REVIEW: Geological Evidence of Oxygenic Photosynthesis and the Biotic Response to the 2400-2200 Ma “Great Oxidation Event”
J. William Schopf1,2,3
1Department of Earth, Planetary, and Space Sciences, Center for the Study of Evolution and the Origin of Life, and Molecular Biology Institute, 595 Charles E. Young Drive East, University of California, Los Angeles 90095, USA; fax: (310) 825-0097; E-mail: schopf@ess.ucla.edu2PennState Astrobiology Research Center, University Park, PA 16802 University Park, PA 16802, USA
3University of Wisconsin Astrobiology Research Consortium, Madison, WI 53706, USA
Received September 9, 2013
Fossil evidence of photosynthesis, documented in the geological record by microbially laminated stromatolites, microscopic fossils, and carbon isotopic data consistent with the presence of Rubisco-mediated CO2-fixation, extends to ~3500 million years ago. Such evidence, however, does not resolve the time of origin of oxygenic photosynthesis from its anoxygenic photosynthetic evolutionary precursor. Though it is evident that cyanobacteria, the earliest-evolved O2-producing photoautotrophs, existed before ~2450 million years ago — the onset of the “Great Oxidation Event” (GOE) that forever altered Earth’s environment — O2-producing photosynthesis seems certain to have originated hundreds of millions of years earlier. How did Earth’s biota respond to the GOE? Four lines of evidence are here suggested to reflect this major environmental transition: (1) rRNA phylogeny-correlated metabolic and biosynthetic pathways document evolution from an anaerobic (pre-GOE) to a dominantly oxygen-requiring (post-GOE) biosphere; (2) consistent with the rRNA phylogeny of cyanobacteria, their fossil record evidences the immediately post-GOE presence of cyanobacterial nostocaceans characterized by specialized cells that protect their oxygen-labile nitrogenase enzyme system; (3) the earliest known fossil eukaryotes, obligately aerobic phytoplankton and putative algae, closely post-date the GOE; and (4) microbial sulfuretums are earliest known from rocks deposited during and immediately after the GOE, their apparent proliferation evidently spurred by an increase of environmental oxygen and a resulting upsurge of metabolically useable sulfate and nitrate. Though the biotic response to the GOE is a question new to paleobiology that is yet largely unexplored, additional evidence of its impact seems certain to be uncovered.
KEY WORDS: carbon isotopic fraction, cyanobacterial evolution, microfossils, oldest eucaryotes, oxygen-requiring biosynthesis, Precambrian, stromatolitesDOI: 10.1134/S0006297914030018
Abbreviations: ATP, adenosine triphosphate; GOE, Great Oxidation Event; Ma, millions of years; PAL, present atmospheric level.
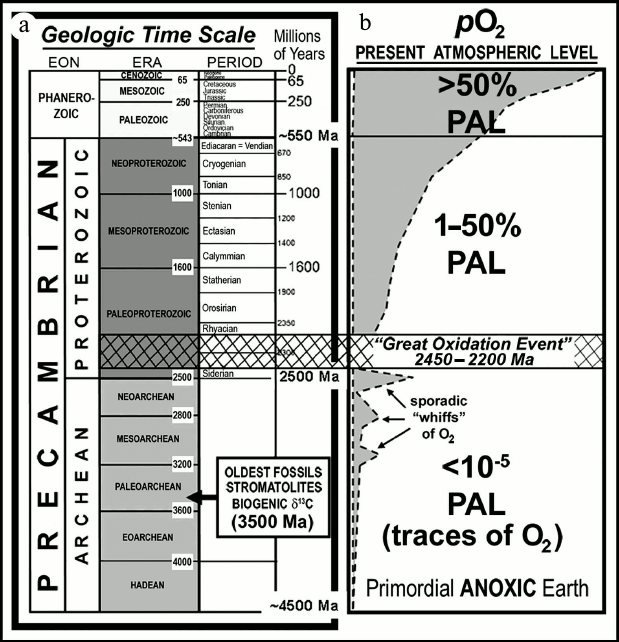
As shown in Fig. 1, geological time is divided into
two major segments: the Phanerozoic Eon, the younger segment
that begins with the first appearance of shelly invertebrate animals
~550 million years (Ma) ago and includes the familiar evolutionary
progressions from algae to seed plants and invertebrates to terrestrial
animals; and the Precambrian Eon, the much longer of the
segments that spans the earlier seven-eighths of Earth history,
extending from the formation of the planet, ~4500 Ma ago, to the
beginning of the Phanerozoic. The Precambrian, in turn, is divided into
two exceedingly long segments — each some 2000 Ma in duration
— the Archean, extending from the formation of the planet
to 2500 Ma ago, and the Proterozoic, spanning the time from 2500
Ma ago to the beginning of the Phanerozoic. The oldest known fossils
date from ~3500 Ma ago [1-5],
with hints of life having been reported from ~3830-Ma-old rocks, among
the oldest known on Earth [6, 7].
Fig. 1. a) Geological time scale. b) Schematic representation of the increase of atmospheric oxygen over geological time and its relation to the “Great Oxidation Event”.
Though it is likely that the earliest forms of life were microbe-like heterotrophs, originating within and metabolically dependent for their foodstuffs on abiotically produced “primordial soup” [8, 9], evidence from the rock record (primarily, microbially produced stromatolites, cellular microscopic fossils, and the carbon isotopic composition of preserved organic matter) establishes that photoautotrophy has served as the foundation of the world’s ecosystem for at least 3500 Ma. The principal unsolved problem is not whether photosynthesis was an exceedingly ancient evolutionary innovation, but, rather, when did oxygen-producing photosynthesis originate, a metabolic process that arose as an evolutionary derivative of a more primitive form of photoautotrophy, anoxygenic photosynthesis, characteristic of non-cyanobacterial photosynthetic bacteria [10, 11].
Among all evolutionary innovations, that having foremost impact on the early ecosystem and subsequent biotic history was the origin of O2-producing photosynthetic cyanobacteria — dating from the earliest, Archean, segment of geological time — the highly reactive O2 produced having provided the biologically useable molecular oxygen required for aerobic respiration, a decidedly more efficient energy-generating process than its anaerobic (fermentative) precursors [9].
The time of origin of this globally altering event can be addressed by answering a single prime question: “When did cyanobacteria originate?” Firm fossil evidence of the existence of cyanobacteria — the earliest-evolved “complete aerobes”, capable both of O2-producing photosynthesis and O2-consuming respiration — would establish that the sequential evolution of metabolic innovations that led to their emergence had by that time already occurred, giving rise to an ancient, but metabolically fully modern, ecosystem [9, 12].
Evidence to answer this question should be expected to be preserved in the Precambrian rock record. Indeed, as is shown here, stromatolites, microbially layered deposits dominated today by filamentous and coccoidal cyanobacteria, are present throughout virtually all of the known geological record; cellularly preserved fossils of cyanobacteria dominate the documented history of Precambrian life; and rock-derived carbon isotopic data are consistent with the presence of photosynthetic microorganisms back to ~3500 Ma ago and, possibly, to >3800 Ma ago.
Nevertheless, as is also shown here, a firm answer to the question of the time of origin of oxygen-producing cyanobacteria is not available: the earliest known stromatolites might have been formed by anoxygenic, rather than O2-producing photosynthesizers; the cyanobacterium-like fossils in rocks as old as ~3500 Ma might be remnants of non-O2-producing microbes; and though a vast amount of carbon isotopic data is consistent with the presence of photosynthesis as early as ~3500 Ma ago, they do not rule out the possibility that the role of primary producer in Earth’s most ancient ecosystems was played by anaerobic, anoxygenic, photosynthetic bacteria rather than by O2-producing cyanobacteria.
It is not surprising that the question of the time of origin of cyanobacteria (and, thus, of O2-producing photosynthesis) is yet unresolved. In contrast with paleontological studies of the Phanerozoic history of life, the basic outlines of which were already known in the mid-1800s [13], successful investigation of the earlier, Precambrian, fossil record dates only from the mid-1960s, more than a 100 years later [14-17]. Although much progress has been made during the ensuing decades (see, for example [18]) — showing that Precambrian microbes were abundant, ubiquitous, metabolically diverse, and biotically predominant — knowledge of the early fossil record remains far from complete.
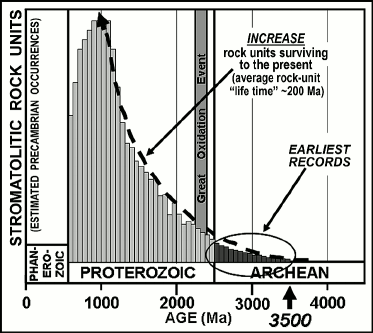
Moreover, due to the “geologic cycle”, the repeated sequence of mountain building, erosion, and deposition of the eroded detritus into sedimentary basins that characterizes Earth’s surface, the average “lifetime” of a geological unit is only some 200 Ma. For this reason, the rock record that has survived to the present rapidly diminishes with increasing geological age, a petering-out that severely limits the abundance of ancient potentially fossiliferous terrains available for study. Thus, it is estimated that about half of the potentially fossil-bearing sedimentary rocks exposed at Earth’s surface date from the Phanerozoic (the most recent one-eighth of geological time); that the remaining ~50% are of Precambrian age, spanning the earlier seven-eighths of Earth history; and that Archean-age rocks — those older than 2500 Ma in which evidence of the earliest oxygenic photosynthesizers is expected to occur — represent only about 5% of the surviving rock record [19]. Indeed, only two sets of such especially ancient, Paleoarchean (~3200-3600-Ma-old; Fig. 1) sedimentary rocks are known to have survived to the present, those of the Western Australia Pilbara Craton and the South African Barberton Mountain Land in and near the Kingdom of Swaziland. Both contain microbial fossils, the biohermal stromatolites they constructed, and isotopically distinctive carbonaceous matter that evidences the presence of photosynthesis. Life’s history and the existence of a photoautotroph-dominated biosphere extend to the very earliest stages of the continuous rock record.
Nevertheless, and although the documented fossil record extends deep into the Precambrian — throughout all of the Proterozoic and much of the Archean — in units older than ~2000 Ma it becomes increasingly sparse as potentially fossil-bearing units become increasingly rare and the fossils that are preserved become increasingly difficult to decipher, due to the geologic cycle-related metamorphic “pressure-cooking” of the surviving rock units.
THE GREAT OXIDATION EVENT (GOE)
Despite the problems posed by the petering-out of the rock and fossil records over geological time, the evidence that has survived is sufficient to establish the presence of appreciable amounts of environmental molecular oxygen — and, by implication, of cyanobacterial oxygen-producing photoautotrophs — at least as early as ~2500 Ma ago.
As discussed by Holland ([20] and references cited therein), iron oxide-rich sedimentary rocks known as banded iron formations (BIFs) are globally abundant during and earlier, but not later than the earliest Proterozoic 2500-to-2300-Ma-old Siderian (Fig. 1). These chemically precipitated sediments — having a distribution especially well known because of their economic importance as the source of iron for the production of steel — are interpreted as having formed from the upwelling of soluble ferrous iron (Fe2+), derived from deep-sea fumaroles over early Earth history, into relatively shallow waters where it reacted with dissolved oxygen to produce insoluble iron-oxides and hydroxides (e.g. Fe2O3, FeO[OH]). Formation of BIFs thus “sponged-up” locally available oxygen as the precipitated oxidized minerals rained onto the ocean floor and, because such BIFs were globally widespread, the abundance of molecular oxygen in the Archean atmosphere and oceans remained vanishingly low. Over time the abundance of BIFs diminished as ocean waters were gradually swept free of ferrous iron, a necessary precursor to the transition to a stable molecular oxygen-containing environment.
The transition from an essentially anoxic to a more oxygen-rich environment — the Great Oxidation Event (GOE) — occurred between ~2500 and ~2200 Ma ago [20, 21]. Beginning near the end of this episode and continuing to the present, sandstones known as red beds have been deposited on land surfaces by meandering rivers and windblown dust. Such units are colored red by the presence of the oxidized iron mineral hematite (Fe2O3) that by occurring as a thin veneer on individual quartz sand gains shows that the atmosphere at the time was oxidizing. In contrast, in terrains older than about 2500 Ma, such red beds are essentially unknown and conglomeratic rocks deposited at the mouths of rivers in shallow-water deltaic settings contain abraded sub-rounded grains of pyrite (FeS2) and uraninite (UO2). If there had been appreciable molecular oxygen in the >2500-Ma atmosphere and near-surface environment, these river-carried detrital grains would have been oxidized and dissolved during transport and deposition. Studies of similarly aged paleosols, preserved ancient soil horizons, confirm this interpretation: paleosols younger than ~2200 Ma show evidence of oxidative weathering whereas those older than ~2500 Ma typically do not [20].
The Archean abundance of banded iron formations, combined with the distinctly differing temporal distributions of red beds and pyritic uraniferous conglomerates and of oxidized and unoxidized paleosols, indicates that there was an increase in the amount of oxygen in Earth’s atmosphere some 2500 to 2200 Ma ago. In recent years, the onset of this transition has been more firmly defined by studies of the ratios of sulfur isotopes preserved in minerals that evidence a rise in atmospheric O2-content at ~2450 Ma ago [22, 23]. Because O2-producing photosynthesis generates more than 99% of the oxygen in Earth’s atmosphere, and because no other large-scale source of molecular oxygen is known, this increase of atmospheric O2 can be firmly attributed to the activities of microbial oxygenic photosynthesizers.
Despite such progress, however, the timing of this major environmental transition sets only a minimum age of ~2450 Ma ago for the origin of O2-producing photosynthesis. In the pre-GOE world the molecular oxygen generated by the earliest cyanobacteria would have been rapidly removed from the environment by its reaction with previously unoxidized substrates (e.g. volcanic gases, unoxidized surface minerals, and BIF-producing huge amounts of dissolved oceanic ferrous iron). And although the development of the Archean atmosphere was no doubt punctuated by sporadic and perhaps localized short-lived “whiffs” of O2 (Fig. 1), such oxygen would have been soon sequestered in rock-forming minerals. Only after such substrates had been more or less completely oxidized — after the “rusting of the Earth” [9] — could the content of atmospheric oxygen have permanently increased, a time-lag from the origin of cyanobacterial O2-producing photosynthesizers that seems certain to have lasted for hundreds of millions of years.
PALEOBIOLOGIC EVIDENCE OF PHOTOSYNTHESIS
Three principal lines of evidence can be used to assess the fossil record of photosynthesis and, of particular interest here, the related question of the time of origin of cyanobacterial oxygenic photosynthesis: stromatolites, cellular microfossils, and the carbon-isotopic chemistry of ancient organic matter. As discussed below, this evidence indicates that O2-producing cyanobacteria were extant earlier than 2450 Ma ago; that such microbes may have been extant 2700 Ma ago; and that cyanobacteria may have been present as early as, or even earlier than, 3500 Ma ago.
Microbial stromatolites. As preserved in the geological record, stromatolites are finely layered rock structures, composed typically of carbonate minerals (e.g. calcite, CaCO3), formed by the microbially mediated accretion of laminae, layer upon layer, from the surface of an ancient seafloor or lake bottom. Their mode of formation has been documented by studies of modern stromatolites, deposits known to microbiologists as “microbial mats”, in which their layered organization reflects the photosynthetic metabolism of mat-building stromatolite-forming microorganisms. Thin (mm-thick) mats composed of such microbes form as the microorganisms multiply and spread across surfaces that are typically intermittently veneered by detrital or precipitated mineral grains that block sunlight. To maintain photosynthesis, mobile members of such communities, such as gliding oscillatoriacean cyanobacteria, move upward through the accumulated mineral matter to establish a new, overlying, microbial mat. The repeated accretion and subsequent lithification of such mats, augmented by other mat-forming microbes (such as colonial chroococcacean and entophysalidacean cyanobacteria and immediately underlying photosynthetic bacteria) can result in the formation of geologically preservable stromatolitic structures that range from small millimetric pustular mounds and columns to large, decimetric, bioherms.
In relatively rare instances, during diagenesis (the series of changes that lead to the lithification and geological preservation of such sedimentary structures), silica dissolved in ground water or the surrounding ocean (converted over time to the mineral quartz, SiO2) can replace the initially precipitated carbonate minerals. If replacement occurs early in the history of a deposit, before the mat-building microorganisms decay and disintegrate, cellularly intact microbes can be preserved. However, the vast majority of fossil stromatolites, unaltered by such replacement, are devoid of cellularly preserved microbes: during diagenesis, carbonate grain-growth crushes and obliterates the stromatolite-forming microorganisms leaving only an amorphous thin coaly residuum of microbe-derived carbonaceous matter.
In the modern world, living stromatolites are known best from refugia, such as hypersaline lagoons, in which the slow-growing microbial mats are not disrupted by grazing or burrowing metazoans. Stromatolites are therefore not especially common in sediments of the Phanerozoic when such invertebrates were abundant. Throughout virtually the entire Precambrian, however, before the origin of metazoans, stromatolites were abundant worldwide in photic-zone carbonate-depositing settings. Known earliest from rock units ~3500 Ma in age, the temporal distribution of stromatolites parallels the surviving Precambrian rock record, stromatolite-bearing units becoming decreasingly abundant as the rock record gradually peters out (Fig. 2). Such structures establish the presence of flourishing photosynthesis-based microbial communities but only rarely do they preserve the cellular fossils that might evidence whether the stromatolite-building photoautotrophs were oxygenic, like cyanobacteria, or anoxygenic, like photosynthetic bacteria.
Fig. 2. Schematic representation of the increase of stromatolite-containing geological rock units over Precambrian time and the corresponding increase of rock units surviving to the present. The late Proterozoic decrease in preserved stromatolitic units is attributable to their disruption by the rise of burrowing and grazing metazoans.
An impressive number of Archean-age geologic units — of particular interest because of their potential bearing on the time of origin of cyanobacteria — contain microbially produced stromatolites. Shown in Fig. 3 (see color insert) are representative examples: sediments of the ~2723-Ma-old Fortescue Group of Western Australia contain domical, pseudocolumnar and branching stromatolites; those of the ~2985-Ma-old Insuzi Group of South Africa include stratiform and conical forms; those of the ~3388-Ma-old Strelley Pool Chert of Western Australia contain domical, stratiform and close-packed conical stromatolites patchily distributed over many tens of square-kilometers; and those of the ~3490-Ma-old Dresser Formation of Western Australia contain domical and stratiform stromatolites. The presence of conical stromatolites in such deposits, reported from 17 of the 48 stromatolitic Archean units now known [3, 24-26], is particularly noteworthy since this distinctive morphology evidently requires the presence of “highly motile mat builders” such as oscillatoriacean cyanobacteria ([27], pp. 342-343).
Fig. 3. Archean (>2500 million year old) stromatolites. Domical, pseudocolumnar, and branching stromatolites overlain by rippled sediments (a1), and a domical stromatolite (a2), from the ~2700-Ma-old Fortescue Group of Western Australia. Stratiform and pseudocolumnar stromatolites (b1) and a conical stromatolite (b2) from the ~3000-Ma-old Insuzi Group of South Africa. Conical stromatolites (c1 and c2) from the ~3430-Ma-old Strelley Pool Chert of Western Australia. Domical (d1) and stratiform (d2) stromatolites from the ~3490-Ma-old Dresser Formation of Western Australia.
Cellular fossils. Two principal processes preserve organic-walled microbial fossils: compression and permineralization. Compression-preserved microorganisms occur in fine-grained detrital sediments such as siltstones and shales, pressed and flattened along bedding planes as the sediment lithified. Although Phanerozoic paleontologists, who focus chiefly on megascopic fossilized remains, have largely neglected such carbonaceous compression-preserved microbes they are appreciably better documented in the Precambrian [28].
The microbial fossil record is best known from microorganisms preserved by permineralization. Of all modes of fossil preservation, this process (known also as petrification) provides the most faithful representation of cellular three-dimensional life-like morphology. Permineralization results from the pervasion of mineral-charged solutions into cells during the early stages of diagenesis, prior to their decay and disintegration. The permeating fluids infill microscopic voids — replacing the watery milieu of the cellular components — to produce a mineral-infused inorganic-organic mix that preserves physically robust structures such as organic-rich cell walls (and a process not unlike that used to epoxy-embed modern microorganisms for study by transmission electron microscopy). As a result, both the organismal morphology and cellular anatomy of such fossils can be preserved in microscopic detail. The most common such permineralizing matrix is silica, fine-grained (cryptocrystalline) quartz and the mineral that comprises the rock-type known as chert. Such microbe-preserving cherts are known from several hundred Precambrian geological units when silica was abundant in the world’s oceans, much earlier than the Phanerozoic appearance of silica-biomineralized sponges, diatoms, and radiolarians that today regulate the oceanic silica budget.
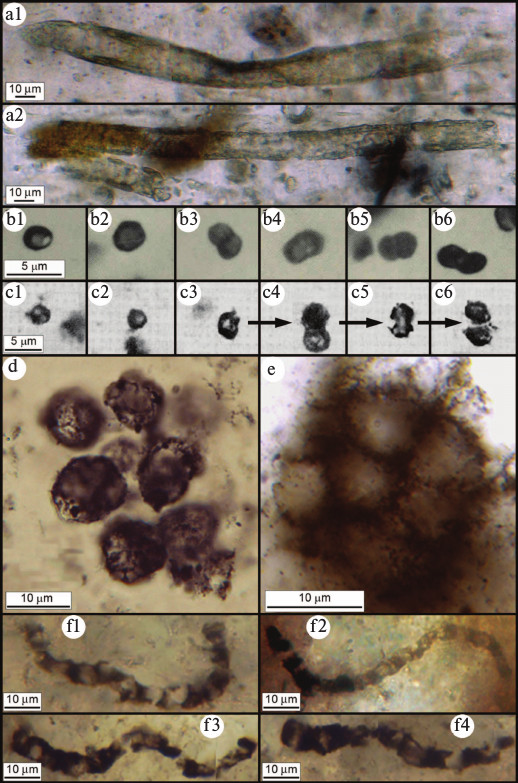
The fossil record of the most abundantly preserved cyanobacterial families (the Oscillatoriaceae, Nostocaceae, Chroococcaceae, and Entophysalidaceae) has recently been reviewed [29], a summary showing that although the cyanobacterial lineage extends deep into the geological past — and that many members of these families have evidently evolved little or not at all over thousands of millions of years [30] — the time of origin of this clade has yet to be established. Because of the continuity of the Proterozoic microbial fossil record, cyanobacteria of this age can be related with considerable certainty to genera and even species of the modern biota. However, because of the incompleteness and fossil-altering metamorphism of the surviving Archean rock record, many such especially ancient fossils have been classed as Bacteria Incertae Sedis (fossil prokaryotes of the Bacterial Domain that cannot be referred with certainty to a particular bacterial group). And though the known record of Archean microbial fossils is sparse relative to that of the Proterozoic, reported from ~50 rock units and comprising only six broad bacterium-like morphotypes [3], 14 of these units — containing many hundreds of documented specimens — date from between 3200- and 3500-Ma-ago, well-evidencing the presence of microbial life this early in Earth history [5, 31-35] (Fig. 4; see color insert).
Fig. 4. Archean (>2500 million year old) microfossils. a) Cylindrical microbial sheaths from the ~2516-Ma-old Gamohaan Formation of South Africa [31]. b) Microbial single cells and dyads from the ~2600-Ma-old Monte Cristo Formation of South Africa [32]. c) Microbial single cells exhibiting, in (c3) through (c6) a preserved cell-division sequence, from the ~3260-Ma-old Swartkoppie Formation of South Africa [33]. d) Colonial coccoidal microbes from the ~3200-Ma-old Farrel Quartzite of Western Australia [34]. e) Colonial coccoidal microbes from the ~3388-Ma-old Strelley Pool Chert of Western Australia [35]. f) Cellular filamentous microbes from the ~3465-Ma-old Apex chert of Western Australia [5].
For virtually all Archean microbial fossils, the uncertainty in their classification stems from their morphological similarity both to cyanobacteria and to non-cyanobacterial microorganisms. The problem in their identification stems primarily from a lack of paleobiologically definitive techniques by which to establish their physiology. Because of microbial mimicry — in this instance, the morphological similarity of oxygenic and non-oxygen-producing microbes [9, 36] — organismal and/or cellular size and shape cannot, in and of themselves, provide firm evidence of physiology [2]. This uncertainty could be resolved were the Archean fossil record, like that of the Proterozoic, sufficiently continuous and well-documented to unambiguously link younger fossils of established affinities to their older and typically less-well preserved evolutionary precursors.
Biomarkers and carbon isotopes. Extraction, isolation, and identification by gas chromatography–mass spectroscopy of organic biomarkers, particularly of various types of hydrocarbons, have provided useful insight into the nature of Precambrian life. For example, identification of the protozoan biomarker tetrahymenol in ~930-Ma-old sediments of the Grand Canyon, Arizona [37], supported by the presence of fossils of testate amoebae in the same Proterozoic sedimentary sequence [38-41], has established a minimum age for the origin of protozoan protists.
In general, however, such studies have not proven useful to Archean-age deposits. The most promising report is that of steranes (hydrogenated derivatives of steroids, such as cholesterol) identified in extracts of ~2700-Ma-old carbonaceous shales of northwestern Australia [42]. This finding is unexpected, since steroids occur almost exclusively in eukaryotic cells [42-44], principally as the platy components of intracellular membranes, and assured fossil eukaryotes (relatively large-celled spheroidal phytoplankton) are known earliest from sediments ~1800 Ma in age [40] that are nearly a billion years younger than the sterane-containing rocks.
However, if the reported steranes date from ~2700 Ma ago, their occurrence would seem to indicate that biologically produced molecular oxygen must have been present in the local environment: steroid biosynthesis involves numerous O2-requiring enzyme-mediated steps (as discussed below, beginning with the cyclization of squalene [43, 44]). The presence of ~2700-Ma-old steranes would therefore imply that O2-producing photosynthesizers must also have been extant since there is no other plausible source for production of the concentrations of free oxygen required for steroid synthesis. Unfortunately, however, interpretation of these biomarkers is open to question. Although there is no doubt that the sterane-containing shales have been dated correctly, potential contamination from modern sources (e.g. from drilling fluids or introduced during laboratory analyses) is an ever-present problem in such studies. Moreover, virtually all organic compounds are soluble to some extent in ground water and for this reason can be introduced into rocks long after their deposition. Because there are no techniques by which to directly date the organic compounds extracted from ancient sediments, it is difficult to show with certainty that such biomarkers are as old as the rock in which they have been detected.
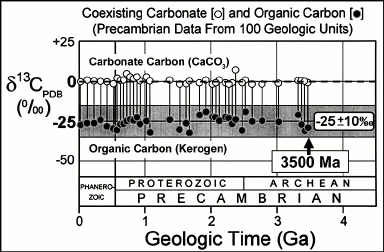
Initiated by the pioneering studies of Park and Epstein [45] and Hoering [46], data have been amassed from thousands of analyses of the carbon isotopic compositions of inorganic carbonate minerals and carbonaceous kerogens coexisting in Precambrian sediments [47]. Based on the relative abundances in such materials of the two stable isotopes of carbon, 12C and 13C, such data show a consistent difference between the inorganic and organic carbon analyzed that extends from the present to ~3500 Ma ago (Fig. 5). The enrichment of the fossil organic matter in the lighter isotope, 12C, relative to coexisting carbonate (a proxy for the seawater-dissolved CO2 required for its formation), and the magnitude of the isotopic difference (expressed as δ13CPDB values) between the inorganic and organic carbon reservoirs that falls typically within a range of 25 ± 10‰, are consistent with the carbon isotopic fractionation that occurs as a result of Rubisco (ribulose bisphosphate carboxylase/oxygenase)-mediated CO2-fixation in O2-producing cyanobacteria [48-51]. Such evidence of carbon isotopic fractionation is well documented in rocks ~3200 to ~3500 Ma in age, the oldest microfossil- and stromatolite-bearing deposits known (Fig. 5).
Fig. 5. Carbon isotopic record over geological time; data from [47].
Despite this strong continuous carbon isotopic evidence of photosynthesis dating to ~3500 Ma ago, it does not necessarily reflect the Archean presence of cyanobacteria. Because of the mixing of carbonaceous matter from diverse biologic sources that occurs during sediment deposition and the alteration of carbon isotopic compositions that can occur during geological metamorphism, the δ13CPDB values of the analyzed kerogen range broadly (±10‰) and, thus, are consistent not only with primary production by cyanobacteria but, as well, by non-O2-producing photosynthetic bacteria. Archean kerogens may have been derived from either or both of these sources. Interpretation of the data is further complicated by the presence in Archean sediments of carbonaceous matter so enriched in 12C as to be plausibly derived only from CH4-metabolizing methanotrophs, indicating that methane-producing Archaea played a significant role in Earth’s early ecosystems [52, 53], and by the presence of C12-rich graphitic carbon in the Eoarchean Isua Supracrustal rocks at Akilia Island off southwestern Greenland that hints at the existence of photosynthesis ~3830 million years ago [7] but that may be of younger metamorphic origin.
Synopsis. As summarized above, the geological record of stromatolites, microscopic fossils, and carbonaceous matter establish that Earth’s ecosystem has been powered by photosynthesis — whether anoxygenic or oxygenic — since at least 3500 Ma ago. It is equally clear, however, that available data do not provide firm evidence by which to determine when O2-producing cyanobacteria originated from their anoxygenic photosynthetic bacterial evolutionary precursors.
In short, the presence of stromatolites, cyanobacterium-like microfossils, and the isotopic evidence preserved in the carbonaceous matter of the especially ancient geological record are consistent with, but do not establish the time of origin of O2-producing photosynthesis. The earliest, Archean, stromatolites might have been formed by photoautotrophic phototaxic anoxygenic photosynthetic bacteria, rather than by the cyanobacteria that dominate the upper surfaces of such structures today; unlike the demonstrably cyanobacterial fossils in younger, Proterozoic, Precambrian sediments, the filamentous and coccoidal microfossils of Archean terrains might represent remains of anoxygenic photoautotrophs; and the carbon isotopic data available from Archean sediments are consistent with the presence of oxygenic photoautotrophs, anoxygenic photoautotrophs, or both.
It is certain that O2-producing photosynthesis evolved earlier, and evidently much earlier than the rise of atmospheric oxygen that marks the onset of the Great Oxidation Event ~2450 Ma ago [20-23, 54], but how much earlier has yet to be established.
BIOTIC RESPONSE TO THE GOE
In the history of life, the single most important environmental change over all of geological time was the Great Oxidation Event (GOE) that by providing the biologically useable oxygen required for aerobic respiration, a much more efficient energy-generating process than its anaerobic precursors set the stage for the evolutionary development of an advanced, stable, obligately aerobic eukaryote-dominated biosphere. As summarized above, geologically based understanding of the occurrence and timing of the GOE stems largely from the work of H. D. Holland who over many years developed this concept and coined its name [20].
Four lines of evidence are here suggested to reflect a biotic response to this world-altering event.
1) The occurrence of anaerobic (rather than oxygen-requiring) reactions in the earliest-evolved parts of widespread metabolic and biosynthetic pathways that is interpreted to reflect an increase in the availability of molecular oxygen.
2) The rRNA phylogeny-correlated fossil evidence of the evolution of cyanobacteria that documents the immediately post-GOE presence of taxa having specialized cells that protect their oxygen-labile nitrogen fixing enzyme system.
3) The earliest known fossil occurrences of obligately aerobic eukaryotes that closely post-date the GOE.
4) Recently discovered fossilized microbial sulfuretums cellularly preserved in rocks deposited during and immediately after the GOE, their occurrence evidently spurred by an increase of environmental oxygen and a resulting upsurge of metabolically useable sulfate, nitrate, and hydrogen sulfide.
The biotic response to the GOE is a question new to paleobiology that can be expected to be addressed by additional fossil evidence and data provided by molecular biologic and genomic studies of modern microbes.
Metabolic and biosynthetic pathways. Of the many such pathways that illustrate a transition from anaerobic to O2-requiring, three examples are summarized here, those of the metabolic process of aerobic respiration (Fig. 6) and the biosyntheses of fatty acids (Fig. 7) and of steroid hormones and carotenoids/xanthophylls (Fig. 8). In the absence of a reliable “molecular clock” such pathways do not provide firm evidence of the timing of the global transition from anaerobic to aerobic conditions. Nevertheless, they do provide convincing evidence of an evolutionary response to a change in the availability of biologically useable molecular oxygen.
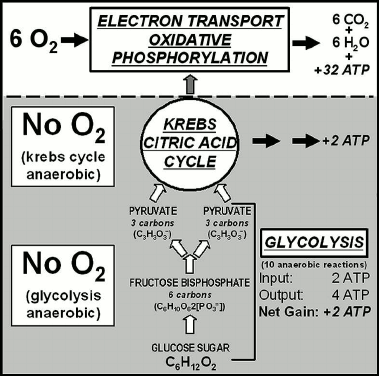
Fig. 6. Metabolic pathway of aerobic respiration showing that the early parts are anaerobic, use of molecular oxygen being a later evolutionary development [9, 43].
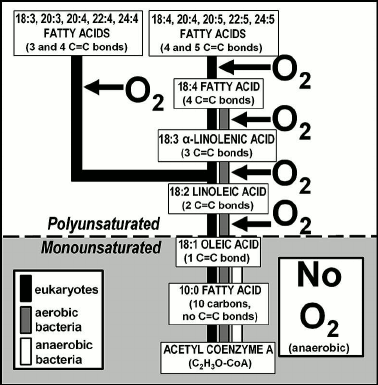
Fig. 7. Phylogeny-correlated biosynthetic pathways of fatty acids showing that the early parts are anaerobic, use of molecular oxygen being a later evolutionary development [43].
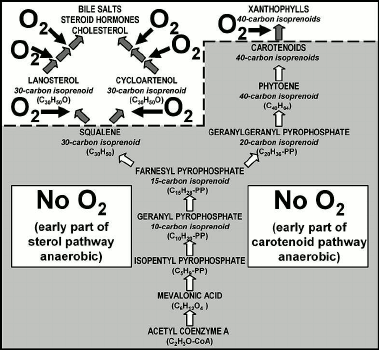
Fig. 8. Biosynthetic pathways of sterols (left) and carotenoids/xanthophylls (right) showing that the early parts of both pathways are anaerobic, use of molecular oxygen being a later evolutionary development [43].
Aerobic respiration, carried out in the mitochondria of eukaryotes (but characteristic also of aerobic prokaryotes such as cyanobacteria) is a three-part process: glycolysis, followed by the Krebs citric acid cycle and then the electron transport system of oxidative phosphorylation (Fig. 6). Glycolysis, the first of the three parts, is an early-evolved ten-step anaerobic metabolic pathway present in all organisms — a biochemical relict of Earth’s earliest biota that inhabited a pre-GOE anoxic world before the origin of O2-generating photosynthesis — that produces minimal cellular energy, two molecules of adenosine triphosphate (ATP) per molecule of glucose metabolized. The second part, the citric acid cycle, similarly produces only two energy-rich ATP molecules from each metabolized glucose molecule. But by oxidizing the products of the glycolysis-citric acid cycle to CO2 and H2O, the third part of the process, the later added-on oxygen-using electron transport system, yields 32 additional ATP molecules (Fig. 6) with the total system yielding 36 ATP molecules (Fig. 6).
Such “anaerobic first-aerobic second” metabolic organization is exhibited also by biosynthetic pathways. Figure 7 summarizes the phylogenetic distribution of the biosynthesis of fatty acids, principal components of cellular membranes. Modern anaerobic bacteria, extant examples of the types of microbes that inhabited the pre-GOE Earth, carry the pathway to 18-carbon oleic acid, a fatty acid that includes a single carbon-carbon double bond (Fig. 7). In contrast, in later-evolved aerobic bacteria and obligately aerobic eukaryotes this pathway has been extended to produce polyunsaturated fatty acids that contain 2-5 symmetrically distributed double bonds (Fig. 7), their biosynthesis carried out by “oxidative desaturation”, the enzyme-mediated formation of double bonds from the addition of one atom of O2 to two atoms of fatty acid-derived hydrogen and the release of a molecule of H2O.
As shown in Fig. 8, the biosyntheses of steroids and carotenoids/xanthophylls are much the same — anaerobic first, aerobic second. The biological significance of steroids such as cholesterol, an essential structural component of animal cell membranes and a precursor for the biosynthesis of bile salts and steroid hormones (e.g. estrogen, testosterone) is well established as is the presence of the accessory pigments carotenoids and xanthophylls in cyanobacteria and in red, brown, and green algae [55-57]. In such microbial and algal photoautotrophs, carotenoids, which protect chlorophyll from photo-oxidation by reacting with nascent oxygen, are the biosynthetic precursors of xanthophylls. In the biosyntheses of steroids and carotenoids/xanthophylls, as in the examples outlined above, use of molecular oxygen has been added to an earlier evolved anaerobic pathway (Fig. 8).
The timing of the origin of the metabolic and biosynthetic use of molecular oxygen has yet to be deciphered. Although O2-requiring steroid synthesis (Fig. 8) may have post-dated the Great Oxidation Event — in bacteria, the earlier evolved anaerobic part of the pathway leading to formation of hopanoids rather than steroids [44] — the aerobic respiration characteristic of cyanobacteria and other O2-using microbes must have pre-dated the GOE, given that the increase of environmental oxygen was a result of cyanobacterial photosynthesis. Similarly, O2-use in the biosyntheses of polyunsaturated fatty acids and xanthophylls, both of which are present in cyanobacteria, presumably also originated earlier than the GOE. Indeed, O2-using metabolic and biosynthetic pathways may have been widespread among microbes prior to the Great Oxidation Event — particularly, if facultative oxygen-users clustered about cyanobacterium-dominated “oxygen oases” where they could use oxygen when available (e.g. during daylight hours) but in its absence could revert to fermentation. The timing of the use of molecular oxygen in such pathways, quite clearly an “add-on” to earlier-evolved anaerobic processes, may ultimately be addressed by development of a reliable genome-based molecular clock.
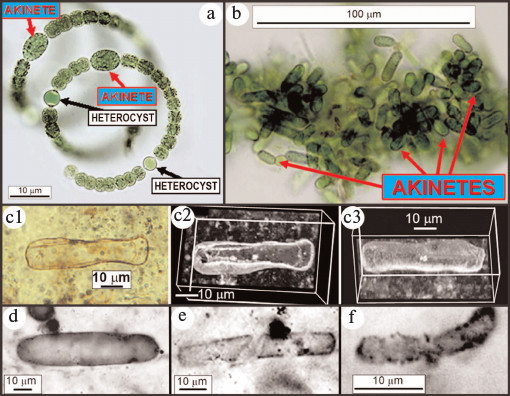
Cyanobacterial evolution. Of the four taxonomic families of cyanobacteria most abundant in modern and fossil stromatolitic assemblages, rRNA phylogenies indicate that three (the Chroococcaceae, Oscillatoriaceae, and Entophysalidaceae) originated early in Earth history whereas the fourth, the Nostocaceae, was of later vintage [58]. Unlike the other three families, later-evolved nostocaceans are characterized by the presence of heterocysts (Fig. 9a; see color insert), specialized spheroidal thick-walled cells that enclose the oxygen-labile N2-fixing nitrogenase enzyme system that hydrogenates N2 to NH3 for synthesis of amino acids.
Fig. 9. Modern nostocacacean cyanobacteria and modern and fossil akinetes. a) Modern spirally coiled Anabaena exhibiting heterocysts and akinetes. b) Modern akinetes (stress-resistant resting cells) produced after disintegration of their parent trichomes. c) Fossil akinete (Archaeoellipsoides) from the ~750-Ma-old Chichkan Formation of Kazakhstan, in (c2) and (c3) shown in confocal laser scanning micrographs [59]. Fossil akinetes from the ~1500-Ma-old Billyakh Group of Siberia (d), the ~1650-Ma-old McArthur Group of Western Australia (e), and the ~2100-Ma-old Franceville Group of Gabon (f) [60].
Triple-bonded diatomic nitrogen, having a bond energy of 226 kcal/mol, is reputed to exhibit among the strongest bonds in nature (appreciably stronger, for example, than the 111 kcal/mol required for breakage of the H–O bond of H2O during oxygenic photosynthesis). Indeed, because of the lack of reactivity conferred by its exceptionally strong bonding, N2 was named “azote” (meaning “without life”) by the French chemist Antoine Lavoisier, a property that explains its absence from common rock-forming minerals and its resulting accumulation in Earth’s atmosphere. Breakage of the triple bond of N2 during biological nitrogen fixation therefore requires a large input of cellular energy, 12 ATP molecules, and a powerful electron-providing reductant, a need met in nostocaceans, as in all N2-fixing microbes, by the iron–sulfur protein ferredoxin:
ferredoxin
N2 + 6e- + 12 ATP + 12
H2O →→→→→ 2
NH4+ + 12 ADP + 4 H+.
The nitrogenase enzyme complex is widely distributed among extant members of early evolved microbial lineages, present not only in cyanobacteria but also in their evolutionary precursors, anoxygenic photosynthetic bacteria, as well as other anaerobic prokaryotes including methanogenic Archaeans. This phylogenetic distribution, together with the inactivation of the complex by molecular oxygen and its absence in eukaryotes, later-evolved obligate aerobes, evidences its early evolutionary origin.
Significantly, and understandable given the energy requirements of the system, fixation of atmospheric nitrogen is used only as a last resort by nostocaceans and other N2-fixing microbes. In all such organisms, ammonia, NH3, hydrogenated to amino-grade, is the preferred nitrogen-source and use of nitrate, NO3-, ranks second, breakage of its N–O bonds requiring 55 kcal/mol, much less than that needed to break the triple bond of N2. On the early Earth, however, neither ammonia nor nitrate was plentiful, ammonia being rapidly photodissociated in the upper atmosphere by the high UV-flux of the primordial Sun (with the hydrogen thus liberated being lost from Earth’s gravitational field) and the production of nitrate from electric discharge (lightning), a significant nitrogen source for the modern biosphere, being prohibited by the absence of O2 from Earth’s anoxic early atmosphere.
Given these constraints and the need by early microbes for enzyme-comprising amino acids, fixation of atmospheric nitrogen — despite its high energetic requirements — was of selective advantage. This situation changed at the GOE when atmospheric oxygen became relatively abundant resulting in formation of a UV-absorbing ozone (O3) layer in the upper atmosphere that shielded ammonia from photodissociation and enabled production of nitrate by lightning discharge. The fossil record shows that the biota then responded, for cyanobacteria by the origin of heterocystous nostocaceans. Because such heterocysts lack photosystem II and thus do not generate oxygen, have thick walls that inhibit inflow of oxygen from adjacent photosynthesizing cells, and rapidly remove nascent oxygen by carrying out aerobic respiration at a rate four-to-five-times greater than the O2-producing cells, they comprise a specialized micro-anaerobic environment within aerobic O2-producing microorganisms [29, 43]. Of paleontological importance, nostocaceans (unlike members of the earlier evolved cyanobacterial families) also produce abundant stress-resistant spores, akinetes (Fig. 9, a and b; see color insert), relatively large robust elongate cells that are preservable in the fossil record. As shown in Fig. 9 (c-f) [59, 60], fossilized akinetes are known as early as, but not earlier than 2100 Ma ago, suggesting that the evolutionary development of akinete-producing heterocystous nostocaceans was a response to the immediately preceding GOE.
Earliest eucaryotes. All eukaryotes are obligately aerobic (or, for anaerobic cellular endosymbionts such as Giardia and mammalian gut-inhabiting chytrid-related fungi, are evolutionary derivatives of obligate aerobes). Thus, the timing of the origin of eukaryotic cells from their earlier-evolved prokaryotic ancestors might be expected to correlate temporally with the Great Oxidation Event, eukaryotes occurring after but not before the GOE.
Given the vagaries of fossil preservation, however, identification of early-evolved eukaryotes is difficult and their most telling attribute, their cell nucleus, is almost never preserved, having been degraded and leached away together with other cytoplasmic components during fossilization. At present, therefore, the earliest occurrence of eukaryotes can be dated most reliably by fossils having relatively large eukaryote-sized cells and eukaryote-like organismal morphology. With regard to cell size, an exhaustive comparison of extant unicellular coccoid prokaryotes and eukaryotes has documented a distinct difference between the groups, the average diameter of such prokaryotes being <3 µm, appreciably smaller than the >10 µm diameter characteristic of coccoid eukaryotes [36]. Thus, the presence of large-celled (60-to-220-μm-diameter) spheroidal phytoplanktonic fossils in ~1800-Ma-old siltstones of northern China (Fig. 10a; see color insert) [61] provides convincing evidence of eukaryotes, as does the occurrence of larger, spirally coiled “alga-like” cellular filaments (Grypania) in ~1600-Ma-old shales of central India (Fig. 10b) [62] and is suggested — albeit less convincingly, because of their lack of preserved cells — by coiled carbonaceous strands in ~1875 (or ~2100-Ma-)-old sediments of northern Michigan, USA (Fig. 10d) [63].
Fig. 10. Early eukaryotes. a) Large-celled (60-to-220-μm-diameter) phytoplanktonic acritarchs from the ~1800-Ma-old Changzhougou Formation of North China [61]. b) Sinuous alga (Grypania) from the ~1600-Ma-old Rohtas Formation of India [62] and (c) a model of its coiled morphology. d) Grypania-like fossils from the >1875-Ma-old Negaunee Iron Formation of Michigan, USA [63].
These earliest known occurrences of fossil eukaryotes, closely post-dating the Great Oxidation Event, are consistent with and plausibly interpreted as evidence of an evolutionary opportunistic biotic response to the GOE.
Mid-Precambrian sulfuretums. The recent discovery of mid-Precambrian anaerobic sulfur-cycling microbial assemblages is similarly consistent with a biological response to an increase of oxygen at the Great Oxidation Event. These sulfuretums, preserved in ~2300-Ma- [64] and ~1800-Ma-old ([65], photo 14-3) carbonaceous cherts of Western Australia, are associated with and closely follow the 2450-2200-Ma ago GOE. Geological studies show that the cellularly preserved biotas of both units occur in marine, anoxic, deep water sub-surface sediments, distant from the then-extant shoreline and evidently well below the photic zone [66]. This regional geology-based interpretation, requiring the assemblages to be anaerobic and non-photosynthetic, is confirmed by the cobweb-like organization of their randomly interwoven, hundreds of micron-long microbial filaments, a fabric that differs greatly from the well-layered structure of photoautotroph-dominated stromatolites in which the preserved microbes are invariably much shorter, having been disrupted by wave action in their relatively shallow-water photic zone setting.
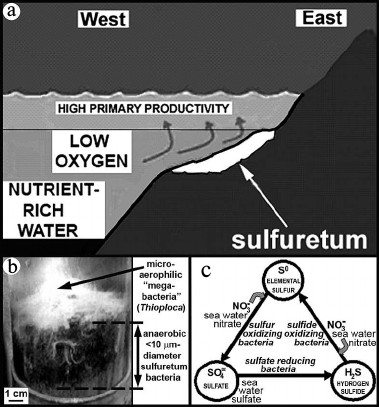
The puzzle originally presented by the off-shore deep-water anaerobic setting and cobweb-like organization of these mid-Precambrian microbial communities — decidedly different from any of the hundreds of Precambrian microbial assemblages previously reported — has been resolved. Their modern analogue, first reported in 2007 [67, 68], is the anoxic sediment-enclosed sub-surface part of extant microbial sulfuretums documented off the western coast of South America from Chile to Panama (Fig. 11a).
Fig. 11. Modern surfuretums. a) Ecological setting and (b) a core sample of a modern sulfuretum off the west coast of Chile (modified after [67, 68]). c) The metabolic sulfur cycle in the anoxic (sub-sediment) part of sulfuretums.
Modern sulfuretums, globally widespread in modern seafloor settings, are composed of an anaerobic, anoxic mud-inhabiting microbial community that is commonly overlain near and above the sediment surface by interlaced microaerophilic larger-celled “megabacteria” such as Beggiatoa and Thioploca (Fig. 11b). Because of their large (>100-µm-diameter) cell size, highly unusual for prokaryotic microbes, the megabacteria of such assemblages are relatively well known. However, the mid-Precambrian fossil assemblages, composed of <10-µm-diameter filamentous microbes essentially identical to those of the lower anoxic parts of the modern communities, do not contain fossilized megabacteria — perhaps because such large-celled microbes were of later evolutionary origin.
As shown in Fig. 11c, the anaerobic sub-sediment components of such sulfur-cycling communities require sulfate, to be biologically reduced to hydrogen sulfide, and nitrate, to oxidize the resultant H2S to elemental sulfur and, subsequently, from So to SO4. Before the GOE, neither sulfate nor nitrate was plentiful, the photochemical formation of both being inhibited a lack of atmospheric oxygen. With the increase of environmental oxygen at the GOE, however, these nutrients and H2S, produced by an increase in the abundance of sulfate reducing bacteria spurred by the GOE-promoted increased availability of sulfate, would have provided the reactants needed to power the proliferation of sulfuretums.
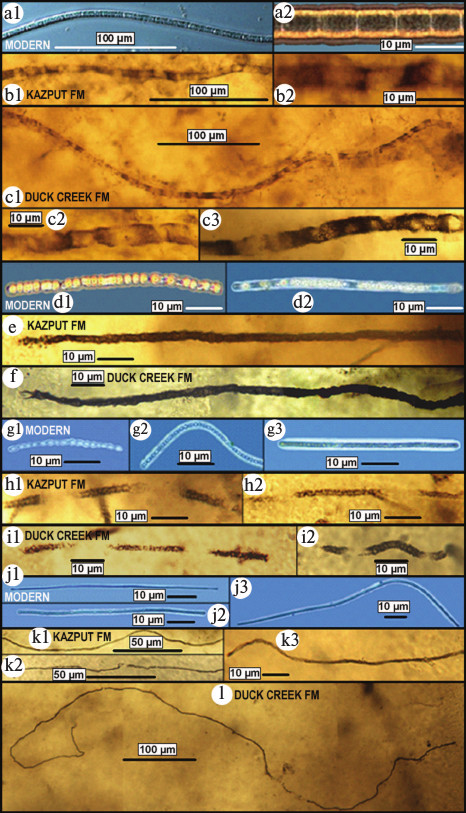
Thus, these earliest known GOE-related fossil sulfur-cycling communities provide strong support of an evolutionary response to the mid-Precambrian increase of biologically useable molecular oxygen. Their evolutionary significance, however, is greater. As shown in Fig. 12 (see color insert), each of the four major microbial components of the two fossil assemblages exhibits a one-to-one morphological correspondence to those of the sub-surface anoxic part of modern sulfuretums. In other words, these fossils indicate that not only did the microbes of the ~2300-Ma-old deposit evolve little or not at all during the five hundred million years before preservation of the ~1800-Ma-old assemblage but that such sulfuretum-comprising microbes remained essentially unchanged over the more than two thousand million years between their mid-Precambrian preservation and the present. Inhabitants of a cold quiescent, anoxic, sub-seafloor deep-water environment that has persisted since early stages of Earth history, the colorless sulfur bacteria of such communities have evidently evolved little over geological time.
Fig. 12. Comparison of microbes of modern sulfuretums off the west coast of Chile (a, d, g, j) [67, 68] with those of fossilized sulfuretums of the ~2300-Ma-old Kazput Formation (Turee Creek Group) (b, e, h, k) and ~1800-Ma-old Duck Creek Formation (c, f, h, l) of Western Australia. a-c) Broad (~8-μm-diameter) microbes having elongate cells. d-f) Medium-diameter (~4-μm-wide) microbes having bead-shaped cells. g-i) Narrow (~2-μm-diameter) microbes composed of ball-shaped cells. j-l) Very narrow (~1-μm-diameter) long microbes.
As surprising as this fossil-documented absence of evolutionary change may seem, its occurrence for Precambrian microbes is well established [30], in this instance illustrating the “null” hypothesis required of Darwinian evolution: if there is no change in the physical-biological environment of a well-adapted ecosystem there will be no evolution of the form, function and metabolic requirements of its components.
SUMMARY AND CONCLUSIONS
The time of origin of oxygen-producing photosynthesis is not resolved. Although the presence of stromatolites, cyanobacterium-like microfossils, and 12C-enriched organic matter in the earliest rock record are consistent with the presence of O2-producing photoautrophs, they are insufficient to establish its existence. It is certain that oxygen-producing photosynthesis evolved earlier (and perhaps much earlier) than the increase of atmospheric oxygen marked by the onset of the Great Oxidation Event ~2450 Ma ago, but how much earlier has yet to be determined.
Nevertheless, the biological response to the global increase of oxygen at the GOE — an event that forever altered the history of life — is beginning to come into focus, here illustrated by four examples: (1) the anaerobic to aerobic transition of metabolic and biosynthetic pathways; (2) rRNA phylogeny-correlated fossil evidence of O2-protection in immediately post-GOE-preserved cyanobacteria; (3) the earliest fossil occurrences of obligately aerobic eukaryotes; and (4) recent discoveries of mid-Precambrian anaerobic microbial sulfuretums dependent on the GOE-promoted increase of required nutrients.
The biotic response to the GOE is a question ripe for further exploration. Progress toward this goal can be expected to accrue from more detailed studies of the fossil record, as yet insufficiently mined, but also from molecular biologic and genomic studies of modern microbes — a particularly promising source of additional data. An important problem in the history of life, additional evidence of the biological impact of the GOE seems certain to be uncovered.
For the invitation to prepare this manuscript I thank A. A. Krasnovsky, Jr., V. P. Skulachev, and R. D. Ozrina, and for his generous arrangements for my participation in the symposium dedicated to the 100th anniversary of the birth of academician Alexander A. Krasnovsky, I thank M. S. Kritsky of the Russian Academy of Sciences Bach Institute of Biochemistry. I also thank V. A. Gallardo (University of Concepción, Chile) for providing the images of modern sulfuretum bacteria shown in Fig. 12 (a, d, g, and j), and J. Shen-Miller, A. K. Garcia, and A. B. Kudryavtsev for thoughtful review of the manuscript.
REFERENCES
1.Schopf, J. W. (1992) in The Proterozoic
Biosphere, a Multidisciplinary Study (Schopf, J. W., and
Klein, C., eds.) Cambridge University Press, New York, pp. 25-39.
2.Schopf, J. W. (1993) Science, 260,
640-646.
3.Schopf, J. W. (2006) Phil. Trans. Roy. Soc.
B, 361, 869-885.
4.Schopf, J. W., Kudryavtsev, A. B., Czaja, A. D.,
and Tripathi, A. B. (2007) Precam. Res., 158,
141-155.
5.Schopf, J. W., and Kudryavtsev, A. B. (2012)
Gondwana Res., 39, 761-771.
6.Mojzsis, S., Arrenhius, G., McKeegan, K. D.,
Nutman, A. P., and Friend, C. R. L. (1996) Nature, 384,
55-59.
7.McKeegan, K. D., Kudryavtsev, A. B., and Schopf, J.
W. (2007) Geology, 35, 591-594.
8.Oparin, A. I. (1938) The Origin of Life,
McMillan, New York.
9.Schopf, J. W. (1999) Cradle of Life, The
Discovery of Earth’s Earliest Fossils, Princeton University
Press, Princeton, New Jersey.
10.Blankenship, R. E. (1992) Photosynth.
Res., 33, 91-111.
11.Blankenship, R. E., and Hartman, H. (1998)
Trends Biochem. Sci., 23, 94-97.
12.Schopf, J. W. (1996) in Evolution and the
Molecular Revolution (Marshall, C. R., and Schopf, J. W., eds.)
Jones and Bartlett, Boston, pp. 73-105.
13.Darwin, C. R. (1859) On the Origin of Species
by Means of Natural Selection, John Murray, London.
14.Barghoorn, E. S., and Tyler, S. A. (1965)
Science, 147, 563-577.
15.Cloud, P. E. (1965) Science, 148,
27-45.
16.Barghoorn, E. S., and Schopf, J. W. (1965)
Science, 150, 337-339.
17.Schopf, J. W. (1968) J. Paleontol.,
42, 651-688.
18.Schopf, J. W., and Bottjer, D. J. (eds.) (2009)
World Summit on Ancient Microscopic Fossils, Special Issue,
Precam. Res., 173 (Nos. 1-4).
19.Garrels, R. M., and Mackenzie, F. T. (1971)
Evolution of Sedimentary Rocks, Norton, New York.
20.Holland, H. D. (2002) Geochim. Cosmochim.
Acta, 66, 3811-3826.
21.Canfield, D. E. (2005) Annu. Rev. Earth
Planet. Sci., 33, 1-36.
22.Farquhar, J., Bao, H., and Thiemens, M. (2000)
Science, 289, 756-759.
23.Farquhar, J., Peterson, M., Johnson, D. T.,
Strauss, H., Masterson, A., Weichert, U., and Kaufman, A. J. (2007)
Nature, 449, 706-709.
24.Hofmann, H. J. (2000) in Microbial
Sediments (Riding, R. E., and Awramik, S. M., eds.)
Springer-Verlag, Berlin, pp. 315-327.
25.Hofmann, H. J., Grey, K., Hickman, A. H., and
Thorpe, R. I. (1999) Geol. Soc. Am. Bull., 111,
1256-1262.
26.Allwood, A. C., Walter, M. R., Kamber, B. S.,
Marshall, C. P., and Burch, I. W. (2006) Nature, 441,
714-718.
27.Grotzinger, J. P., and Knoll, A. H. (1999)
Annu. Rev. Earth Planet. Sci., 27, 313-358.
28.Butterfield, N. J. (2009) Precam. Res.,
173, 201-211.
29.Schopf, J. W. (2012) in Ecology of
Cyanobacteria II, Their Diversity in Space and Time
(Whitton, B. A., ed.) Springer-Verlag, Berlin, pp. 15-36.
30.Schopf, J. W. (1994) Proc. Natl. Acad. Sci.
USA, 91, 6735-6742.
31.Klein, C., Beukes, N., and Schopf, J. W. (1987)
Precam. Res., 6, 81-94.
32.Lanier, W. P. (1986) Palaios, 1,
525-542.
33.Knoll, A. H., and Barghoorn, E. S. (1977)
Science, 198, 396-398.
34.Schopf, J. W., Kudryavtsev, A. B., Sugitani, K.,
and Walter, M. R. (2010) Precam. Res., 179, 191-205.
35.Schopf, J. W., and Packer, B. M. (1987)
Science, 237, 70-73.
36.Schopf, J. W. (1992) in The Proterozoic
Biosphere, a Multidisciplinary Study (Schopf, J. W., and
Klein, C., eds.) Cambridge University Press, New York, pp. 195-218.
37.Summons, R. E. (1992) in The Proterozoic
Biosphere, a Multidisciplinary Study (Schopf, J. W., and
Klein, C., eds.) Cambridge University Press, New York, pp. 101-115.
38.Bloeser, B. (1985) J. Paleontol.,
59, 741-765.
39.Bloeser, B., Schopf, J. W., Horodyski, R. J., and
Breed, W. J. (1977) Science, 195, 676-679.
40.Schopf, J. W. (1992) in The Proterozoic
Biosphere, a Multidisciplinary Study (Schopf, J. W., and
Klein, C., eds.) Cambridge University Press, New York, pp. 583-600.
41.Porter, S. M., and Knoll, A. H. (2000)
Paleobiology, 26, 360-385.
42.Brocks, J. J., Logan, G. A., Buick, R., and
Summons, R. E. (1999) Science, 285, 1033-1036.
43.Schopf, J. W. (1978) Sci. Amer.,
239, 110-138.
44.Summons, R. E., Bradley, A. S., Janke, L. L., and
Waldbauer, J. R. (2006) Phil. Trans. Roy. Soc. B, 361,
951-968.
45.Park, R., and Epstein, S. (1963) Geochim.
Cosmochim. Acta, 21, 110-115.
46.Hoering, T. C. (1967) in Researches in
Geochemistry (Abelson, P. H., ed.) Vol. 2, Wiley, New York, pp.
87-111.
47.Strauss, H., and Moore, T. B. (1992) in The
Proterozoic Biosphere, a Multidisciplinary Study (Schopf, J.
W., and Klein, C., eds.) Cambridge University Press, New York, pp.
709-798.
48.Hayes, J. M., Kaplan, I. R., and Wedeking, K. M.
(1983) in Earth’s Earliest Biosphere, Its Origin and
Evolution (Schopf, J. W., ed.) Princeton University Press,
Princeton, NJ, pp. 93-134.
49.Hayes, J. M., DesMarais, D. J., Lambert, I. A.,
Strauss, H., and Summons, R. E. (1992) in The Proterozoic
Biosphere, a Multidisciplinary Study (Schopf, J. W., and
Klein, C., eds.) Cambridge University Press, New York, pp. 81-134.
50.House, C. H., Schopf, J. W., McKeegan, K. D.,
Coath, C. D., Harrison, T. M., and Stetter, K. O. (2000)
Geology, 28, 707-710.
51.House, C. H., Schopf, J. W., and Stetter, K. O.
(2003) Org. Geochem., 34, 345-356.
52.Hayes, J. M. (1983) in Earth’s Earliest
Biosphere, Its Origin and Evolution (Schopf, J. W., ed.)
Princeton University Press, Princeton, NJ, pp. 291-301.
53.Schopf, J. W. (1994) Early Life on Earth
(Bengtson, S., ed.) Columbia University Press, New York, pp.
193-206.
54.Holland, H. D. (2009) Geochim. Cosmochim.
Acta, 73, 5241-5255.
55.Fawley, M. W. (1991) J. Phycol.,
27, 544-548.
56.Graham, J. E., and Bryant, D. A. (2009)
Bacteriology, 2009, 3292-3300.
57.Takaichi, S. (2011) Marine Drugs,
9, 1101-1118.
58.Giovannoni, S. J., Turner, S., Olsen, G. J.,
Barns, S., Lane, D. J., and Pace, N. R. (1988) J. Bacteriol.,
170, 3584-3592.
59.Schopf, J. W., Kudryavtsev, A. B., and Sergeev,
V. N. (2010) J. Paleol., 84, 402-416.
60.Tomitani, A., Knoll, A. H., Cavanaugh, C. M., and
Ohno, T. (2006) Proc. Natl. Acad. Sci. USA, 103,
5442-5447.
61.Lamb, D. M., Awramik, S. M., Chapman, D. J., and
Zhu, S. (2009) Precam. Res., 173, 93-104.
62.Sharma, M., and Shukla, Y. (2009) Precam.
Res., 173, 105-122.
63.Han, T., and Runnegar, B. (1992) Science,
257, 232-235.
64.Van Kranendonk, M. J., Schopf, J. W., Grice, K.,
Walter, M. R., Pages, A., Kudryavtsev, A. B., Gallardo, V. A.,
Espinoza, C., Melendez, I., and Lepland, A. (2012) Astrobiology
Science Conference 2012, Atlanta, Georgia, USA, Abstract #2084,
available online at: http://abscicon2012.arc.nasa.gov/abstracts.
65.Hofmann, H. J., and Schopf, J. W. (1983) in
Earth’s Earliest Biosphere, Its Origin and
Evolution (Schopf, J. W., ed.) Princeton University Press,
Princeton, NJ, pp. 321-360.
66.Wilson, J. P., Fischer, W. W., Johnston, D. T.,
Knoll, A. H., Grotzinger, J. P., Walter, M. R., McNaughton, N. J.,
Simon, M., Abelson, J., Schrag, D. P., Summons, R., Allwood, A.,
Andres, M., Gammon, C., Garvi, J., Sky, R., Schweizer, M., and Watters,
W. A. (2010) Precam. Res., 179, 135-149.
67.Gallardo, V. A., and Espinoza, C. (2007) Int.
Microbiol., 10, 97-102.
68.Gallardo, V. A., and Espinoza, C. (2007) in
Instruments, Methods, and Missions for Astrobiology
X (Hoover, R. B., Levin, G. V., Rozanov, A. Y., and Davies, P. C.,
eds.) Proc. SPIE 66941, pp. 66941H-1–66941H-11.