New-Generation Skulachev Ions Exhibiting Nephroprotective and Neuroprotective Properties
E. Y. Plotnikov1,2#, D. N. Silachev1,2#, A. A. Chupyrkina1,2, M. I. Danshina2, S. S. Jankauskas2, M. A. Morosanova2,3, E. V. Stelmashook2,4, A. K. Vasileva1,2, E. S. Goryacheva2,3, Y. A. Pirogov5, N. K. Isaev1,2,4*, and D. B. Zorov1,2*
1Belozersky Institute of Physico-Chemical Biology, Lomonosov Moscow State University, 119992 Moscow, Russia; fax: (495) 939-3181; E-mail: isaev@genebee.msu.su; zorov@genebee.msu.su2Mitoengineering Research Institute, Lomonosov Moscow State University, 119992 Moscow, Russia
3Faculty of Bioengineering and Bioinformatics, Lomonosov Moscow State University, 119991 Moscow, Russia
4Department of Brain Research, Neurology Research Center, Russian Academy of Medical Sciences, Pereulok Obukha 5, 105064 Moscow, Russia
5Training and Research Interfaculty Center of Magnetic Tomography and Spectroscopy, Lomonosov Moscow State University, 119991 Moscow, Russia
# These authors contributed equally.
* To whom correspondence should be addressed.
Received November 5, 2009
A mitochondria-targeted chimeric compound consisting of a rhodamine derivative linked to a plastoquinone molecule (10-(6′-plastoquinonyl)decylrhodamine, SkQR1) was studied under conditions of acute brain or kidney damage. A protective effect of this compound was demonstrated in a model of focal brain ischemia, rat kidney ischemia/reperfusion, myoglobinuria (rhabdomyolysis, or crush syndrome), and pyelonephritis. We found that a single intraperitoneal injection of SkQR1 diminishes the size of the ischemic zone in the brain and improves performance of a test characterizing neurological deficit in ischemic animals. Control substance not containing plastoquinone appeared to be not neuroprotective. The data show that SkQR1 is a nephroprotectant and neuroprotectant, which can be due to the antioxidative action of this Skulachev cation.
KEY WORDS: Skulachev ions, SkQ, SkQR1, kidney, oxidative stress, mitochondria-targeted antioxidants, mitochondria, rhabdomyolysis, pyelonephritis, brain ischemia, magnetic resonance tomographyDOI: 10.1134/S0006297910020045
Abbreviations: AKI, acute kidney injury; DCF, 2,7-dichlorodihydrofluorescein diacetate; MRI, magnetic resonance imaging; ROS, reactive oxygen species; SkQ, cationic derivatives of plastoquinone; SkQR1, 10-(6′-plastoquinonyl)decylrhodamine 19; TTC, 2,3,5-triphenyl tetrazolium chloride.
Skulachev ions, membrane-penetrating cations previously used as tools to
investigate the now well-established chemiosmotic hypothesis [1], currently have a second life as vehicles for
addressed mitochondrial transport of different substances for
correction of certain undesirable metabolic changes [2-7]. Chimeric molecules formed
from classical Skulachev ions conjugated via a linker with various
physiologically active components, in particular with antioxidants [8], have been designed to prevent sudden changes in
mitochondrial redox status that underlie many pathologies including
aging.
A collection of such chimeric molecules has been synthesized at the Mitoengineering Research Institute at Lomonosov Moscow State University. Plastoquinone was chosen as a redox-active group, and later it was found that this gave the mitochondria-penetrating molecule a number of beneficial effects, first of all defending the system from unwanted consequences of oxidative stress. The latest data suggest that these new-generation Skulachev cations can prevent or correct pathological changes in different tissues (e.g. heart, kidney, brain, and eye) that are characterized by increased level of reactive oxygen species (ROS) [2-8]. We recently presented the first progress in the study of nephroprotective action of substances of the SkQ (cationic derivatives of plastoquinone) family using a model of kidney ischemia [5, 7, 9]. We also showed the neuroprotective effect of Skulachev ions in a model of compression brain ischemia [6, 8]. However, this model is not broadly accepted, so more complete models that correspond to the most frequent clinical forms of ischemic brain damage are needed. We have reasons to assume that SkQs have a wider range of nephroprotective and neuroprotective properties in kidney and brain pathology caused by oxidative stress, e.g. during rhabdomyolysis, pyelonephritis, and temporary occlusion of brain arteries. These studies are timely because kidney diseases, despite several decades of study of the mechanisms of the development of acute kidney injury (AKI) in various clinical pathologies, modern therapeutic means still fail to effectively cure such pathologies. For the majority of kidney dysfunctions, the only effective solution is periodic hemodialysis and, very frequently, the transplantation of a healthy kidney, which, in addition to high cost and high degree of risk does not always solve the problem. The same is true for brain, because acute disturbances of brain blood circulation remains one of the most important medico-social problems due to their high impact on the morbidity and mortality of the population, significant loss of working time, and primary invalidity. Mortality from brain vascular diseases in Russia takes second place, just behind cardiovascular diseases [10]. Considering therapy of kidney and brain pathologies involving oxidative stress, substances of the SkQ family meet all requirements because they protect in a damaged tissue one of the most sensitive intracellular ROS targets, i.e. mitochondria.
MATERIALS AND METHODS
This study used male white rats of weight 300-350 g. They were fed ad libitum.
Model of kidney ischemia. The animals were subjected to 40-min ischemia of the left kidney as described in [11]. The right side nephrectomy was executed together with ischemia. On the second day after ischemia blood samples were taken to determine creatinine concentration using a CellTac blood analyzer (Nihon Kohden Corp., Japan).
Animal model of rhabdomyolysis. Rhabdomyolysis was induced by a standard method by injection of 50% water solution of glycerol (ICN, USA) into the leg muscles of rats as described earlier [12]. Blood samples were taken on the second day after the injection. Blood creatinine concentration was determined using the CellTac blood analyzer.
The ischemia and rhabdomyolysis modeling experiments were done with at least eight animals in each group. Data is presented as mean ± standard error.
In vitro pyelonephritis model. The primary cell culture of kidney tubules was obtained as described earlier [11]. Cells were cultivated in an incubator with a humid atmosphere and 5% CO2 concentration in DMEM/F-12 medium (Invitrogen, USA) containing 10% of fetal bovine serum (PAA, USA) for 1-4 days and then used for the experiments.
The fraction of mononuclear cells of peripheral rat blood was obtained. For this purpose, heparinized blood taken from the jugular vein was centrifuged at 2000g in single-stage density gradient of Ficoll-Urografin of 1.077 g/cm3 density (PanEko, Russia). The suspension of mononuclear cells in complete medium was added to the kidney tubule culture. To induce proinflammatory leukocytic response analogous to pyelonephritis, 100 µl of a suspension of inactivated E. coli bacteria (108-109 cells per ml) was added to the cell culture.
After 1 day of co-cultivation, cells were stained with 10 µM 2,7-dichlorofluorescein (DCF) (Invitrogen), and ROS production in kidney cells was analyzed using a scanning confocal microscopy (LSM510; Carl Zeiss Jena, Germany). The mean intensity of DCF fluorescence in cells was calculated for quantitative analysis by confocal images.
Brain ischemia model. Brain ischemia in rats was induced by introduction of a thread covered with silicon into the middle cerebral artery as described in [13, 14]. Blood flow was closed for 60 min, and then the thread was taken out of the vessel and the blood supply in the middle cerebral artery was restored. During and after the surgery animal body temperature was maintained at the level of 37 ± 0.5°C. Sham-operated animals underwent the same procedures except cutting vessels and introducing the thread.
The volume of brain infarct was determined on the first day to study neuroprotective action of the drug or during 7 days to study the dynamics of ischemic damage formation. This evaluation was accomplished by morphometric analysis of digital images obtained using magnetic resonance imaging (MRI) or by staining brain sections with 2,3,5-triphenyl tetrazolium chloride (TTC). (We have shown earlier that these methods give similar results [15].) All MRI experiments were done as described before [15] using a BioSpec 70/30 instrument (Bruker, Germany) with magnetic field induction of 7 T and gradient system of 105 mT/m.
Behavioral test was performed 1 day before the operation and on the first day after the induction of ischemia. The limb-placing test was used to evaluate the neurological deficit estimated by a conventional 14-point scale [16] with modifications [17]. The test had 7 limb-placing tasks that assess the sensorimotor integration of forelimb and hindlimb responses to tactile and proprioceptive stimulation. The task was scored in a following way: the rat performed normally, 2 points; the rat performed with a delay exceeding 2 sec, and/or incompletely, 1 point; and the rat which did not perform normally, 0 points.
Animals were handled and experimental procedures were done according to generally accepted international guidance for animal care.
The results are presented as mean ± standard error. The normality of sign distribution in the selection was estimated using the Shapiro–Wilk W-criteria. To compare data in behavior tests, we used the Mann–Whitney U-criteria (for independent selections). To estimate the statistical mean of the differences of infarct volumes, we used t-criteria at significance level p < 0.05.
RESULTS
The SkQ rhodamine derivative 10-(6′-plastoquinonyl)decylrhodamine 19 (SkQR1) [8] is highly nephroprotective in three models of kidney failure (ischemia, rhabdomyolysis, and pyelonephritis) and strongly neuroprotective in a model of focal brain ischemia.
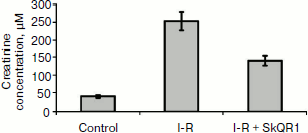
Improvement of kidney function after unilateral ischemia. Hypoxia (ischemia) followed by reoxygenation (reperfusion) (I-R) is one of the most broadly used models of oxidative stress [18]. To study the action of SkQR1 on kidney, we used a model of unilateral 40-min ischemia of the left kidney together with a removal of the right kidney. We used the following scheme of SkQR1 injection for the therapy: preliminary intraperitoneal drug injection 3 h before the ischemia, injection 1 h after the ischemia, and three times each 12 h at the dose 100 nmol/kg per injection. Therefore, the total dose of SkQR1 to the animal was 500 nmol/kg. On the second day after I-R without treatment, we observed the development of AKI, the creatinine level in blood serum increasing up to 250 µM. SkQR1 treatment according to the scheme 500 nmol/kg 48 h after I-R decreased the level of creatinine in blood almost twofold (Fig. 1). These results support the protective action of SkQR1 during 90-min ischemia with preliminary injection 1 day before ischemia, which we showed before; in this case SkQR1 effectively prevented ROS generation in kidney tissue after I-R [8].
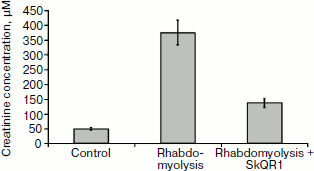
Improvement of kidney function after myoglobinuria. Study of the development of AKI during rhabdomyolysis showed that the development of critical kidney failure is revealed during first 2 days by the increased level of products of nitrogen metabolism in blood, in particular, creatinine, which reaches its peak on the second day (Fig. 2). For the therapy we used a scheme analogous to the treatment of ischemic AKI: intraperitoneal injection of the drug 1 h after the induction of rhabdomyolysis and then three times at 12-h intervals at dose 100 nmol/kg per injection. Therefore, the animal received a total dose of 400 nmol SkQR1 per kg. During SkQR1 treatment according to this scheme, 48 h after glycerin injection we observed a decrease in creatinine level in blood by more than 2.5-fold compared to rhabdomyolysis without treatment (Fig. 2).Fig. 1. Development of acute kidney failure after 40-min I-R of a single kidney. The increase in creatinine concentration in blood on the second day after I-R indicates the development of AKI. Influence of SkQR1 treatment (100 nmol/kg intraperitoneally 3 h before ischemia and then in 1, 12, 24, and 36 h after the initiation of reperfusion) on kidney functioning. For groups I-R and (I-R + SkQR1), p < 0.01.
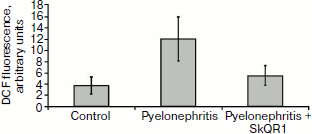
Protective action during pyelonephritis modeling in vitro. We found that during co-cultivation of kidney cells with activated leukocytes, the intensity of DCF fluorescence increased almost threefold compared to control (Fig. 3), which indicates the development of oxidative stress in the cell model of pyelonephritis. Moreover, we found that the strongest change in ROS level takes place in the primary kidney culture and not in the leukocytes. In cells that were preliminarily incubated with 10 nM SkQR1 within 2 h, ROS production (evaluated by DCF fluorescence) decreased in kidney cells almost to control values. This fact supports the idea that the protective action is mostly targeted to kidney cells and to the decrease in ROS generation through mitochondria-dependent protective signal pathways. Therefore, according to the confocal microscopy data, we conclude that mitochondrial antioxidant SkQR1 protects kidney cells from oxidative stress under conditions modeling inflammation in kidney during pyelonephritis.Fig. 2. Development of AKI (increase in creatinine level in blood) on the second day after rhabdomyolysis modeling. The influence of intraperitoneal SkQR1 introduction (100 nmol/kg at 1, 12, 24, and 36 h after the initiation of rhabdomyolysis) on kidney functioning. For groups Rhabdomyolysis and (Rhabdomyolysis + SkQR1), p < 0.01.
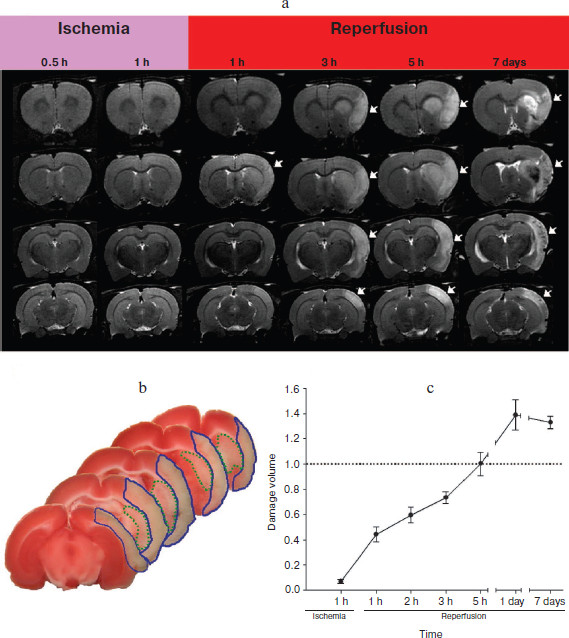
Dynamics of development of ischemic focus in brain. The analysis of T2-weighted images obtained during ischemia by MRI did not reveal significant changes in signal after 30 and 60 min. Remarkable change in signal intensity was observed only more than 1 h after the initiation of reperfusion; moreover, the intensity of the signal further increased and reached its maximum on the first day. On the 7th day T2-weighted images were heterogeneous, there were regions with both increased signal (explained by increased level of liquid in the analyzed object) and decreased signal (Fig. 4a; see color insert). Morphometric analysis of T2-weighted images revealed the dynamics of the ischemic lesion formation (Fig. 4b; see color insert). As seen on the graph, maximum volume of ischemic zone (including damage to cortex and striatum) was observed on the first day, and to the 7th day it was somewhat decreased, which is connected with a decrease in tissue swelling.Fig. 3. Model of pyelonephritis in vitro. Changes in intensity of DCF fluorescence reflecting ROS level in kidney cells after 24 h of co-cultivation with activated leukocytes. The influence of preincubation of kidney cells with 10 nM SkQR1 1 h before the addition of leukocytes and bacteria. For the groups Pyelonephritis and (Pyelonephritis + SkQR1), p < 0.05.
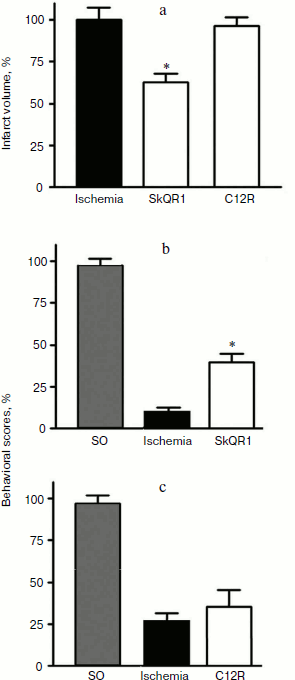
Influence of preliminary SkQR1 treatment on the volume of brain damage and neurological deficit of ischemic rats. SkQR1 in dose 1 µmol/kg was injected into rats intraperitoneally 24 h before the induction of ischemia. One day after the induction of ischemia, the rat was subjected to the limb-placing test, and then brain sections were stained with TTC. Moreover, the evaluation of the ischemic area revealed that SkQR1 treatment led to statistically significant decrease of the infarct volume from (327 ± 25) to (205 ± 17) mm3, which fully correlated with the data of sensorimotor performance of the legs of these animals (Fig. 5). The results obtained in the limb-placing test revealed that intraperitoneal injection of SkQR1 significantly decreased neurological deficit of the ischemic animals. The intact rats before the induction of ischemia scored 14 in this test, sham-operated animals scored 13.5 ± 0.5, after ischemia this value was only 1.4 ± 0.2 for the ischemic group without treatment that received physiological solution instead of SkQR1. A single injection of the drug significantly increased these values to 5.5 ± 0.7 (Fig. 5).Fig. 4. Formation of volume of infarct after transient occlusion of the middle cerebral artery in rats. a) T2-weighted MRI. Temporal changes in magnetic resonance images of representative brain for four different brain sections are shown; arrows indicate area of damage. b) Standard picture of brain cortex damage (outlined with blue line) and underlying striatum (outlined with dotted green line) on initiation of ischemia by the occlusion of a middle cerebral artery with a thread. The data obtained on the first day after ischemia is presented; TTC staining was used to reveal the ischemic focus on sections. c) Changes in total volume of ischemic damage measured on T2-weighted images, including damage to cortex and striatum. Volumes normalized by data obtained 5 h after the reperfusion.
Influence of preliminary C12R treatment on the volume of brain damage and neurological deficit of ischemic rats. C12R drug, which is SkQR1 without the quinone residue, was injected intraperitoneally in dose 1 µmol/kg one day before the induction of ischemia. Morphometric analysis of the images obtained one day after the operation did not reveal significant changes between the volume of infarct of rats who received physiological solution (338 ± 44 mm3) and C12R (288 ± 20 mm3). No decrease in behavioral scores in the limb-placing test one day after the induction of ischemia was observed (Fig. 5).Fig. 5. Influence of Skulachev ions on infarct volume (a) and performance in the limb-placing test (b, c) of rats after transient artery occlusion. Black columns, ischemia; white columns, ischemia + SkQR1 treatment; gray columns, sham-operated (SO). The evaluation of the damage area for SkQR1 was done using TTC staining and for C12R by MRI with analysis of T2-weighted images. For panel (a), * p < 0.05; for panels (b, c), * p < 0.001 (6-11 animals).
DISCUSSION
The goal of this study was to assess protective effect of SkQR1 on organs totally different in structure and function, i.e. kidney and brain. We supposed that of the pathologies of different organs after oxidative stress might be consolidated to create a universal protection strategy. This supposition allowed us to discuss ischemic brain and kidney damage, rhabdomyolysis and pyelonephritis, in one study, as both of these pathologies are characterized by increased level of ROS in the tissue.
For kidney it has been proved that the primary mechanism of cell damage and death in nephrons after oxidative stress, in particular during I-R, involves mitochondrial damage [11] caused by the induction of nonspecific mitochondria permeability, followed by increased ROS generation [19] and cell death, probably through apoptosis [9, 11]. Two other kidney pathologies, myoglobinuria (rhabdomyolysis, or crush syndrome) and pyelonephritis, might also be considered as nephrological diseases with oxidative stress involved. The main participants of this chain of events, leading to mitochondrial dysfunction and oxidative stress, are ROS. It is obvious that in this situation antioxidants can decrease the consequences of oxidative stress, eliminating the excess of free radicals, thus preventing damage to kidney cells and tissue [2, 20]. In general, increasing cell tolerance towards damaging ROS action in any way (e.g. by ischemic preconditioning) is a basic approach to prevent acute kidney failure and is very much in demand in modern medicine, including such fields like surgery, urology, and transplantation.
The same arguments can be used for antioxidant strategy for pathologies caused by brain ischemia. It should be noted that though the mechanisms of ischemic brain damage where ROS play the key role [21] are actively studied, the problem of pharmacological correction of this pathology remains one of the main problems of experimental and clinical neurology and neurosurgery, which is a reason to create an adequate model for the study of ischemia and the development of novel neuroprotectors. The model of unilateral intravascular occlusion of the middle cerebral artery by a Kapron thread that is inserted through a. carotis communis to the place of origin of a. cerebri media from a. carotis interna [13, 22], is the most similar to clinical forms of ischemic brain injury. The performed study of ischemic brain damage evaluated using MRI revealed that the induction of focal ischemia in the middle cerebral artery allows us to obtain a standard by volume and localization of ischemic focus, whose basic volume forms within 24 h of reperfusion. This means that we can evaluate the action of neuroprotectors already at this step of ischemic brain damage. However, it should be admitted that even using this model for testing pharmacological drugs did not make it possible to create highly effective neuroprotectors. In this context, the search for novel pharmacological drugs to treat pathologies related to acute failures of brain blood circulation is an important problem of experimental and clinical neurology.
According to these ideas, antioxidants should be potential nephroprotectors and neuroprotectors in pathologies mediated by oxidative stress. However, until now there were no convincing clinical data about the ability of antioxidants to prevent ischemic damage [23], which is partially explained by the problem of transporting these drugs to the place of ROS generation at the right moment and in sufficient amount to decrease oxidative stress [24]. In this context, the most promising would be a principally new trend of antioxidants development for the therapy of diseases with oxidative stress involved – the development of mitochondria-targeted antioxidants [2-8, 25-27]. It was shown that these drugs improve kidney and heart functioning after ischemia and protect neurons from consequences of a transient compression of part of the brain [6].
We expanded the possible range of nephroprotective properties of penetrating cations of the SkQ family (in particular, SkQR1), showing that not only ischemia-driven kidney pathologies can be prevented (and probably reversed) by Skulachev ions, but also pathological changes in mitochondria in modeling myoglobinuria and pyelonephritis are significantly normalized by Skulachev ions. Apparently, the type of ROS source in all these three cases is not of particular importance (during I-R and rhabdomyolysis mitochondria are the main source of ROS, and in case of pyelonephritis and antigenic stimulation ROS are produced mainly by the leukocytic NADPH-oxidase system).
Using the model of compression brain ischemia, we showed earlier neuroprotective properties of SkQR1 [6]. However, though this model is quite simple in use, it does not fully correspond to the most frequent clinical forms of ischemic brain damage during focal ischemia, and it does not allow evaluation of the effect of the drug on compensation of neurological disturbances caused by ischemia in animals. Considering this, the data on neuroprotective properties of SkQR1 obtained earlier in the model of compression ischemia had to be confirmed in another model, which was actually done in this study. Using the model of focal ischemia we found that a single introduction of SkQR1 before ischemia results in decrease in infarct volume on average by 37%, and also in significant decrease in neurological deficit in ischemic animals. Earlier a positive anti-ischemic effect of another mitochondrial antioxidant, MitoQ, was demonstrated in a heart ischemia model [26]; however, this substance failed to protect brain from ischemic damage [27].
The SkQR1 molecule consists of two parts, “head” responsible for penetration of the molecule into mitochondrion and antioxidant “tail”. Because we cannot exclude that the part of the molecule not carrying quinone residue can also manifest biological activity, we studied possible neuroprotective effect of C12R, a conjugate of rhodamine with a linker chain of 12 carbon atoms. However, we did not observe neuroprotective effect of C12R on the model of focal ischemia.
Our results show that SkQR1 has a significant nephroprotective and neuroprotective action that might be determined by the antioxidant action of the molecule. However, we cannot exclude other mechanisms of SkQR1 action, for example, through a mechanism of the induction of ischemic tolerance that might also cause an increase in cell and tissue tolerance towards oxidative stress.
The authors congratulate V. P. Skulachev with his anniversary and wish long years of productive life in science.
This study was supported by the Research Institute of Mitoengineering and the Russian Foundation for Basic Research (grants 08-04-00762-a, 08-04-01667-a, 09-04-01096-a).
REFERENCES
1.Liberman, E. A., Topali, V. P., Tsofina, L. M.,
Jasaitis, A. A., and Skulachev, V. P. (1969) Nature, 222,
1076-1078.
2.Skulachev, V. P. (2007) Biochemistry
(Moscow), 72, 1385-1396.
3.Cocheme, H. M., Kelso, G. F., James, A. M., Ross,
M. F., Trnka, J., Mahendiran, T., Asin-Cayuela, J., Blaikie, F. H.,
Manas, A. R., Porteous, C. M., Adlam, V. J., Smith, R. A., and Murphy,
M. P. (2007) Mitochondrion, 7, 94-102.
4.Antonenko, Y. N., Avetisyan, A. V., Bakeeva, L. E.,
Chernyak, B. V., Chertkov, V. A., Domnina, L. V., Ivanova, O. Yu.,
Izyumov, D. S., Khailova, L. S., Klishin, S. S., Korshunova, G. A.,
Lyamzaev, K. G., Muntyan, M. S., Nepryakhina, O. K., Pashkovskaya, A.
A., Pletjushkina, O. Yu., Pustovidko, A. V., Roginsky, V. A.,
Rokitskaya, T. I., Ruuge, E. K., Saprunova, V. B., Severina, I. I.,
Simonyan, R. A., Skulachev, I. V., Skulachev, M. V., Sumbatyan, N. V.,
Sviryaeva, I. V., Tashlitsky, V. N., Vassiliev, J. M., Vyssokikh, M.
Yu., Yaguzhinsky, L. S., Zamyatnin, A. A., Jr., and Skulachev, V. P.
(2008) Biochemistry (Moscow), 73, 1273-1287.
5.Anisimov, V. N., Bakeeva, L. E., Egormin, P. A.,
Filenko, O. F., Isakova, E. F., Manskikh, V. N., Mikhelson, V. M.,
Panteleeva, A. A., Pasyukova, E. G., Pilipenko, D. I., Piskunova, T.
S., Popovich, I. G., Roshchina, N. V., Rybina, O. Yu., Saprunova, V.
B., Samoylova, T. A., Semenchenko, A. V., Skulachev, M. V., Spivak, I.
M., Tsybul’ko, E. A., Tyndyk, M. L., Vyssokikh, M. Yu., Yurova,
M. N., Zabezhinsky, M. A., and Skulachev, V. P. (2008) Biochemistry
(Moscow), 73, 1329-1342.
6.Bakeeva, L. E., Barskov, I. V., Egorov, M. V.,
Isaev, N. K., Kapelko, V. I., Kazachenko, A. V., Kirpatovsky, V. I.,
Kozlovsky, S. V., Lakomkin, V. L., Levina, S. B., Pisarenko, O. I.,
Plotnikov, E. Y., Saprunova, V. B., Serebryakova, L. I., Skulachev, M.
V., Stelmashook, E. V., Studneva, I. M., Tskitishvili, O. V.,
Vasilyeva, A. K., Victorov, I. V., Zorov, D. B., and Skulachev, V. P.
(2008) Biochemistry (Moscow), 73, 1288-1299.
7.Neroev, V. V., Archipova, M. M., Bakeeva, L. E.,
Fursova, A. Zh., Grigorian, E. N., Grishanova, A. Yu., Iomdina, E. N.,
Ivashchenko, Zh. N., Katargina, L. A., Khoroshilova-Maslova, I. P.,
Kilina, O. V., Kolosova, N. G., Kopenkin, E. P., Korshunov, S. S.,
Kovaleva, N. A., Novikova, Yu. P., Philippov, P. P., Pilipenko, D. I.,
Robustova, O. V., Saprunova, V. B., Senin, I. I., Skulachev, M. V.,
Sotnikova, L. F., Stefanova, N. A., Tikhomirova, N. K., Tsapenko, I.
B., Shchipanova, A. I., Zinovkin, R. A., and Skulachev, V. P. (2008)
Biochemistry (Moscow), 73, 1317-1328.
8.Skulachev, V. P., Anisimov, V. N., Antonenko, Y.
N., Bakeeva, L. E., Chernyak, B. V., Erichev, V. P., Filenko, O. F.,
Kalinina, N. I., Kapelko, V. I., Kolosova, N. G., Kopnin, B. P.,
Korshunova, G. A., Lichinitser, M. R., Obukhova, L. A., Pasyukova, E.
G., Pisarenko, O. I., Roginsky, V. A., Ruuge, E. K., Senin, I. I.,
Severina, I. I., Skulachev, M. V., Spivak, I. M., Tashlitsky, V. N.,
Tkachuk, V. A., Vyssokikh, M. Y., Yaguzhinsky, L. S., and Zorov, D. B.
(2009) Biochim. Biophys. Acta, 1787, 437-461.
9.Plotnikov, E. Y., Vasileva, A. K., Arkhangelskaya,
A. A., Pevzner, I. B., Skulachev, V. P., and Zorov, D. B. (2008)
FEBS Lett., 582, 3117-3124.
10.Suslina, Z. A., and Piradov, M. A. (2008) in
Stroke: Diagnostics, Treatment, and Prophylaxis [in Russian],
MEDpress-inform, Moscow.
11.Plotnikov, E. Y., Kazachenko, A. V., Vyssokikh,
M. Y., Vasileva, A. K., Tcvirkun, D. V., Isaev, N. K., Kirpatovsky, V.
I., and Zorov, D. B. (2007) Kidney Int., 72,
1493-1502.
12.Plotnikov, E. Y., Chupyrkina, A. A., Pevzner, I.
B., Isaev, N. K., and Zorov, D. B. (2009) Biochim. Biophys.
Acta, 1792, 796-803.
13.Longa, E. Z., Weinstein, P. R., Carlson, S., and
Cummins, R. (1989) Stroke, 20, 84-91.
14.Koizumi, J., Yoshida, Y., Nakazawa, T., and
Ooneda, G. (1986) Jpn. J. Stroke, 8, 1-8.
15.Silachev, D. N., Uchevatkin, A. A., Pirogov, Yu.
A., Zorov, D. B., and Isaev, N. K. (2009) Bull. Exp. Biol. Med.,
147, 232-237.
16.De Ryck, M., van Reempts, J., Borgers, M.,
Wauquier, A., and Janssen, P. A. (1989) Stroke, 20,
1383-1390.
17.Jolkkonen, J., Puurunen, K., Rantakomi, S.,
Harkonen, A., Haapalinna, A., and Sivenius, J. (2000) Eur. J.
Pharmacol., 400, 211-219.
18.Cadenas, E., and Sies, H. (1985) Adv. Enzyme
Regul., 23, 217-237.
19.Zorov, D. B., Filburn, C. R., Klotz, L. O.,
Zweier, J. L., and Sollott, S. J. (2000) J. Exp. Med.,
192, 1001-1014.
20.Koyner, J. L., Sher-Ali, R., and Murray, P. T.
(2008) Nephron. Exp. Nephrol., 109, 109-117.
21.Cao, W., Carney, J. M., Duchon, A., Floyd, R. A.,
and Chevion, M. (1988) Neurosci. Lett., 88, 233-238.
22.Hunter, J., Green, A. R., and Cross, A. J. (1995)
Trends Pharmacol. Sci., 16, 123-128.
23.Kromhout, D. (2001) J. Nutr. Health Aging,
5, 144-149.
24.Becker, L. B. (2004) Cardiovasc. Res.,
61, 461-470.
25.Antonenko, Y. N., Roginsky, V. A., Pashkovskaya,
A. A., Rokitskaya, T. I., Kotova, E. A., Zaspa, A. A., Chernyak, B. V.,
and Skulachev, V. P. (2008) J. Membr. Biol., 222,
141-149.
26.Adlam, V. J., Harrison, J. C., Porteous, C. M.,
James, A. M., Smith, R. A., Murphy, M. P., and Sammut, I. A. (2005)
FASEB J., 19, 1088-1095.
27.Hobbs, C. E., Murphy, M. P., Smith, R. A., and
Oorschot, D. E. (2008) Pediatr. Int., 50, 481-488.