Arguments Against Non-programmed Aging Theories
T. C. Goldsmith
Azinet LLC, Box 239, Crownsville MD, 21032, USA; fax: 1-410-403-2283; E-mail: tgoldsmith@azinet.com
Received June 13, 2013
Until recently, non-programmed theories of biological aging were popular because of the widespread perception that the evolution process could not support the development and retention of programmed aging in mammals. However, newer evolutionary mechanics theories including group selection, kin selection, and evolvability theory support mammal programmed aging, and multiple programmed aging theories have been published based on the new mechanics. Some proponents of non-programmed aging still contend that their non-programmed theories are superior despite the new mechanics concepts. However, as summarized here, programmed theories provide a vastly better fit to empirical evidence and do not suffer from multiple implausible assumptions that are required by non-programmed theories. This issue is important because programmed theories suggest very different mechanisms for the aging process and therefore different mechanisms behind highly age-related diseases and conditions such as cancer, heart disease, and stroke.
KEY WORDS: senescence, ageing, gerontology, biological aging theoryDOI: 10.1134/S0006297913090022
For much of the past century, it was widely thought that the evolution process was directed entirely by individual cost and benefit. An evolved trait had to benefit the ability of individual organisms (or their direct descendants) to survive and/or reproduce. It was also widely agreed that deterioration and death associated with aging did not provide any individual benefit in gradually aging mammals. Programmed aging requires that there be an evolutionary benefit from purposely limiting lifespan. Therefore, programmed mammal aging was considered to be “impossible” and non-programmed aging theories dominated scientific thought on the subject although numerous issues remained.
However, beginning in 1962 a series of evolutionary mechanics theories appeared proposing that wider benefit to groups [1] (group selection), kin [2] (kin selection), the propagation of genes [3] (e.g. selfish gene theory), or the evolution process [4] (evolvability theory) could offset some degree of individual disadvantage and result in evolution and retention of an individually adverse trait like mammal aging. Programmed aging theories then appeared [5-8] that proposed that an organism design that purposely limited the life of the organism generally provided non-individual benefits to most organisms including mammals. The non-individual-benefit theories also provided explanations for observations of other individually-adverse organism traits including altruism, excessive male puberty age, some aspects of inheritance mechanisms, and sexual reproduction in addition to the lifespan observations. The new theories were largely based on relatively recent genetics discoveries.
Some non-programmed aging proponents countered that all of the non-individual-benefit theories were invalid because of the mechanics of mutation propagation. However, multiple proposals appeared [4] providing propagation solutions for the non-individual-benefit theories.
Finally, the zero net evolutionary disadvantage of aging proposed by non-programmed proponents weakened their argument against programmed aging based on non-individual benefit as outlined below.
Consequently, proponents of non-programmed aging have largely abandoned attacks on the non-individual-benefit theories. Some senior and vocal proponents of non-programmed aging have even conceded that the non-individual-benefit theories may be valid [9, 10]. Further, logical attacks on specific programmed aging theories based on the many proposed non-individual benefits of a purposely limited lifespan have not appeared. Instead, the remaining proponents of non-programmed aging contend that their theories provide equivalent performance in matching observations and should have sole consideration by medical researchers [9, 10]. Major difficulties with this idea are described below. Researchers choosing the wrong theory of aging could significantly delay progress in preventing and treating age-related diseases.
COMMON FEATURES OF PROGRAMMED AND NON-PROGRAMMED THEORIES
The following outlines areas for which there is agreement between modern programmed and non-programmed aging theories.
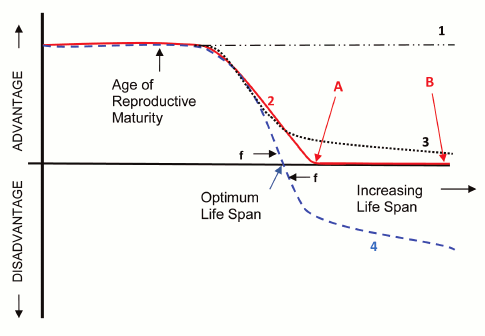
Intrinsic and extrinsic causes of mortality. Darwin’s [11] evolutionary mechanics concept “survival of the fittest” (curve 1, dash-dotted line in Fig. 1) did not suggest that the evolutionary value of surviving and reproducing varied with the age of an organism. According to Darwin, organisms were trying to live as long as possible and breed as much as possible and were acquiring traits through the evolution process that helped in this quest. This concept logically leads to the idea that observed lifespans are the result of fundamental limitations such as laws of physics or chemistry that cannot be overcome by the evolution process. However, lifespans in different mammal species were observed to vary over a huge range of at least 100 to 1, and fish lifespans were seen to vary over a range of at least 600 to 1, from weeks to centuries. In the following 93 years theorists were unable to provide a plausible explanation, based on Darwin’s evolutionary mechanics, as to why there was such an enormous difference in lifespans between different species. This led to modern programmed and non-programmed aging theories, all based on modifications to Darwin’s mechanics, which propose that the evolutionary benefit of survival and reproduction declines with age following reproductive maturity.
In 1952 Medawar [12] introduced the now generally accepted idea that the evolution process must incorporate some relationship between internal and external causes of mortality. The evolutionary benefit of overcoming internal limitations on lifespan and reproduction (i.e. senescence) declined in proportion to the relative importance of external causes to internal causes. For example, we can all agree that there would be zero evolutionary benefit to overcoming internal limitations on lifespan or reproduction beyond the age at which 100% of the individuals in a wild population could be expected to be dead from external causes such as predators, environmental conditions, or lack of habitat or food supply. Medawar proposed that the evolutionary benefit of living and reproducing longer declined to zero at some species-specific age as shown in Fig. 1 (curve 2). Issues associated with the concept of overcoming internal lifespan and reproductive limitations are discussed further below.
Aging and reproductive maturity. We can also all agree with Medawar’s proposal that internal factors that caused even very slight degradation in survival or reproductive fitness prior to the age at which the particular species could complete its first reproduction would be very highly selected against as shown in Fig. 1. We can agree that a species that died of old age prior to reaching puberty would not be viable. Modern programmed and non-programmed theories of aging agree that the age at which an organism is first capable of reproducing is the most important factor in determining the lifespan needed by that organism, although many other species-specific internal and external factors affect the needed lifespan.
Evolutionary disadvantage of aging. Major programmed and non-programmed evolutionary theories of aging concur that at some species-particular age the net (counting all tradeoffs) evolutionary disadvantage of aging must be effectively zero as shown in Fig. 1, curves 2 (solid line) and 4 (dashed line).
Fig. 1. Evolutionary cost/benefit vs. lifespan.
This is true because in the case of essentially any species we can find some similar species with a longer or shorter lifespan, and it is therefore apparent that a species can evolve whatever lifespan is needed by that species. If at that age there was an even very slight advantage to a longer lifespan, the species would presumably have evolved a longer lifespan. Proponents of non-programmed aging contend that beyond the age (point A) at which the benefit of further survival and reproduction declines to zero, there is no further decline as shown in curve 2. Living longer creates no evolutionary disadvantage.
Programmed aging proponents contend that based on modern evolutionary mechanics concepts that allow for non-individual cost/benefit, further life and reproduction conveys an evolutionary disadvantage as shown in curve 4. According to programmed theories, aging and other lifespan limiting traits are beneficial features of organisms that evolved because they cause the possessing species to have an evolutionary advantage. According to non-programmed theories and curve 2, there is evolutionary force only toward achieving the age at which the value of further survival and reproduction declines to zero. According to programmed theories there is evolutionary force toward both achieving the zero-point age and not exceeding it. Beyond the zero-point there is evolutionary force toward limiting lifespan, which leads to the evolution of mechanisms that purposely limit lifespan to a particular optimum value.
The difference between programmed and non-programmed theories is thus essentially the difference between zero benefit of longer life (curve 2) and at least a slight disadvantage of a longer life (curve 4). Proponents of non-programmed aging arguing against non-individual-benefit theories or associated programmed aging theories therefore have to show that all of the non-individual benefit theories and all of the associated programmed aging theories are utterly invalid to the point where none of them could provide an even minute benefit to a purposely limited lifespan – a very difficult case to make.
MEDAWAR’S DILEMMA
A major problem with Medawar’s concept immediately appeared: lifespans of most organisms appeared to be much shorter than could be justified with the above analysis showing that the residual benefit of further survival and reproduction would not be zero until the age at which 100% of a wild cohort would be dead from external causes. Further, gradual aging in many organisms caused obviously significant negative effects on survival and reproductive fitness at much younger ages than death, per se. The evolutionary value of survival and reproduction as a function of age considering only individual-benefit evolutionary mechanics theory therefore appeared to be more like the one shown in Fig. 1, curve 3 (dotted line). For programmed theories this is not a problem because the proposed negative evolutionary effects of a longer life could trade off against the residual positive (individual fitness) effects of a longer life in order to reach the net zero-point at a plausible age as shown in curve 4. Non-programmed aging proponents developed the linkage concept described below in efforts to explain early death and gradual aging.
PERFECTLY RIGID LINKAGE
Many non-programmed aging theorists beginning with Williams’ antagonistic pleiotropy theory [13] in 1957 proposed that there was some unbreakable or perfectly rigid linkage between some individually beneficial trait(s) and the assumed to be adverse trait of aging. The linked beneficial trait(s), sometimes conveniently unspecified, would compensate for the adverse effects of aging allowing aging to be retained despite its presumably adverse effects and allowing a net zero-point to be obtained at a plausible age per curve 2. Many such theories exist to the effect that aging is an unavoidable side-effect of growth, or an unavoidable side-effect of some mechanism that acts to prevent cancer, or an unavoidable side-effect of (insert your own favorite individually beneficial property here).
Of course, the evolution process would presumably be acting to break the linkage to result in an organism design that accomplished the beneficial function without the putatively adverse effect. We can use the term perfectly rigid to describe a linkage that was so unbreakable that the operation of the evolution process would be unable to break the linkage even after extremely long periods of operation (billions of years). This would be necessary because, according to non-programmed theories, aging and death have generally been an evolutionary disadvantage at least since the appearance of eukaryotes. If the linkage had been broken, even in the primordial past, subsequent species would have presumably inherited the beneficial property without the adverse property.
There are a series of arguments against the unbreakable linkage concept. The most obvious is that if coincidental linkages of the sort described here were common, evolution as we observe it in complex organisms would be impossible. As organisms became more complex there would be more and more potential linkages between the increasing number of traits. Does it not seem like a stupendously incredible coincidence that perfectly rigid linkages only adversely affected the ability of organisms to evolve longer lifespans and did not affect their ability to evolve all of their other species-unique traits? Similar organisms like mammals have obviously been able to evolve a huge variety of designs in order to fit into their respective ecological settings, somehow without being bothered by linkage problems.
Another argument is that the differences between similar organisms having major lifespan differences appear to be relatively minor. Some fish lives X years, some other similar fish lives 10X years. Gross lifespan differences between otherwise similar organisms were obviously a problem: Why were the lifespans different if most other (including putatively linked) traits were so similar?
Linkages between different traits caused by the mechanisms of biological inheritance do indeed exist and have varying rigidity, that is, varying resistance to the unlinking force exerted by the evolution process. However, analysis [4] shows that linking mechanisms such as antagonistic pleiotropy suggested by non-programmed proponents are not perfectly rigid and do not explain the indefinite retention of an adverse characteristic.
The perfectly rigid linkage argument was developed at a time when modern non-individual-benefit evolutionary mechanics theories did not exist, and it would not withstand careful scientific analysis today.
Programmed aging theories do not require perfectly rigid linkages that implausibly only affect the evolution of a longer lifespan.
OPTIMUM SPECIES LIFESPAN
As described above, species lifespans appear to vary at least as much if not more than other evolved organism traits. We presume that any organism design parameter such as tail length has an optimum value for a particular species and that therefore there is evolutionary force toward developing and maintaining the optimum value. For tails there is therefore evolutionary force toward both achieving the optimum length and not exceeding it. Per Fig. 1, curve 4, programmed aging theories assume there is evolutionary force (f) toward both achieving and not exceeding a species-unique optimum value for lifespan in a manner similar to other evolved characteristics and that therefore we could expect the observed large inter-species lifespan variations.
However, non-programmed theories (curve 2) propose that there is no evolutionary force toward limiting lifespan and that therefore any lifespan beyond the zero-point (point A) would satisfy the requirements of their theory including point B or any lifespan longer than point A including functional immortality. How then to explain why organisms ended up with just the lifespan needed by that particular organism (point A) leading to the huge lifespan variations? If a species had a longer-lived ancestor, would it not inherit the longer lifespan? Non-programmed aging theories consequently require the assumption that undirected random processes (e.g. random mutations, stochastic changes, etc.) would degrade the lifespan of each organism until it matched point A for that species.
At the outset, it seems implausible that undirected random processes would be as efficient as evolutionary processes specifically directed at an optimum lifespan in resulting in the lifespan observations. Discussion of actual biological mechanisms of aging (below) exposes more severe implausibility with the non-programmed scenario.
AGING MECHANISM ISSUES
As described below, programmed and non-programmed aging theories logically lead to radically different concepts regarding the biological mechanisms responsible for aging and age-related diseases. Discussion of mechanisms discloses additional problems with non-programmed theories.
Aubrey de Grey has advanced an argument [10] (to my knowledge the only argument) against programmed aging that does not depend on assuming the utter invalidity of all of the post-1962 non-individual-benefit evolutionary mechanics theories and/or all of the dependent programmed aging theories. Consequently, it is worth taking the time to describe the many flaws in this argument.
Active (programmed) and passive (non-programmed) maintenance and repair theories of aging both assume generic deteriorative processes (wear and tear, oxidation, telomere shortening, etc.) exist that would cause deterioration in any organism unless actively opposed by the organism. These processes include disease-specific processes such as those that result in cancer (unopposed adverse mutations), or heart disease (arterial deposits or other artery damage) that are very different and presumably involve very different maintenance and repair activities. The passive non-programmed aging theory proposes that the very large differences in lifespan between different mammals result entirely from differences in the operation of their maintenance and repair activities that act to oppose the deteriorative processes. De Grey’s argument is that active and passive mechanisms both result in organisms having the specific lifespan needed by a particular species and that they are consequently functionally identical. If this were true (it is not), there would be no evolutionary motivation to evolve and retain the more complex lifespan regulation mechanisms proposed by proponents of programmed aging. De Grey proposes that his aging mechanism concept (which is compatible with non-programmed aging theories) is valid even if programmed aging theories and their underlying evolutionary mechanics concepts such as group selection or evolvability are valid. De Grey’s argument is directed only at the observed inter-species lifespan differences and ignores many other observations. It also has many logical issues described below. De Grey also ignores requirements and predictions of specific programmed aging theories in his claim that his passive mechanism would be functionally identical to an active mechanism in satisfying a programmed aging theory.
In the passive maintenance and repair concept suggested by de Grey and shown in Fig. 2, aging is ultimately the result of generic deteriorative processes such as oxidation, molecular disruption, genetic transcription faults, mechanical damage, and other natural processes that cause deterioration in biological systems. The gross life span differences are explained by the presence of a large number of independent anti-deterioration functions that act to prevent damage from or repair damage resulting from the generic deteriorative processes. A particular longer-lived mammal species possesses more effective anti-deterioration functions than a shorter-lived species and consequently is able to slow the monotonic accumulation of damage.
Fig. 2. Passive aging mechanism functional diagram.
This concept suggests that organisms possess a potentially large number of maintenance and repair functions that are functionally independent and evolved independently in order to produce the lifespan needed by the organism. If, for example, cancer at too young an age was preventing a species from obtaining the particular lifespan needed by that species, the species would evolve better anti-cancer mechanisms. If heart disease at too young an age was a problem, the species would evolve better anti-heart-disease functions. It is understood that the mechanisms ultimately responsible for cancer, heart disease, and other diverse manifestations of aging are very different and consequently the associated maintenance and repair mechanisms are presumably very different. If for some reason a species needed a shorter lifespan as required by a programmed aging theory, or did not need as long a lifespan as specified by a non-programmed theory, de Grey suggests that all of its maintenance and repair mechanisms would eventually be gradually degraded by random unopposed mutations until the target lifespan was obtained. Therefore, de Grey suggests his passive maintenance concept would satisfy both programmed and non-programmed theories of aging in regard to obtaining the species-specific lifespan needed by each species.
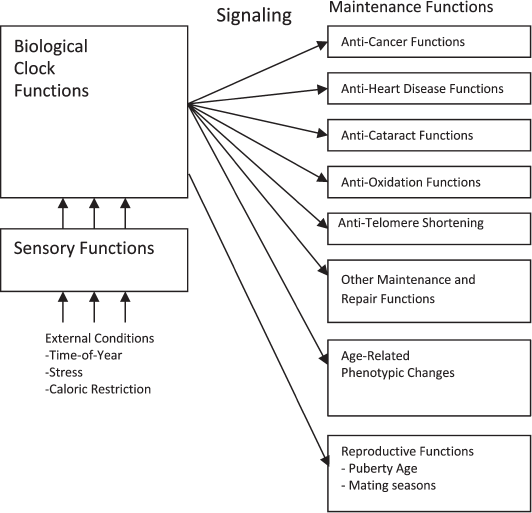
Figure 3 describes a programmed aging concept in which maintenance and repair functions are further controlled by a biological clock mechanism. The clock directs the various maintenance and repair mechanisms to decrease their effects as a species-specific function of age in order to result in the species-specific lifespan. The clock mechanism can in turn be adjusted by sensory functions that can detect and respond to external conditions that alter the optimum lifespan for the organism.
Fig. 3. Programmed aging mechanism functional diagram.
This concept encompasses the idea that not all phenotypic changes associated with aging necessarily involve “damage” or generic deteriorative processes per se. An extension of the growth program could purposely program decreases in muscle strength, reductions in reproductive activity, and other phenotypic changes that are seen as adverse under traditional evolutionary mechanics because these changes create an evolutionary advantage according to a programmed theory of aging. If the program can direct increases in muscle strength and reproductive activity it could obviously also direct decreases in muscle strength and reproductive activity. Programmed decreases in some biological function, sometimes by means of programmed cell death (apoptosis), are common. A tadpole’s tail both grows and shrinks according to its program.
As indicated in Fig. 3, reproductive functions such as age of initial reproductive maturity and mating seasons are also controlled by a biological clock that is adjusted by external conditions such as mating seasons that occur at a particular time of year. There is no scientific disagreement with the idea that reproductive functions are controlled by such a system. The proposal here is that an aging function that evolved because it served a purpose would logically be implemented in essentially the same manner as the reproductive functions and would also involve a biological clock, signaling, and mechanisms that allow for sensing of external and internal (see below) conditions that affect optimum lifespan.
The passive aging mechanism proposed by de Grey and others requires a number of assumptions.
First, it assumes the existence of a potentially large number of different deteriorative processes that would cause organism deterioration unless actively opposed by the organism. If there were no deteriorative processes, presumably aging would not occur according to non-programmed theories. There is no scientific disagreement with this idea and many such processes including wear and tear, oxidation, and telomere shortening in addition to many disease-specific processes have been identified.
Second, it assumes that living organisms possess a potentially large number of different maintenance and repair functions that counteract the deteriorative processes. Again there is no scientific disagreement, and many such functions have been identified. Wounds heal, hair grows, and skin cells are replaced. It is generally accepted that merely maintaining life requires the expenditure of energy and resources. We need to keep breathing even when asleep.
Third, much more controversial and counter-intuitive, it assumes that their proposed evolution process would result in each of the maintenance and repair functions varying in effectiveness over a huge range in order to explain the huge differences in lifespan between otherwise biochemically similar organisms. The anti-cancer mechanisms, the anti-heart disease mechanisms, the anti-cataract mechanisms, and so forth must all be different in humans than in dogs in order to explain the gross difference in the ages at which these symptoms appear.
This idea is very counter-intuitive because it would appear that maintenance and repair functions are generally binary in nature: an organism either can perform the function or it cannot. Once an organism evolved the capability for performing some maintenance and repair function, would not that particular problem be solved for it and its descendant species regardless of the age of the organism? Some reptiles can replace a lost limb, mammals cannot. It is not obvious why “replace skin cells” or other maintenance and repair process would be a different process in an 80-year-old then in a 2-year-old or a different process in a mouse than in a man. If the process is not incrementally different, why would the function need an incrementally different design as organism lifespan incrementally increases? It is easy to see how random mutations or even a single mutation could completely destroy the functioning of a complex maintenance and repair mechanism. It is much harder to see how random mutations could result in a 600 to 1 variation in the effectiveness of such a mechanism to correspond with observed variations in fish lifespan.
This difficulty is progressively more severe with shorter-term maintenance processes. If we discontinue a particular maintenance or repair process, how long does it take for the resulting organism deterioration to produce a significant degradation in survival or reproductive characteristics and therefore be significant in an evolutionary sense? We could use this as a measure of “term”. Now imagine that some process has a term of one year. Perhaps in rats this process is 90% efficient and in dogs it is 99% efficient, and so forth. For reasons described above this is implausible but maybe possible. The problem is that most obvious maintenance and repair functions (hair grows, wounds heal, etc.) have short terms. Sleep is widely accepted as a maintenance and repair function and has a term as defined above of less than 24 h. Are we to believe that the sleep process is 99.999% effective in rats and 99.99999% effective in humans? What would be the specific mechanics of such a process that accommodated such fine variation in response to random mutations?
If indeed there existed deteriorative processes that had relatively long terms we would expect differences in symptoms of aging between short and long-lived organisms. For example, if the mechanisms that cause cancer inherently took a long time to operate, we would not expect to see cancer in relatively short-lived organisms like dogs. This has not proved to be true. Symptoms of aging are very similar between dogs and humans but occur at very different ages.
To summarize, the binary and short-term nature of maintenance and repair functions suggests that if a mammal evolved maintenance and repair mechanisms adequate for a lifespan of three years, that the identical mechanisms would be good enough for a lifespan of three hundred years! The issues described above suggest that non-programmed aging would work better in very short-lived organisms. Perhaps such an organism would have a very short lifespan because it simply did not possess relatively longer-term maintenance and repair mechanisms. This too fails to match observations: Caenorhabditis elegans, with a lifespan of 21 days, has a complex programmed aging mechanism [14]!
According to all of the evolutionary programmed and non-programmed theories of aging, the most important single factor that determines how long a particular species needs to live is the age at which it is first capable of reproducing. As described above, it is very widely accepted that this reproductive function is controlled by a biological program that in turn is capable of detecting and adjusting to external conditions such as planetary seasons. It would therefore appear to be obviously beneficial for an organism to be able to alter its lifespan to accommodate changes in its own age of reproductive maturity or other reproductive parameter. An aging program would be able to do this where the non-programmed concept would not.
Many temporary or local external conditions could plausibly temporarily affect the optimum lifespan for an organism including harsh environmental conditions, predation, decreased food supply, overpopulation, and other forms of stress. An organism that could temporarily alter its lifespan in response to these external conditions would have obvious evolutionary advantages. For example, harsh environmental conditions or heavy predation could increase the externally caused mortality in a population. An organism that could change its internally imposed lifespan to compensate would thus have an advantage. The ability of organisms to adjust biological mechanisms in response to local and temporary external conditions is very common. In connection with lifespan, the programmed mechanism would have this capability while the non-programmed mechanism would not.
The aging mechanism proposed by de Grey and other non-programmed aging proponents (multiple different maintenance and repair mechanisms that independently evolved) suggests that mechanisms behind major symptoms of aging are functionally independent and that there may be few if any potentially treatable common factors between otherwise unrelated symptoms of aging such as heart disease and cancer.
Programmed aging mechanisms such as described above suggest that there are many elements of the aging mechanism (clock, signals, sensing features, etc., i.e. the “program”) that are common to many symptoms of aging and represent targets for intervention. Evidence overwhelmingly supports the programmed mechanism. Stress in the form of caloric restriction or exercise [15] generally delays aging. Single-gene human genetic diseases such as Hutchinson–Guilford progeria [16] and Werner syndrome [17] greatly accelerate multiple symptoms of aging. Some pharmaceutical agents such as statins [18] have been shown to have a beneficial effect on both heart disease and cancer.
The existence of apparently non-aging (negligibly senescent) organisms [19] acts to confirm the idea that, once evolved, maintenance and repair mechanisms could result in at least extremely long and possibly indefinitely long lifespans. Under the programmed mechanism concept, such observations can be explained as failures in the programming mechanism (e.g. in the clock or other common element) that caused the organism to fail to limit its lifespan. According to programmed aging theories the negligibly senescent species would be at an evolutionary disadvantage and therefore likely to become extinct, an idea that is compatible with the relative rarity of negligibly senescent species in comparison to the much larger number of similar senescent species. Non-programmed aging proponents are forced to implausibly contend that the negligibly senescent species for some conveniently unspecified reason needed an extremely long life and therefore evolved extremely efficient maintenance and repair mechanisms. Perhaps they are 99.999999999999999% efficient!
EMPIRICAL EVIDENCE
Observations overwhelmingly favor programmed lifespan regulation mechanisms. Genes have been discovered that cause aging [20]. Many species such as salmon, octopus, and many semelparous species have very explicit suicide mechanisms [21]. Examples of signaling and detection of external conditions in explicit lifespan regulation mechanisms have been discovered [14].
Traditionally, proponents of non-programmed mammal aging have claimed that all such instances are examples of special cases where an organism needed a programmed lifespan limitation according to their aging theory based on individual-benefit-only mechanics. They propose that according to their theory, these special cases do not apply to most mammals and that therefore humans and most mammals possess non-programmed aging mechanisms despite all the contrary evidence. They consequently dismiss examples of obviously programmed lifespan limitation in non-mammals as irrelevant to human aging.
In contrast, programmed aging theories generally contend that most complex species would benefit from a purposely limited lifespan. Some theories [6, 22] supply arguments that more complex organisms such as mammals have a greater need for a purposely limited lifespan than simpler organisms and even suggest that gradual aging would have evolutionary benefits over biological suicide or semelparity. Non-programmed aging proponents, having desisted from attempts to show that programmed theories and their underlying evolutionary mechanics concepts are invalid, cannot so easily dismiss non-mammal observations. If an octopus needs a suicide mechanism, why would a more complex organism not need lifespan regulation? Arguing against this logic would require that they argue against specific programmed aging theories or their underlying evolutionary mechanics concepts, and they have declined to do so.
Any claims that non-programmed passive aging mechanisms satisfy the need for programmed aging should make at least some attempt to describe how passive mechanisms would satisfy the many relevant observations described above as well as some attempt to satisfy the requirements of specific programmed aging theories or explain why those requirements are invalid. Programmed aging theories say a complex programmed lifespan regulation mechanism would evolve because of the many evolutionary advantages it would convey. If this is not the case, why not? If non-programmed mechanisms are generally as effective as programmed mechanisms, why is there so much direct evidence of programmed mechanisms?
Since the development of the first formal programmed aging theory in 1882 [23], the main issue with aging theory has always been the underlying evolutionary mechanics. Traditional individual-benefit-only evolutionary mechanics (as modified by Medawar’s ideas) leads to non-programmed mammal aging theories. Non-programmed aging theories were developed during an era when it was a given that programmed mammal aging was “impossible” because of the constraints of the then universally accepted individual-benefit-only evolutionary mechanics theory.
The newer non-individual-benefit theories lead rather inevitably to programmed aging theories. As described here and in more detail elsewhere [4], programmed aging theories provide a vastly better fit to empirical evidence and do not require the consecutive implausible assumptions required by non-programmed theories. There are many plausible group, kin, or evolvability benefits to a purposely limited lifespan.
The programmed and non-programmed theories have grossly different predictions regarding aging mechanisms and therefore methods for treating and preventing massively age-related diseases. The importance of this issue deserves careful attention by a wide scientific community.
REFERENCES
1.Wynne-Edwards, V. (1986) Evolution Through Group
Selection, Blackwell.
2.Hamilton, W. (1963) Am. Naturalist,
97, 354-356.
3.Dawkins, R. (1986) The Selfish Gene (revised
edition), Oxford University Press.
4.Goldsmith, T. (2013) The Evolution of Aging
(3rd Edn.) Azinet Press, Crownsville, MD, USA.
5.Goldsmith, T. (2012) Biochemistry (Moscow),
77, Phenoptosis Issue (No. 7), 729-732.
6.Skulachev, V. (1997) Biochemistry (Moscow),
62, 1191-1195.
7.Mittledorf, J. (2006) Evol. Ecol. Res.,
8, 561-574.
8.Libertini, G. (2008) Sci. World J.,
8, 182-193.
9.Kirkwood, T., and Melov, S. (2011) Curr.
Biol., 21, R701-R707.
10.De Grey, A. (2007) Ann. N. Y. Acad. Sci.,
1119, 296-305.
11.Darwin, C. (1859) On the Origin of
Species, John Murray, Albemarle Street, London.
12.Medawar, P. B. (1952) An Unsolved Problem of
Biology, H. K. Lewis & Co., London.
13.Williams, G. (1957) Evolution, 11,
398-411.
14.Apfeld, J., and Kenyon, C. (1999) Nature,
402, 804-809.
15.Spindler, S. (2005) Mech. Ageing Dev.,
126, 960-966.
16.Eriksson, M., Brown, W. T., Gordon, L. B., Glynn,
M. W., Singer, J., Scott, L., Erdos, M. R., Robbins, C. M., Moses, T.
Y., Berglund, P., Dutra, A., Pak, E., Durkin, S., Csoka, A. B.,
Boehnke, M., Glover, T. W., and Collins, F. S. (2003) Nature,
423, 293-298.
17.Gray, M., Shen, J., Kamath-Loeb, A., Blank, A.,
Sopher, B., Martin, G., Oshima, J., and Loeb, L. (1997) Nature
Genet., 17, 100-103.
18.Poynter, J., Gurber, S. B., Higgins, P. D. R.,
Almog, R., Bonner, J. D., and Rennert, H. S. (2005) New England J.
Med., 352, 2184.
19.Bennett, J. T., Boehlert, G. W., and Turekian, K.
K. (1982) Maritime Biol., 71, 209-215.
20.Arantes-Oliveira, N., Apfeld, J., Dillin, A., and
Kenyon, C. (2002) Science, 295, 502-505.
21.Wodinsky, J. (1977) Science, 198,
948-951.
22.Goldsmith, T. (2010) Russ. J. Gen. Chem.,
80, 1434-1446.
23.Weismann, A. (1882) Uber die Dauer des
Liebens, Fischer, Jena.