction (reaction 1, Fig. 1) serves as the point of branching. In the dark a side reaction is observed—the transformation of this intermediate into a short-wavelength chlorophyll form Chl675/670 (reaction 6 in the scheme of Fig. 1) [5, 6, 8, 11]. This reaction goes at a noticeable rate only at temperatures above 273 K. It can also be observed under conditions of illumination of etiolated leaves by low-intensity light at temperatures above 0°C. The energy migration from the form Chl675/670 to Chld684/676 is evidence of it being in close proximity to the center of biosynthesis of the main mass of the pigment [18]. Analysis of pigment extracts by thin layer chromatography has shown that the product of the side dark reactions is not chlorophyllide but chlorophyll [5, 6]. Consequently, the etherification of the Chld molecule occurs not only during the Shibata shift (as believed earlier) but also at the stage of the discussed dark reaction of the formation Chl675/670 from the product of the first photoreaction, the rate of the shift being by an order of magnitude higher. The existence in the Chl biosynthesis process of two ways of chlorophyllide etherification, fast and slow, was also shown elsewhere [39, 40].
Judging by the fact that this reaction is manifested by the short-wavelength shift and also by the resistance of the product Chl675/670 to the disaggregating actions and higher (as compared to that of Chld690/680) extractive ability of this product [5, 6], the side reaction is apparently accompanied by pigment disaggregation. That is confirmed by studies of circular dichroism spectra of the homogenates obtained from illuminated etiolated leaves at this stage of the process [7]. It can be assumed that the same as at Shibata shift stage disaggregation promotes the enzymatic process of etherification the pigment molecule. Besides, it appears that the terminal form of chlorophyll formed as a result of the side reaction earlier designated as Chl675/670, in reality represents two forms of chlorophyll, Chl671/668 and Chl675/668 [41, 42].
Using low-temperature fluorescence spectroscopy [41-43], it has been shown that as a result of the “side reaction” alongside with Chl a, pheophytin a (Pheo679/675) is synthesized (reaction 6), which is shown by typical fluorescence excitation and fluorescence emission spectra of the illuminated etiolated leaves and of pigment extracts.
The reaction of pheophytin formation has biosynthetic rather than destructive character: it proceeds only in leaves when the pigment–protein complexes are intact and is not revealed even in the homogenates from the etiolated leaves. Additional studies showed a greater complication with the “side reaction” of the process [44]. Pheophytin appeared to be able to undergo conversion to chlorophyll Chl671/668 in the course of the dark reaction, and this reaction is photoconvertible.
The complex of pheophytin and chlorophyll of etiolated leaves is identical in a number of properties to the pheophytin-containing RC of PS II [41-44]. However, a high quantum yield of Pheo fluorescence of greening leaves indicates the different states of Pheo molecules in these structures from those in RC.
Biosynthesis of Long-Wavelength Chlorophyll, a Possible Component Of PS II RC (reaction sequence 7 in Fig. 1)
The formation of the long-wavelength chlorophyll form, in spectral characteristics close to the chlorophyll a component of the PS II RC, was first observed under illumination of etiolated leaves under conditions of heat shock [45, 46]. In those studies, a new dark reaction of the product of photoreduction of protochlorophyllide Chlde684/676 was localized at the point of reaction chain branching. The reaction is manifested by the bathochromic shift of the spectral bands and accompanied by the etherification of the pigment to chlorophyll: Chld684/676 → Chl688/680. After the completion of the reaction, fast (20-30 sec) and full quenching of the fluorescence of its product occurred: Chl688/680 → Chl-/680. The authors supposed that the final product of this chain of dark reactions, non-fluorescent chlorophyll Chl-/680, is the pigment of the PS II RC, P-680 [45, 46].
Studies of Chl biosynthesis in juvenile (3-4-day-old) etiolated leaves provided additional data on the mechanism of biosynthesis of P-680 under natural conditions [47, 48]. It was shown that in these juvenile leaves, like in 7-10-day-old leaves grown under conditions of heat shock, the intermediate Chld684/676 at room temperature participates in two dark reactions: Chld684/676 → Chl675/670 (“side reaction”) and Chld684/676 → Chl688/680 → Chl-/680. The peculiarity of the process in the juvenile plants was accumulation of Chld684/676 during the first 3-5 sec of illumination with white light without marked changes in the absorption and fluorescence bands of PChld655/650, although photoconversion of protochlorophyllide (judging by the extracts) took place. Experiments using monochromatic light in the long-wavelength region (680 nm), or with white light illumination at different temperatures, showed that the effect is accounted for by the photoconversion of an earlier unknown weakly fluorescent long-wavelength form of PChld686/676 into PChld653/648 (reaction 8) which, as a result of the next light reaction, is transformed into Chld684/676. Thus, at the early stages of plant development, alongside with the main branching chain of reactions, a parallel branching process was found leading to the synthesis of the non-fluorescent Chl-/680 (perhaps the pigment of PS II RC) from the long-wavelength PChld686/676 via the stage of the formation of the active form of PChld653/648.
Thus, the native form of Chld684/676, the product of the first photochemical reaction, formed from PChld655/650, presents a point of triple branching of the biosynthesis chain of the minor forms of chlorophyll and pheophytin connected with PS II RC and the main mass of chlorophyll of the antenna.
Biosynthesis of Chlorophyll of PS I RC
Studies of Chl formation in the cells of the heterotrophic mutant Chlorella vulgaris B-15 with full genetic block of the dark chlorophyll synthesis [49, 50] revealed photoactivity of another long-wavelength minor form of protochlorophyll, PChl682/672 (etherified form). A considerable amount of this form is accumulated in the cells of the mutant during dark cultivation. Under illumination of the mutant cells in the temperature range from –70°C to 26°C, alongside with earlier described [51, 52] photoreactions (PChld655/650 → Chld695/684 and PChld640/635 → Chld680/670), the photoconversion of the long-wavelength form PChl682/672 into the stable (terminal) form Chl715/696 was revealed [49, 50]. It has been proposed that Chl715/696 is the pigment of the core of PS I. In favor of this hypothesis is the position of its long-wavelength band (696 nm), close to the position of the absorption band P700 (697 nm of chlorophyll of PS I RC in Chlamydomonas [53]). Photoactivity of PS I (photostimulated release of H2) starts to be manifested simultaneously with Chl715/696 formation in the light [54].
The efficiency of electron excitation energy transfer from carotenoids to PChl682/672 and Chl715/696 in the course of the photoreaction did not change significantly [49, 50], which is possible with the preserved constant intermolecular distances and mutual orientation of chromophores of carotenoids and porphyrins. Based on this, the authors hypothesized that in the isolated mutant cells the structure of the core of PS I already existed, containing PChl682/672 instead of chlorophyll, and intercomplex photoreduction of protochlorophyll to chlorophyll completes the formation of the core. As POR is known to be specific to the substrate and not to reduce the etherified protochlorophyll [55], it can be assumed that in this case protochlorophyll photoreduction involves some earlier unknown type of protochlorophyllide-oxidoreductase.
Photobiosynthesis of Chlorophyll in Embryo Plant Leaves
In chlorophyll biosynthesis in embryo leaves of dicotyledonous plants, the pathways of Chl biosynthesis differ from the reaction sequences described above for the juvenile and mature etiolated plants. In the dark, in the embryo leaves predominantly the short-wavelength form of the precursor is accumulated characterized by a band of low-temperature fluorescence at 632-635 nm. Simultaneously, there exists a longer-wavelength form with a low-temperature fluorescence band at 653-655 nm. In the fluorescence spectra measured at room temperature, the main maximum is situated at 636-637 nm [56-58]. The short-wavelength form of the embryo leaves appeared to be photoactive. Its photoconversion into the short-wavelength form Chl675/670 occurs under the action of light. In accord with the data of [56-58], the conversion proceeds via the formation of the intermediate Chld684/676, i.e. by the pathway of the “side” reaction (dotted arrows in Fig. 1). In the embryo leaves the “side” reaction goes at a high rate: during 5 sec [56]. It is known that in proplastids of very young (2-3-day-old) leaves the prolamellar bodies (PLB) are still absent, only small stromal membranes are formed [57, 59-61]. The high rate of the short-wavelength shift Chld684/676 → Chl675/670 observed in the embryo leaves at room temperature [56] is indicative of fast pigment release from the active site of POR.
CYCLIC SCHEME OF TRANSFORMATIONS OF COMPONENTS OF THE ACTIVE
PROTOCHLOROPHYLLIDE–ENZYMIC COMPLEX IN THE PROCESS OF CHLOROPHYLL
BIOSYNTHESIS
At present much attention is given to studies of the behavior of the protein component of the active complex and hydrogen donor NADPH in protochlorophyllide photoreduction.
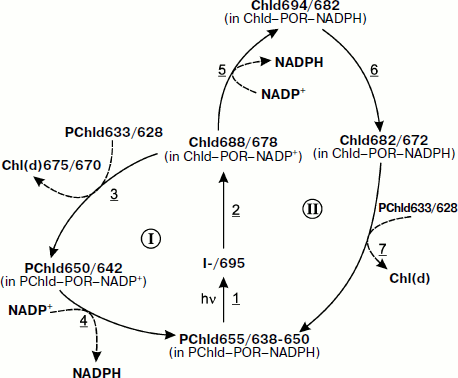
Already in the early studies of pigment–protein complex of protochlorophyllide, a “shuttle” (repeated) mechanism of the photoenzyme function in the chlorophyll biosynthesis was proposed. In a recent paper [57], a general scheme of Chl formation was proposed including two pathways of Chl biosynthesis and of regeneration of the photoactive complex protochlorophyllide – two cycles of repeated use of POR (Fig. 2).
A scheme by Schoefs and Franck, in contrast to the scheme given in Fig. 1, includes only one light-dependent reaction—the formation of chlorophyllide with absorption maximum at 678 nm, fluorescence maximum at 688 nm, which is transformed into a longer-wavelength form via a dark reaction. As at room temperature and at high light intensity it is difficult to observe separately two sequential photoreactions [3-6], the spectral characteristics of the primary chlorophyllide form recorded by Schoefs and Franck (absorption maximum at 678 nm, fluorescence maximum at 688 nm) in the leaves illuminated by an intense flash of light apparently correspond to the simultaneous accumulation of the products of both the first and the second reactions. From comparative analysis of the spectral characteristics of the intermediates, it can be concluded that Cycle I in the scheme by Schoefs and Franck reflects the same process as the side reaction (reaction 6 in the scheme in Fig. 1 in our paper). Cycle II corresponds to the direct sequence of reactions (reactions 1-4, Fig. 1).Fig. 2. Pathways of chlorophyll biosynthesis and regeneration of the photoactive complex of protochlorophyllide [17]. The figures indicate the positions of the maxima of fluorescence spectra (first figure) and absorption spectra (second figure) of pigment forms; I-/695 is a non-fluorescent intermediate.
Generalizing the results obtained by different investigators, Schoefs and Franck came to the conclusion that Cycle I is manifested in the case when the concentration of non-photoactive chlorophyllide is rather high relative to that of the formed chlorophyllide. According to Schoefs and Franck, in this case chlorophyllide rapidly leaves the enzyme, being replaced by protochlorophyllide in the aggregate of the POR complex. This pathway is observed in the membranes of etioplasts, in the embryo leaves, in the isolated PLB, under the low light intensity. Cycle II corresponds to the complete photoconversion of the protochlorophyllide pool (the ratio of non-photoactive protochlorophyllide to the formed chlorophyllide is rather low). The authors suppose that, in this case, chlorophyllide remains bound to the enzyme for a longer time, and during this time disruption of the enzyme aggregate occurs (at the stage of the short-wavelength “Shibata shift”).
BIOSYNTHESIS OF CHLOROPHYLL IN GREEN PLANT LEAVES
All the above-described results of the studies of biosynthesis of chlorophyll native forms were obtained in the investigations of the process in greening etiolated leaves. Naturally, a question arose about the sameness or difference of the pathways and mechanisms of chlorophyll photobiosynthesis in the greening leaves and on Chl accumulation and renewal of its pool in mature green plant leaves, when the main pigment mass is synthesized. A high rate of Chl biosynthesis must simultaneously provide not only accumulation of the pigment in growing leaves but also compensate for its loss in destruction processes, in particular, in photodestruction which is especially intensive in bright light. That is why the problem of chlorophyll biosynthesis mechanisms directly in normal green leaves has long attracted attention of researchers. However, here great difficulties appear, because the accumulation and transformation of the minute amounts of the chlorophyll precursor we have to study against the background of strongly absorbing and fluorescing chlorophyll already accumulated in the green leaves (Chl concentration is several orders of magnitude higher than stationary concentration of the precursor).
Studies of Mechanisms of Chlorophyll Photobiosynthesis in Green Leaves after Placing Plants in Darkness
Using low-temperature (77 K) fluorescence spectrometry that decreases the overlapping of the spectral bands belonging to chlorophyll and protochlorophyllide, we acquired the possibility to overcome these difficulties. The first study of the spectral forms of protochlorophyll(ide) in green leaves and their photochemical activity was the paper by Litvin and coworkers [62]. The changes in the low-temperature spectra of green leaves of beans and of the pigment extracts after placing them in darkness and subsequent illumination were studied. They found out that when placed in the dark, green bean leaves accumulated chlorophyll precursor spectroscopically identical to the main form of protochlorophyllide in etiolated leaves (fluorescence maximum at 655 nm). This band disappeared rapidly on subsequent illumination of the leaves. The quantitative measurements of concentration of the precursor accumulated in the dark, by the spectra of the extracts (using an inner standard) and the comparison to the data of the rate of chlorophyll renewal obtained with the isotope method [63] showed that the process of Chl renewal in the green leaves occurred through the same main form of the precursor as in the in etiolated leaves. Accumulation of the photochemically active protochlorophyllide with a fluorescence maximum at 655 nm in the darkened green leaves was later confirmed in a number of studies [64-67].
The studies of Chl biosynthesis in green leaves in our laboratory were continued only in 2002. To elucidate the participation of different protochlorophyllide forms in chlorophyll a biosynthesis in green leaves, we studied low-temperature fluorescence spectra and fluorescence excitation spectra of green leaves and of their extracts obtained from several plant species, with darkening and subsequent illumination [68]. It was found that in 16 h of darkening three forms of protochlorophyllide were accumulated with the same spectral parameters as the forms in the etiolated leaves (fluorescence maxima at 633, 642, and 655 nm). The main photoactive form was PChld655/650. The pigment with the band at 642 nm disappeared very slowly. In green leaves, like in juvenile etiolated seedlings, the biosynthesis of Chl a involved the precursor form PChld653/648, which in turn is a product of photochemical reaction of the minor long-wavelength form of protochlorophyllide with an absorption maximum at about 680 nm. Consequently, in green leaves, like in etiolated leaves, two pathways of biosynthesis are present:

Studies of Mechanisms of Chlorophyll Photobiosynthesis in Green Leaves Grown in Natural Photoperiodic Conditions
For understanding the whole picture of chlorophyll biosynthesis in plant leaves, it was important to study the process in mature green leaves grown under natural photoperiodic conditions. It appeared that under periodic illumination the apparatuses of Chl biosynthesis are different in young (up to seven days) leaves from mature ones. During the dark period in the young leaves PLB are formed, while they are not in more mature leaves [57].
Schoefs and coworkers [67] studied Chl biosynthesis in the mature (20 days) secondary bean leaves grown under normal periodic illumination (light, 16 h; darkness, 8 h). For initiation of photoconversion of the chlorophyll precursor, the leaves were illuminated by powerful flashes of light at different time of day and night. Difference fluorescence spectra were measured and analyzed: light (flash) minus darkness (before the flash). It was shown that during the light period (illumination of the leaves was performed in the middle of the 16 h light period) under the action of the flash protochlorophyllide with fluorescence maximum at 653 nm underwent transformation. Identical results were obtained for barley and maize. The photoactive form of protochlorophyllide was also found in the greening barley leaves after 2-3 h of illumination when PLB were destroyed and thylakoids developed [70]. The authors supposed that the active form PChld653/648 in green leaves is connected with thylakoids.
In studies of Chl biosynthesis in young green bean leaves (more than 1 day) under conditions of periodic illumination (light, 16 h; darkness, 8 h), in the fluorescence spectra two protochlorophyllide bands were observed: at 632-636 and 653-654 nm, along with Chl maxima at 685, 695, and 735 nm [57]. At the end of the first light period, the shorter-wavelength band prevailed. Using an original technique of studies of the kinetic changes of the absorbance of the leaves in the far red region (at 700 nm) the authors found that in young bean leaves the transformations of the active protochlorophyllide form (fluorescence at 654 nm) under the action of an intense flash of light proceeded through the same intermediate stages as in the greening etiolated leaves. In 6-day-old leaves grown under periodic illumination, as a result of illumination with a powerful flash of light, the primary form of chlorophyllide is formed from the active protochlorophyllide (according to the authors, Chld688/678) which can further transform either into a short-wavelength product Chl675/670 or into a longer-wavelength form Chld694/682 (that mainly corresponds to the scheme proposed by us in Fig. 1). During the first light period (1-day-old leaves), the short-wavelength form is formed predominantly (like in embryo leaves). With the increase in the number of light–dark cycles after the illumination with the flash, the process goes by the second pathway: the long-wavelength form is formed (fluorescence maximum at 694 nm) which, like in the greening etiolated leaves, undergoes the transformation characterized by a short-wavelength spectral shift (Shibata shift). Under illumination of leaves with a flash during the light period, this shift is completed during 1 min, i.e. by an order of magnitude faster than in the greening leaves. At the same high rate etherification of chlorophyllide and regeneration of the active form of protochlorophyllide proceed. Under illumination of the leaves during the dark period (when PLB are formed), the rate of the short-wavelength shift decreases.
Thus, it can be concluded that in green plant leaves grown under the normal photoperiodic conditions the light-dependent synthesis of chlorophyll involves the active long-wavelength form of PChld654/648, close in spectral characteristics to its main active form in greening etiolated leaves. Like in the greening leaves, in the green leaves there are no fewer than two pathways of chlorophyll biosynthesis as dependent on the state of the biosynthetic apparatus and its structure (the formation of PLB and thylakoids) that in turn are dependent on the age of the leaves and are different during the light and dark periods.
SEQUENCE OF REACTIONS IN PHOTOREDUCTION OF PROTOCHLOROPHYLLIDE IN
ARTIFICIAL TRIPLE COMPLEXES
NADPH–PROTOCHLOROPHYLLIDE–POR
Recently, an increasing number of researchers use for studies of the mechanism of photoreduction reaction artificial triple complexes including protochlorophyllide, NADPH, and the POR photoenzyme, serving as a model of photoactive complexes in the living systems. However, the spectral properties of the reconstructed triple complexes markedly differ from their properties in etiolated leaves. In the artificial systems, the fluorescence and absorption maxima of protochlorophyllide are shifted towards shorter wavelengths as compared to the main active form PChld655/650 of whole etiolated leaves.
In a number of papers, active triple complexes with an absorption band near 630 nm were studied [71-74]. Under the action of light the short-wavelength protochlorophyllide transformed into chlorophyllide with an absorption band near 670 nm, shifted towards shorter wavelengths with respect to the corresponding band of the etiolated leaves of plants. Some authors supposed that in vitro POR catalyzed the reaction in a monomeric complex [72, 74]. However, it was shown using chromatography that the molecular mass of the purified POR in solution corresponds to the dimer of the enzyme [73].
In the studies of photochemical activity of the reconstructed triple complexes characterized by fluorescence band at 633 nm, using “light minus dark” difference spectra, in addition to the photoconversion of short-wavelength protochlorophyllide, a slight conversion of longer-wavelength form with a fluorescence maximum at 645 nm was found [75]. In spectral characteristics and the ability for photoreduction at low temperature, this form of protochlorophyllide appeared to be similar to one of the active forms of protochlorophyllide in vivo—PChld645/639.
In our cooperative study with Griffiths and Timofeev [76] using a high concentration of protochlorophyllide, artificial triple complexes were obtained (PChld–POR–NADPH) characterized by a longer-wavelength absorption band (at 648 nm). In the fluorescence spectrum, maxima at 651 and 708 nm were observed. The position of the first is close to that of the maximum of one of the active forms of protochlorophyllide in vivo (653 nm). The fluorescence band at 708 nm apparently consists of two components—a vibratory satellite corresponding to the fluorescence band at 651 nm and a fluorescence maximum belonging to large protochlorophyllide aggregates (their number is not great, which is indicated by the absence of the appropriate band in the absorption spectrum). The high intensity of the long-wavelength band can be explained by effective energy migration from PChld651/648 to the aggregated form. As a result of illumination with intense white light at 77 K, the quenching of both fluorescence bands was observed (without appearance of new bands). This can be accounted for by the formation of non-fluorescent intermediate accompanied by the appearance of an EPR singlet signal with g-factor of a free electron in the spectrum. The efficient quenching of the band at 708 nm is apparently explained by a decrease in the energy migration after photoconversion of PChld651/648 into the non-fluorescent intermediate. With an increase in the temperature the non-fluorescent intermediate turned into chlorophyllide with fluorescence maximum at 695 nm, which corresponds to the position of the maximum of the primary fluorescent intermediate formed in vivo. The formation of chlorophyllide was identified by the spectra of methanol extracts obtained from illuminated samples.
In the works of Heyes and coworkers [77-79], artificial triple complexes were obtained with two active protochlorophyllide forms characterized by absorption bands of approximately equal intensity at 630 and 642 nm and corresponding fluorescence bands at 631 and 644 nm. The longer-wavelength form PChld644/642 was active at very low temperatures: at 180 K it was observed to photoconvert (under the action of a flash of light) into a primary non-fluorescent intermediate with an absorption band near 696 nm [78, 79]. On increasing the temperature (to about 200 K), the non-fluorescent intermediate turned into an intermediary product with an absorption band at 681 nm and a fluorescence band at 684 nm. It should be noted that both the position of the maximum of the non-fluorescent intermediate and the temperature dependence of its further dark transformation coincide with those detected in vivo [80-89]. The temperature 200 K is a critical temperature at which dynamic changes in the enzyme begin, called “glass transition”. This change in the enzyme is apparently connected with the dark transformation of the non-fluorescent intermediate into the primary product. With further increase in temperature above 200 K, in the triple complex, the second dark reaction took place, characterized by short-wavelength shift of the absorption and fluorescence bands to 671 and 674 nm, respectively. By the spectral characteristics of the intermediates and the final product, the chain of protochlorophyllide transformation reactions in the triple complex is analogous to the sequence of Chl biosynthesis reactions in vivo, proceeding at low light intensities and leading to the formation of the short-wavelength form of chlorophyll (“side branch”: reactions 1 and 6 in scheme on Fig. 1).
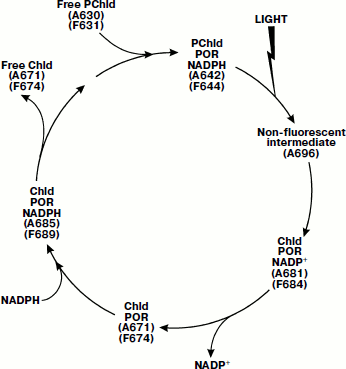
During low-temperature spectroscopic studies of triple complexes including the thermophilic protochlorophyllide-oxidoreductase [90], several dark reactions were detected on the pathway of PChld644/642 transformation into chlorophyllide. Increasing the temperature of the sample after the primary photoreaction of protochlorophyllide at 185 K revealed four dark stages (see scheme in Fig. 3). Comparison of the spectral characteristics of the intermediates to those of the complexes of Chld–POR–NADPH (or NADP+) allowed the authors to interpret the formation of the intermediates. A hypothetical scheme was proposed including proper photoreduction of protochlorophyllide and four dark stages connected with NADPH oxidation, NADP+ release from the complex, the subsequent incorporation of NADPH into the complex, and finally, with the release of free chlorophyllide and incorporation of a new protochlorophyllide (see scheme in Fig. 3).
For studies of the kinetics of the intermediate reactions of the cycle, laser pulses (6 nsec, 450 nm) were used to initiate the photoreduction of protochlorophyllide [91]. The release of NADPH from the complex appeared to be a two-phase process; for this process, the rate constants of dark reactions were calculated. Heyes and coworkers showed the formation of the triple complex of NADPH, protochlorophyllide, and POR included six stages of conformational changes of the enzyme molecule [92].Fig. 3. Scheme of the catalytic cycle of protochlorophyllide-oxidoreductase [90]. The figures indicate the positions of the maxima in the absorption spectra (A) and fluorescence spectra (F) of pigment–protein complexes.
CONCLUSION
Here it seems reasonable to describe briefly the possible near and distant perspectives of those aspects of the mechanism of the terminal stage of biosynthesis and its role in the photosynthesis apparatus formation that were dealt with in the review.
First of all this is elucidation of the mechanisms of individual dark and photochemical reactions and decoding of real structures of the pigment–protein complexes involved in these reactions with known spectral characteristics. The complexity of the problem, as we have already mentioned, is due to the lability of the pigment–protein structures. However, one can hope that new preparative methods and other approaches will develop, in particular, physical methods whose opportunities have been demonstrated in the latest studies of model triple systems.
In the scheme (Fig. 1), only final results of different pathways of biosynthesis of chlorophyll and pheophytin are indicated; they point to the significance of the existence of such a complicated, branching reaction chain. One can hope that in the near future “the missing link” will be found between the system of biosynthesis of pigment–protein complexes and the system of self-building of the functioning of the photosynthetic apparatus.
A great challenge is also the question of the process of formation of the pigment structures preceding the terminal photo-dependent stage of chlorophyll biosynthesis, i.e. of pigment–protein complexes of protochlorophyllide. Here an important role can be played by increasing the sensitivity of fluorescence methods allowing the study of their formation and functioning in living cell at all stages of plant development, beginning with a seed, under physiological conditions.
Undoubtedly, the problem of photobiosynthesis of chlorophyllous pigments in completely developed leaves requires further study. A small number of the existing papers concerning these problems is only the beginning of the studies of the process, providing a global role of photosynthesis in the biosphere and in agriculture.
Finally, the direction of studies only slightly touched on in the review is regulation of pigment synthesis and biogenesis of photosynthetic structures by its coupling with other cellular processes. Besides, in the fundamental and applied aspects it is useful to employ already accumulated information on the rate and the pathways of different branches of the biosynthetic process as dependent on exterior conditions (temperature, illumination, the stage of plant development, etc.) in order to control the terminal stage of chlorophyll biosynthesis and building of the photosynthetic apparatus.
REFERENCES
1.Litvin, F. F., Belyaeva, O. B., and Ignatov, N. V.
(1998) Biol. Membr. (Moscow), 15, 490-503.
2.Litvin, F. F., Belyaeva, O. B., and Ignatov, N. V.
(2000) Uspekhi Biol. Khim., 40, 3-42.
3.Belyaeva, O. B. (1996) Molecular Biophysics
[in Russian], Nauka, Moscow, pp. 156-158.
4.Litvin, F. F., and Belyaeva, O. B. (1968)
Biokhimiya, 33, 928-936.
5.Litvin, F. F., and Belyaeva, O B. (1971)
Biokhimiya, 36, 615-622.
6.Litvin, F. F., and Belyaeva, O. B. (1971)
Photosynthetica, 5, 200-209.
7.Mathis, P., and Sauer, K. (1972) Biochim.
Biophys. Acta, 267, 498-511.
8.Mathis, P., and Sauer, K. (1973) Plant
Physiol., 51, 115-119.
9.Litvin, F. F., Efimtsev, E. I., Ignatov, N. V., and
Belyaeva, O. B. (1976) Fiziol. Rast., 23, 17-24.
10.Litvin, F., Ignatov, N., Efimtsev, E., and
Belyaeva, O. (1978) Photosynthetica, 12, 375-381.
11.Oliver, R. P., and Griffiths, W. T. (1928)
Plant Physiol., 70, 1019-1025.
12.Sironval, C., Kuyper, Y., Michel, J. M., and
Brouers, M. (1967) Stud. Biophys., 5, 43-50.
13.Henningsen, K. W., and Thorne, S. W. (1974)
Physiol. Plant., 30, 82-89.
14.El Hamouri, B., and Sironval, C. (1980)
Photochem. Photobiophys., 1, 219-223.
15.El Hamouri, B., and Sironval, C. (1981) Plant
Sci. Lett., 21, 375-379.
16.Franck, F., Bereza, B., and Boddi, B. (1999)
Photosynth. Res., 59, 56-61.
17.Schoefs, B., and Franck, F. (2008) Photosynth.
Res., 96, 15-26.
18.Ignatov, N. V., and Litvin, F. F. (1981)
Biofizika, 26, 664-668.
19.Shibata, K. (1956) Carn. Inst. Wash. YB,
55, 248-250.
20.Shibata, K. (1957) J. Biochem. (Tokyo),
44, 147-173.
21.Monteverde, N. A., and Ljubimenko, V. N. (1911)
Izv. Imp. Akad. Nauk (St. Petersburg), Ser. VI, 5,
73-101.
22.Krasnovsky, A. A., Bystrova, M. I., and Sorokina,
A. V. (1961) Dokl. AN SSSR, 136, 1227-1230.
23.Vorobyeva, L. M., Bystrova, M. I., and
Krasnovsky, A. A. (1963) Biokhimiya, 28, 524-534.
24.Boardman, N. K. (1966) in The Chlorophylls
(Vernon, L. P., and Seely, G. R., eds.) Academic Press, NY, pp.
437-479.
25.Schopfer, P., and Siegelman, H. B. (1968)
Plant Physiol., 43, 990-996.
26.Lang, F., Vorobyeva, L. M., and Krasnovsky, A. A.
(1971) Mol. Biol. (Moscow), 5, 366-374.
27.Nielsen, O. F., and Kahn, A. (1973) Biochim.
Biophys. Acta, 292, 117-129.
28.Henningsen, K. W., Thorne, S. W., and Boardman,
N. K. (1974) Plant Physiol., 53, 419-425.
29.Butler, W. L. (1961) Arch. Biochem.
Biophys., 92, 287-295.
30.Thorne, S. W. (1971) Biochim. Biophys.
Acta, 226, 113-127.
31.Thorne, S. W. (1971) Biochim. Biophys.
Acta, 226, 128-134.
32.Henningsen, K. W., Kahn, A., and Houssier, C.
(1973) FEBS Lett., 37, 103-109.
33.Schultz, A., and Sauer, K. (1972) Biochim.
Biophys. Acta, 267, 320-340.
34.Raskin, V. I. (1981) Photoreduction of
Protochlorophyllide [in Russian], Nauka i Tekhnika, Minsk.
35.Wiktorsson, B., Ryberg, M., Gough, S., and
Sundqvist, C. (1992) Physiol. Plant., 85, 659-669.
36.Belyaeva, O. B., Karneeva, N. V., Stadnichuk, I.
N., and Litvin, F. F. (1975) Biokhimiya, 40, 951-961.
37.Fradkin, L. I. (1988) in Biogenesis of Pigment
Apparatus of Photosynthesis (Litvin, F. F., ed.) [in Russian],
Nauka i Tekhnika, Minsk, pp. 164-191.
38.Fradkin, L. I., and Shlyk, A. A. (1978) Zh.
Prikl. Spektrosk., 29, 1029-1039.
39.Domanski, V. P., and Rudiger, W. (2001)
Photosynth. Res., 68, 131-139.
40.Domanski, V., Rassadina, V., Gus-Mayer, S.,
Wanner, G., Shoch, S., and Rudiger, W. (2003) Planta,
216, 475-483.
41.Ignatov, N. V., and Litvin, F. F. (1993)
Biochemistry (Moscow), 58, 1074-1083.
42.Ignatov, N. V., and Livin, F. F. (1993)
Biochemistry (Moscow), 58, 1210-1219.
43.Ignatov, N. V., and Litvin, F. F. (1994)
Photosynth. Res., 42, 27-35.
44.Ignatov, N. V., and Litvin, F. F. (1995)
Photosynth. Res., 46, 445-453.
45.Ignatov, N. V., Gostimsky, S. A., Satina, L. Ya.,
and Litvin, F. F. (1998) Biochemistry (Moscow), 63,
1319-1327.
46.Ignatov, N. V., Satina, L. Ya., and Litvin, F. F.
(1999) Photosynth. Res., 62, 185-195.
47.Ignatov, N. V., and Litvin, F. F. (2002)
Biochemistry (Moscow), 67, 949-955.
48.Ignatov, N. V., and Litvin, F. F. (2002)
Photosynth. Res., 71, 195-207.
49.Ignatov, N. V., and Litvin, F. F. (1995)
Biochemistry (Moscow), 60, 1429-1438.
50.Ignatov, N. V., and Litvin, F. F. (1996)
Photosynth. Res., 50, 271-283.
51.Lebedev, N. N., Djelepova, I. D., and Krasnovsky,
A. A. (1991) Biofizika, 36, 1022-1030.
52.Wang, W. J. (1979) Plant Physiol.,
63, 1102-1106.
53.Karapetyan, N. V., Rakhimberdieva, M. G., Bukhov,
N. G., and Gyurjan, I. (1980) Photosynthetica, 14,
48-54.
54.Boichenko, V. A., and Litvin, F. F. (1990)
Biokhimiya, 55, 1309-1318.
55.Griffiths, W. T. (1980) Biochem. J.,
186, 267-278.
56.Schoefs, B., and Franck, F. (1993) J. Exp.
Bot., 44, 1053-1057.
57.Schoefs, B., and Franck, F. (2008) Photosynth.
Res., 96, 15-26.
58.Dubrovsky, V. T., and Litvin, F. F. (2008)
Biol. Membr. (Moscow), 25, 203-209.
59.Klein, S., and Schiff, J. A. (1972) Plant
Physiol., 49, 619-626.
60.Deng, X.-W., and Gruissem, W. (1987) Cell,
49, 379-384.
61.Khandakar, K., and Bradbeer, J. W. (1989)
Cytologia, 54, 409-417.
62.Litvin, F. F., Krasnovsky, A. A., and Rikhireva,
G. T. (1959) Dokl. AN SSSR, 127, 699-701.
63.Shlyk, A. A. (1965) Metabolism of Chlorophyll
in Green Plants [in Russian], Nauka i Tekhnika, Minsk.
64.Garab, G. T., Sundquist, C., and Faludi-Daniel,
A. (1980) Photochem. Photobiol., 31, 491-503.
65.Lebedev, N. N., Siffel, P., and Krasnovsky, A. A.
(1985) Biofizika, 30, 44-49.
66.Lebedev, N. N., Siffel, P., and Krasnovsky, A. A.
(1985) Photosynthetica, 19, 183-187.
67.Schoefs, B., Bertrand, M., and Franck, F. (2000)
Photochem. Photobiol., 72, 85-93.
68.Ignatov, N. V., and Litvin, F. F. (2002)
Biochemistry (Moscow), 67, 949-955.
69.Ignatov, N. V., and Litvin, F. F. (2002) Biol.
Membr. (Moscow), 19, 153-159.
70.Franck, F., and Strazlka, K. (1992) FEBS
Lett., 309, 73-77.
71.Knaust, R., Seyfried, B., Schmidt, L., Schulz,
R., and Senger, H. (1993) J. Photochem. Photobiol. B: Biol.,
20, 161-166.
72.Birve, S., Selstam, E., and Johansson, L. (1996)
Biochem. J., 317, 549-555.
73.Martin, G. E. M., Timko, M. P., and Wilks, H. M.
(1997) Biochem. J., 325, 139-145.
74.Klement, H., Helfrich, M., Oster, U., Schoch, S.,
and Rudiger, W. (1999) Eur. J. Biochem., 265,
862-874.
75.Lebedev, N., and Timko, M. (1999) Proc. Natl.
Acad. Sci. USA, 96, 17954-17959.
76.Belyaeva, O. B., Griffiths, W. T., Kovalev, Yu.
V., Tmofeev, K. N., and Litvin, F. F. (2001) Biochemistry
(Moscow), 66, 173-177.
77.Heyes, D. J., Ruban, A. V., Wilks, H. M., and
Hunter, C. N. (2002) PNAS, 99, 11145-11150.
78.Heyes, D. J., Ruban, A. V., and Hunter, C. N.
(2003) Biochemistry, 42, 523-528.
79.Heyes, D. J., Heathcote, P., Rigby, S. E. J.,
Palacios, M. A., and Grondelle, R., and Hunter, C. N. (2006) J.
Biol. Chem., 281, 26847-26853.
80.Raskin, V. I. (1976) Vesti AN BSSR,
Ser. Biol. Nauk, 5, 43-46.
81.Losev, A. P., and Lyal’kova, N. D. (1979)
Mol. Biol. (Moscow), 13, 837-844.
82.Dujardin, E., and Correia, M. (1979)
Photobiochem. Photobiophys., 1, 25-32.
83.Dujardin, E., Correia, M., and Sironval, C.
(1981) in Proc. 5th Congr. on Photosynthesis: Chloroplast
Development (Akoyunoglou, G., ed.) Philadelphia, 1981, Vol. 5, pp.
21-29.
84.Franck, F., and Mathis, P. (1980) Photochem.
Photobiol., 32, 799-803.
85.Inoue, Y., Kobayashi, T., Ogawa, T., and Shibata,
K. (1981) Plant. Cell Physiol., 22, 197-204.
86.Belyaeva, O. B., and Litvin, F. F. (1980)
Biofizika, 25, 617-623.
87.Belyaeva, O. B., and Litvin, F. F. (1981)
Photosynthetica, 15, 210-215.
88.Belyaeva, O. B., Personova, E. R., and Litvin, F.
F. (1983) Photosynth. Res., 4, 81-85.
89.Ignatov, N., Belyaeva, O., and Litvin, F. (1993)
Photosynth. Res., 38, 117-124.
90.Heyes, D. J., and Hunter, C. N. (2004)
Biochemistry, 43, 8265-8271.
91.Heyes, D. J., and Hunter, C. N. (2007) J.
Biol. Chem., 282, 32015-32020.
92.Heyes, D. J., Menon, B. R. K., Sakuma, M., and
Scrutton, N. (2008) Biochemistry, 47, 10991-10998.