Substitution of Ether Linkage for Ester Bond in Phospholipids Increases Permeability of Bilayer Lipid Membrane for SkQ1-Type Penetrating Cations
T. M. Il’yasova1, T. I. Rokitskaya1, I. I. Severina2, Y. N. Antonenko1*, and V. P. Skulachev1
1Belozersky Institute of Physico-Chemical Biology, Lomonosov Moscow State University, 119991 Moscow, Russia; E-mail: antonen@mail.genebee.msu.ru2Biological Faculty, Lomonosov Moscow State University, 119991 Moscow, Russia
* To whom correspondence should be addressed.
Received June 7, 2012
Using dialkylphospholipid (diphytanyl phosphatidylcholine) instead of the conventional diacylphospholipid (diphytanoyl phosphatidylcholine) in planar lipid bilayer membranes (BLM) led to an increase in the diffusion potential of the penetrating cation plastoquinonyl-decyl-triphenylphosphonium (SkQ1), making it close to the Nernst value, and accelerated translocation of SkQ1 across the BLM as monitored by the kinetics of a decrease in the transmembrane electric current after applying a voltage (current relaxation). The consequences of changing from an ester to an ether linkage between the head groups and the hydrocarbon chains are associated with a substantial reduction in the membrane dipole potential known to originate from dipoles of tightly bound water molecules and carbonyl groups in ester bonds. The difference in the dipole potential between BLM formed of the ester phospholipid and that of the ether phospholipid was estimated to be 100 mV. In the latter case, suppression of SkQ1-mediated proton conductivity of the BLM was also observed.
KEY WORDS: BLM, phospholipid, proton conductivity, dipole potential, SkQ1DOI: 10.1134/S0006297912090118
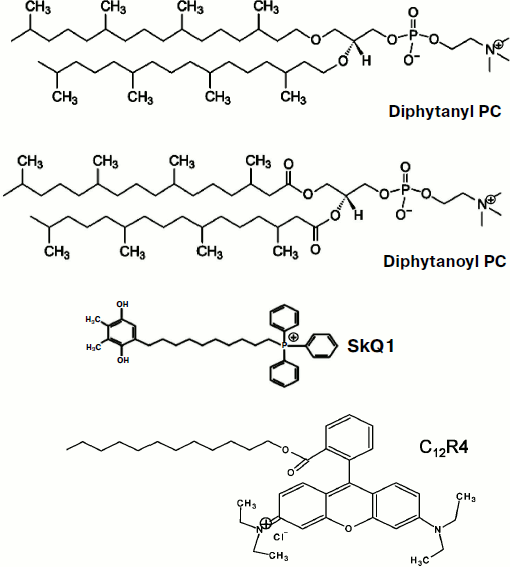
Abbreviations: BLM, planar bilayer phospholipid membrane; C12R4, dodecylrhodamine B cation; diphytanoyl PC, 1,2-diphytanoyl-sn-glycero-3-phosphocholine; diphytanyl PC, 1,2-di-O-phytanyl-sn-glycero-3-phosphocholine; SkQ1, plastoquinonyl triphenylphosphonium cation.
Yefim A. Liberman became famous as an outstanding experimenter due to
his work on planar bilayer phospholipid membranes (BLM) [1-3]. These experiments, launched
in the 1960s, contributed to the proof of the chemiosmotic hypothesis
of oxidative phosphorylation, the author of which, Peter Mitchell, was
later awarded the Nobel Prize. In our experiments we also used BLM as a
test system for studies of penetrating properties of the new class of
antioxidants of SkQ1 type and their analogs synthesized in our
laboratory [4-8]. While
conducting these experiments on the BLM, we encountered certain
difficulty: at low SkQ concentrations the value of SkQ diffusion
potential did not reach the values calculated on the basis of the
Nernst equation (about 60 mV per 10-fold gradient of the cation), and
increasing the concentration of the tested cation led to
destabilization of the BLM due to the detergent effect of this type of
compounds. More stable “colored” (thick) phospholipid
membranes provided higher values of diffusion potentials. However, it
is the “black” BLM, and not the “colored”
membranes, that can serve as an adequate model of biological membranes,
which always have areas of phospholipid bilayer.
In the early work of Liberman and Topaly (1969), it was noted that similar in structure penetrating cations and anions (tetraphenylphosphonium and tetraphenylborate) have completely different permeability through neutral membranes, the permeability of anions being substantially higher [9]. They suggested the formation of a layer of dipoles at the membrane-water interface to be responsible for this phenomenon; these dipoles were assumed to be oriented with their positive charge towards the inside of the membrane. Further research conducted in different laboratories not only confirmed this suggestion, but also made it possible to determine the contribution of various phospholipid components in the formation of this dipole potential [10]. In particular, it was shown that a significant proportion of the dipole potential is formed by carbonyls, which are part of the ester bond of fatty acids and glycerol in phospholipids [11]. Synthetic phospholipids differing only in the type of this bond (esters and ethers) were shown to have dipole potentials with about 120 mV difference, the ether potential value being smaller. In this paper, we present results of studies of permeability of SkQ1 and its analogs for BLM made of phospholipids with residues of phytanic acid (diphytanoyl PC) or residues of phytol (diphytanyl PC; Fig. 1). In accordance with the predictions of the dipole potential theory, the permeability of the studied cations was substantially higher in the case of diphytanyl PC (when compared to diphytanoyl PC).
Fig. 1. Structures of diphytanyl PC, diphytanoyl PC, C12R4, and SkQ1.
MATERIALS AND METHODS
The permeability of planar bilayer phospholipid membranes (BLM) was measured in a Teflon cell with two compartments separated by a Teflon septum with a hole 0.6-mm in diameter, where a membrane was formed from one of the two phospholipids: diphytanoyl PC or diphytanyl PC (both at the concentration of 20 mg/ml in decane). Electrical parameters were measured by two AgCl electrodes immersed in a cell on the two sides of the membrane using a Keithley 6617 amplifier (Keithley, USA) for measuring transmembrane electrical potential difference and the patch-clamp amplifier OES-2 (Opus, Russia) for measuring the current through the membrane.
RESULTS AND DISCUSSION
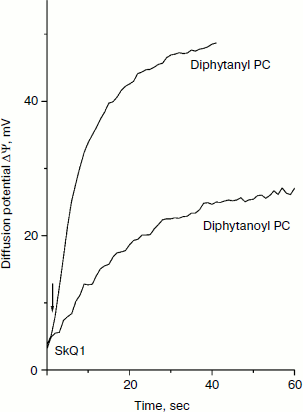
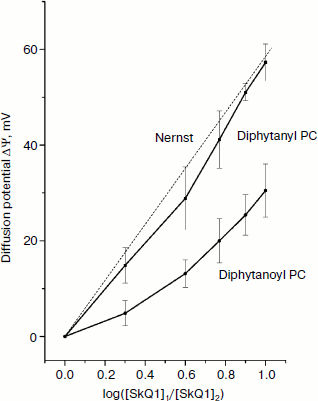
The structures of the compounds used in this study are shown in Fig. 1. Figure 2 shows the kinetics of the generation of SkQ1 diffusion potential on BLM composed of either diphytanoyl PC or diphytanyl PC. The potential was positive in the compartment with the lower SkQ1 concentration. The process was initiated by the addition of 1 μM SkQ1 to one of the compartments that originally contained 0.1 μM SkQ1. The kinetics of the potential generation for diphytanyl PC was faster than that for diphytanoyl PC (see Fig. 2). In addition, the magnitude of the stationary potential was greater for diphytanyl PC. Figure 3 presents the values of SkQ1 diffusion potential as a function of the concentration of this cation in one of the two compartments of the experimental cell separated by the BLM. SkQ1 concentration in the other compartment was constant at 0.1 μM. As seen from the figure, the potential values on the diphytanoyl PC BLM were significantly lower than Nernstian (dashed line), which corresponds to the results of our previous experiments [3-7]. However, for a diphytanyl PC BLM the potentials were close to Nernstian values.
Fig. 2. Kinetics of generation of SkQ1 diffusion potential. The initial SkQ1 concentration in both cell compartments was 0.1 μM. One micromolar SkQ1 was added at zero time to one of the compartments. The incubation mixture contained 10 mM Tris, 10 mM MES, and 10 mM KCl (pH 7.0).
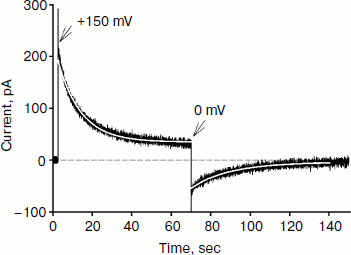
Figure 4 shows the time dependence of current through the BLM in the presence of SkQ1 in response to an application of 150 mV potential to the diphytanyl PC membrane. The application of the potential difference leads to the appearance of a current that decreases with time (current relaxation). As shown earlier, this process is associated with redistribution of the SkQ1 cation between the two sides of the membrane in response to the application of the transmembrane electric field [8]. Significant binding of SkQ1 at the “membrane–water” interface is an important condition for current relaxation. The gray curve in Fig. 4 represents an approximation to the data with a monoexponential curve I = I + (I0 – I)·exp(–t/τ). The characteristic relaxation time (τ) for this process was 11 sec. The subsequent recording shows the current relaxation in response to the removal of the potential. The gray curve in this part of the figure represents an approximation with a monoexponential model with a relaxation time of 23 sec. It should be noted that we could not obtain reliable records of SkQ1 current relaxation for the diphytanoyl PC membrane. For membranes prepared from total E. coli phospholipid, the relaxation time values were significantly higher (τ was close to 60 sec) [8]. Thus, we conclude that the data on the current relaxation also suggest a significantly higher permeability of SkQ1 through the diphytanyl PC membrane than through the diphytanoyl PC membrane.Fig. 3. Dependence of SkQ1 diffusion potential on the concentration gradient of this cation. In one of the compartments [SkQ1] = 0.1 μM. SkQ1 concentration in the other compartment is given on the abscissa. Incubation mixture as in Fig. 2.
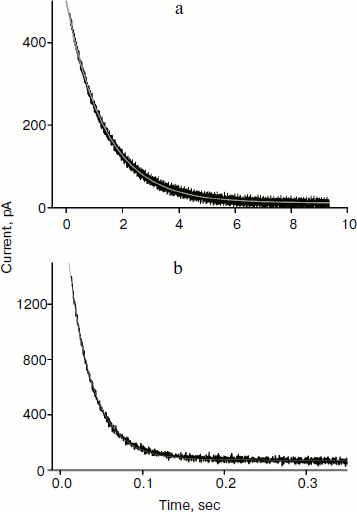
Reliable data on cation permeability for the studied lipids were obtained for the cationic derivative of rhodamine B – C12R4 (Fig. 1). Figure 5 shows recordings of the current induced by C12R4 in response to the application of 100 mV potential for membranes composed of these lipids. The relaxation kinetics are clearly much faster for diphytanyl PC (the time scale for the upper panel is 10 sec, while for the bottom panel it is 0.35 sec). The characteristic relaxation time τ for diphytanyl PC is 0.03 sec, and for diphytanoyl PC it is 1.36 sec (gray curves in Fig. 5).Fig. 4. Recording of current through a diphytanyl PC BLM in the presence of 2 μM SkQ1 upon application to the membrane of +150 mV, followed by removal of the potential. The gray curves are monoexponential approximations with time constants of 11 and 23 sec. Medium composition: 100 mM KCl, 10 mM Tris, pH 7.4.
As noted above, the higher cation permeability through the diphytanyl PC membranes is caused by the lower value of the dipole potential of lipids that have ether linkages and therefore contain no carbonyl groups. The magnitude of the dipole potential change can be estimated in our experiments on the basis of the ratio of permeabilities of C12R4 for these two membranes in accordance with the equation [12]Fig. 5. Recording of the current in diphytanoyl PC BLM (a) or diphytanyl PC BLM (b) in the presence of 0.1 μM C12R4 upon application to the membrane of +100 mV potential. The gray lines are monoexponential curves with time constants of 1.36 sec (a) and 0.03 sec (b). Medium composition: 100 mM KCl, 10 mM Tris, pH 7.4.
The question arises whether the magnitude of the dipole potential of artificial membranes corresponds to that of natural membranes. Unfortunately, the literature provides no quantitative estimates of the dipole potential on the plasma membranes of cells or mitochondria, although there are a few studies indicating the involvement of the dipole potential in a number of processes taking place in plasma membranes [14]. We think that the presence of peptides and proteins in natural membranes should substantially decrease the dipole potential determined by phospholipids, since they inevitably disturb the continuity of the layer of dipoles of carbonyl groups of phospholipid ester bonds. So the dipole potential of mitochondrial membranes should be closer to that of the diphytanyl PC BLM, and the permeability of these membranes to SkQ1 should be higher than that of diphytanoyl PC BLM.
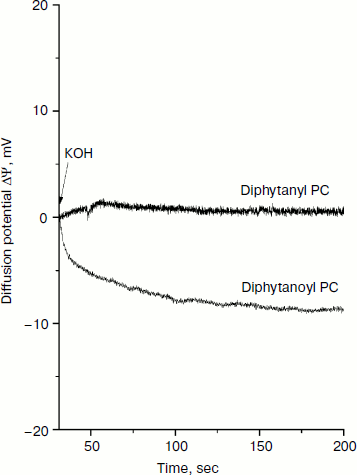
In addition to the influence of the nature of the phospholipid on the permeability of the cations, we also examined the generation of the diffuse potential of hydrogen ions caused by formation of ΔpH on the BLM. The fact is that in earlier experiments we observed some induction of proton conductivity of membranes caused by SkQ1. This effect could be attributed to the combined action of the penetrating cation SkQ1 and fatty acids [15], which appear in the phospholipid as a result of its spontaneous hydrolysis. Since diphytanyl PC contains residues of only phytol, spontaneous hydrolysis would lead to the formation of phytol, which cannot be protonated (in contrast to a fatty acid anion that can bind H+, this process being necessary for the creation of proton conductivity of a BLM). Figure 6 shows the generation of diffusion potential of H+ in the presence of 1 μM SkQ1. Addition of KOH to one of the compartments caused a pH change of 1 unit. As a result, a small but measurable potential (positive in the compartment with lower H+ concentration) was generated for the diphytanoyl PC BLM. In the case of the diphytanyl PC membrane, no potential could be observed, i.e. SkQ1 did not cause the appearance of measurable proton conductivity in such a BLM. Our results support the conclusion that SkQ1-induced proton conductivity of a BLM is determined by the presence of fatty acids produced in the course of hydrolysis of ester bonds between the residues of fatty acid and glycerol in the phospholipid molecule.
Determining the conditions for obtaining Nernstian diffusion potentials for an SkQ1 gradient on a BLM opens up the possibility to use such a BLM as an SkQ1-sensitive electrode for measuring the absorption of SkQ1 by animal cells, mitochondria, and bacteria. This possibility to measure SkQ1 concentration has several advantages over other analytical methods such as chromatography. In this case, it becomes possible to directly measure the kinetics of SkQ1 absorption or release by living systems without the stage of sampling, separation of chemical components, and their subsequent analysis. Such measurements were effective for measuring the concentration of tetraphenylphosphonium cation in the incubation medium to estimate the value of membrane potential in isolated mitochondria and permeabilized hepatocytes [3, 16, 17].Fig. 6. Generation of H+ diffusion potential in the presence of 1 μM SkQ1 in both cell compartments separating a BLM. Initially, the pH in both compartments was 7.0. At 75 sec after the start of the experiment, KOH was added to one of the compartments so as to shift the pH in this compartment to 8.0.
The authors express their gratitude to G. A. Korshunova and N. V. Sumbatyan for the synthesis of the penetrating cations and to D. A. Cherepanov for his suggestion to study diphytanyl PC.
REFERENCES
1.Skulachev, V. P., Sharaf, A. A., and Liberman, E.
A. (1967) Nature, 216, 718-719.
2.Liberman, E. A., Topaly, V. P., Tsofina, L. M.,
Jasaitis, A. A., and Skulachev, V. P. (1969) Nature, 222,
1076-1078.
3.Liberman, E. A., and Skulachev, V. P. (1970)
Biochim. Biophys. Acta, 216, 30-42.
4.Antonenko, Y. N., Avetisyan, A. V., Bakeeva, L. E.,
Chernyak, B. V., Chertkov, V. A., Domnina, L. V., Ivanova, O. Y.,
Izyumov, D. S., Khailova, L. S., Klishin, S. S., Korshunova, G. A.,
Lyamzaev, K. G., Muntyan, M. S., Nepryakhina, O. K., Pashkovskaya, A.
A., Pletjushkina, O. Y., Pustovidko, A. V., Roginsky, V. A.,
Rokitskaya, T. I., Ruuge, E. K., Saprunova, V. B., Severina, I. I.,
Simonyan, R. A., Skulachev, I. V., Skulachev, M. V., Sumbatyan, N. V.,
Sviryaeva, I. V., Tashlitsky, V. N., Vassiliev, J. M., Vyssokikh, M.
Y., Yaguzhinsky, L. S., Zamyatnin, A. A., Jr., and Skulachev, V. P.
(2008) Biochemistry (Moscow), 73, 1273-1287.
5.Skulachev, V. P., Anisimov, V. N., Antonenko, Y.
N., Bakeeva, L. E., Chernyak, B. V., Erichev, V. P., Filenko, O. F.,
Kalinina, N. I., Kapelko, V. I., Kolosova, N. G., Kopnin, B. P.,
Korshunova, G. A., Lichinitser, M. R., Obukhova, L. A., Pasyukova, E.
G., Pisarenko, O. I., Roginsky, V. A., Ruuge, E. K., Senin, I. I.,
Severina, I. I., Skulachev, M. V., Spivak, I. M., Tashlitsky, V. N.,
Tkachuk, V. A., Vyssokikh, M. Y., Yaguzhinsky, L. S., and Zorov, D. B.
(2009) Biochim. Biophys. Acta, 1797, 878-889.
6.Skulachev, M. V., Antonenko, Y. N., Anisimov, V.
N., Chernyak, B. V., Cherepanov, D. A., Chistyakov, V. A., Egorov, M.
V., Kolosova, N. G., Korshunova, G. A., Lyamzaev, K. G., Plotnikov, E.
Y., Roginsky, V. A., Savchenko, A. Y., Severina, I. I., Severin, F. F.,
Shkurat, T. P., Tashlitsky, V. N., Shidlovsky, K. M., Vyssokikh, M. Y.,
Zamyatnin, A. A., Jr., Zorov, D. B., and Skulachev, V. P. (2011)
Curr. Drug Targets, 12, 800-826.
7.Lyamzaev, K. G., Pustovidko, A. V., Simonyan, R.
A., Rokitskaya, T. I., Domnina, L. V., Ivanova, O. Y., Severina, I. I.,
Sumbatyan, N. V., Korshunova, G. A., Tashlitsky, V. N., Roginsky, V.
A., Antonenko, Y. N., Skulachev, M. V., Chernyak, B. V., and Skulachev,
V. P. (2011) Pharm. Res., 28, 2883-2895.
8.Rokitskaya, T. I., Klishin, S. S., Severina, I. I.,
Skulachev, V. P., and Antonenko, Y. N. (2008) J. Membr. Biol.,
224, 9-19.
9.Liberman, Y. A., and Topaly, V. P. (1969)
Biofizika, 14, 452-461.
10.Brockman, H. (1994) Chem. Phys. Lipids,
73, 57-79.
11.Gawrish, K., Ruston, D., Zimmerberg, J.,
Parsegian, V. A., Rand, R. P., and Fuller, N. (1992) Biophys.
J., 61, 1213-1223.
12.Malkov, D. Y., and Sokolov, V. S. (1996)
Biochim. Biophys. Acta, 1278, 197-204.
13.Berkovich, A. K., Lukashev, E. P., and
Melik-Nubarov, N. S. (2012) Biochim. Biophys. Acta, 1818,
375-383.
14.Cladera, J., Martin, I., Ruysschaert, J. M., and
O’Shea, P. (1999) J. Biol. Chem., 274,
29951-29959.
15.Severin, F. F., Severina, I. I., Antonenko, Y.
N., Rokitskaya, T. I., Cherepanov, D. A., Mokhova, E. N., Vyssokikh, M.
Y., Pustovidko, A. V., Markova, O. V., Yaguzhinsky, L. S., Korshunova,
G. A., Sumbatyan, N. V., Skulachev, M. V., and Skulachev, V. P. (2010)
PNAS, 107, 663-668.
16.Kamo, N., Muratsugu, M., Hongoh, R., and
Kobatake, Y. (1979) J. Membr. Biol., 49, 105-121.
17.Labajova, A., Vojtiskova, A., Krivakova, P.,
Kofranek, J., Drahota, Z., and Houstek, J. (2006) Anal.
Biochem., 353, 37-42.