REVIEW: Long-Wavelength Chlorophylls in Photosystem I of Cyanobacteria: Origin, Localization, and Functions
N. V. Karapetyan1*, Yu. V. Bolychevtseva1, N. P. Yurina1, I. V. Terekhova1, V. V. Shubin1, and M. Brecht2,3
1Bach Institute of Biochemistry, Russian Academy of Sciences, Leninsky pr. 33, 119071 Moscow, Russia; fax: (495) 954-2732; E-mail: nkarap@inbi.ras.ru; bolychev@inbi.ras.ru; terekhova@inbi.ras.ru2University Tubingen, Institute of Physical and Theoretical Chemistry (IPTC), Auf der Morgenstelle 18, 72076 Tubingen, Germany; fax: +49 (7071) 295-490; E-mail: marc.brecht@uni-tuebingen.de
3Zurich University of Applied Sciences, Institute of Applied Mathematics and Physics, Technikumstrasse 13, 8401 Winterthur, Switzerland; fax: +41 (58) 9357-3062; E-mail: bret@zhaw.ch
* To whom correspondence should be addressed.
Received November 7, 2013; Revision received November 28, 2013
The structural organization of photosystem I (PSI) complexes in cyanobacteria and the origin of the PSI antenna long-wavelength chlorophylls and their role in energy migration, charge separation, and dissipation of excess absorbed energy are discussed. The PSI complex in cyanobacterial membranes is organized preferentially as a trimer with the core antenna enriched with long-wavelength chlorophylls. The contents of long-wavelength chlorophylls and their spectral characteristics in PSI trimers and monomers are species-specific. Chlorophyll aggregates in PSI antenna are potential candidates for the role of the long-wavelength chlorophylls. The red-most chlorophylls in PSI trimers of the cyanobacteria Arthrospira platensis and Thermosynechococcus elongatus can be formed as a result of interaction of pigments peripherally localized on different monomeric complexes within the PSI trimers. Long-wavelength chlorophylls affect weakly energy equilibration within the heterogeneous PSI antenna, but they significantly delay energy trapping by P700. When the reaction center is open, energy absorbed by long-wavelength chlorophylls migrates to P700 at physiological temperatures, causing its oxidation. When the PSI reaction center is closed, the P700 cation radical or P700 triplet state (depending on the P700 redox state and the PSI acceptor side cofactors) efficiently quench the fluorescence of the long-wavelength chlorophylls of PSI and thus protect the complex against photodestruction.
KEY WORDS: antenna, long-wavelength chlorophyll, P700, reaction center, fluorescence quenching, photosystem I, cyanobacteriaDOI: 10.1134/S0006297914030067
Abbreviations: LWC, long-wavelength chlorophyll; P700 (P700+), the primary electron donor of photosystem I in the reduced (oxidized) state; PSI, photosystem I.
Photosystem I (PSI) of higher plants, algae, and cyanobacteria is a
membrane complex responsible for light energy transformation into
chemical energy and for the reduction of ferredoxin on the stromal side
and oxidation of plastocyanin (or cytochrome c6) on
the luminal side of thylakoids. The structure of PSI in
photosynthesizing organisms is conservative; however, there are
differences in subunit composition and spectral characteristics of the
antenna [1-3]. The PSI complex
in the membrane of cyanobacteria is organized preferentially as a
trimer [4-7], whereas the PSI
in plants is a monomeric complex encircled by four light-harvesting
complexes LHC1 [3, 8, 9]. Due to LHC1, the PSI antenna in plants contains a
significantly higher number of chlorophyll molecules per P700 than in
PSI in cyanobacteria. As significantly differentiated from higher
plants, cyanobacteria do not have light-harvesting complexes LHC1 and
LHC2, rather they have chlorophyll localized only in the core-complexes
PSI and PSII, as well as a higher content of PSI complexes than PSII in
their thylakoids. In higher plants, the PSI/PSII ratio is 1, whereas in
the unicellular cyanobacteria it is 3, and in the cyanobacterium
Arthrospira (Spirulina) platensis it reaches 5.5
[10]. It is supposed that in cyanobacteria the
greater part of P700 is involved in cyclic electron transfer around the
PSI.
The PSI of higher plants, algae, and cyanobacteria is uniquely characterized by a comparatively high content within the antenna of so-called long-wavelength chlorophylls (LWCs), also known as red or low-energy chlorophylls [11-14]. These chlorophylls absorb at longer wavelengths than P700 (the primary electron donor of PSI). The presence of LWCs within the PSI complexes of different photosynthesizing organisms suggests their physiological importance. LWCs were first detected by Litvin and Krasnovsky in kidney bean leaves at 77 K by the fluorescence emission band at 730 nm [15] and by Brody in the algae Chlorella and Porphyridium by the fluorescence band at 720 nm [16]. Concentrated chlorophyll solution at 77 K has fluorescence with maximum at 715 nm; therefore, it was supposed that chlorophyll aggregates could be responsible for the long-wavelength emission of the PSI chlorophylls [17-19].
For a long time the origin of the PSI long-wavelength chlorophylls was unclear. Based on finding the molecular organization of the PSI trimer in the thermophilic cyanobacterium Thermosynechococcus elongatus at the resolution of 2.5 Å, it was supposed that chlorophyll aggregates (trimers and dimers) within the PSI antenna could be candidates for the LWCs [2]. Picosecond spectroscopy of PSI complexes revealed mechanisms of energy migration within the antenna and of light energy transformation by PSI reaction centers, and LWCs were supposed to play a role in PSI [20]. The structure and functioning of PSI are considered in many reviews [1, 7, 14, 21-26]. In the present review, we consider differences in the structural organization of the PSI complexes in cyanobacteria and plants, the nature of LWCs within the cyanobacterial PSI antenna, and their role in energy migration within the antenna, in charge separation in the reaction center, and in the photoprotective dissipation of excess energy.
STRUCTURAL ORGANIZATION OF PSI COMPLEX IN CYANOBACTERIA
The PSI complex in thylakoids of cyanobacteria is organized preferentially as a trimer as evidenced by the similarity of fluorescence spectra of the cyanobacterium Arthrospira (Spirulina) platensis and the PSI trimers isolated from it [6]. As differentiated from the monomers, the PSI trimers are characterized by low-temperature fluorescence (77 K) with maximum at 760 nm (F760). The X-ray analysis of a crystal of the PSI trimer from the thermophilic cyanobacterium Th. elongatus at the resolution of 2.5 Å has indicated the localization of 96 molecules of chlorophyll a, 22 molecules of β-carotene, and four molecules of lipids, and it also has shown more accurately the orientation of polypeptides and cofactors of the reaction center [2, 26]. The molecular weight of the PSI complex of Th. elongatus is about 1000 kDa, and every monomeric complex in the isolated PSI trimer contains 96 chlorophyll molecules. The PSI trimers of cyanobacteria are disk-shaped with 210 Å diameter and 90 Å thickness. Each monomer in the trimer is an ellipsoid with 130 Å length and 105 Å width and contains 12 protein subunits and 127 cofactors [27]. The monomeric complex in the PSI trimer includes subunits PsaA/PsaB associated with the majority of the antenna chlorophylls, the reaction center cofactors, and also small subunits PsaC, PsaD, PsaE, PsaF, PsaI, PsaJ, PsaK, PsaL, PsaM, and PsaX. Iron-sulfur clusters FA/FB are localized on the stromal polypeptide PsaC. In the luminal part of the complex, plastocyanin or type c cytochrome are bound with polypeptide PsaF. Polypeptides PsaL and PsaI are important for generation of the PSI complex trimer in cyanobacteria [2, 28]; PsaL- and PsaI-deficient mutants are unable to form the PSI trimers [29].
The primary electron donor of the reaction center (P700) is a heterodimer of chlorophyll a and its epimer, eC-A1 and eC-B1 [2]. The distance between the centers of these chlorophylls within P700 is 6.3 Å. At the distance of ~12 Å from P700, a pair of chlorophyll a molecules, eC-A2 and eC-B2, is situated; the third pair of monomeric chlorophylls eC-A3 and eC-B3, which acts as the primary electron acceptor A0, is at the distance of 20-21 Å from the P700 (the distances are indicated between the centers of the molecules). The antenna chlorophylls interact with P700 through the linker chlorophylls B39 and A40 situated at the distances of 13 and 15 Å from the A0 molecules [30]. Secondary acceptors of the electron A1 are constituted by phylloquinones QK, which pass the electron to the iron-sulfur center FX localized near the stromal surface of polypeptide PsaA/PsaB. Six molecules of chlorophyll a and two phylloquinone molecules form two parallel branches of the electron transfer in the PSI reaction center [2].
ORGANIZATION OF PSI ANTENNA IN CYANOBACTERIA
PSI of higher plants and cyanobacteria is characterized by a close relation of the antenna chlorophylls with P700 [31-34], and therefore the PSI reaction center cannot be isolated without the antenna chlorophylls. From 96 chlorophylls of the PSI complex in the cyanobacterium Th. elongatus, 90 molecules are localized within the antenna [2]: 79 molecules are on the polypeptide PsaA/PsaB, 10 molecules are associated with subunits PsaG, PsaL, PsaM, PsaK, and PsaX, and one molecule is bound with phosphatidylglycerol. Distances between nearly all antenna chlorophylls are 7-16 Å, which is favorable for rapid migration of the excitation energy among them [2]. Every monomeric complex of the PSI trimer in Th. elongatus contains 22 molecules of β-carotene associated with polypeptides PsaA/PsaB; however, no carotene molecule is close to P700. Sixty of 90 chlorophylls are in immediate contact with carotenoids, which promotes the quenching of chlorophyll triplets. The majority of β-carotenes are localized near the LWCs [2], which allows carotenoids to perform the protective function. Carotenoids also play a structural role in thylakoid membranes [35] and are involved in the interaction of peripheral subunits with the heterodimer PsaA/PsaB and in trimerization of the PSI monomeric complexes [2].
As mentioned above, the PSI antenna of different cyanobacteria contains 5-10% of LWCs with absorption in the region of 710-750 nm [7]. The PSI complexes of different cyanobacteria are organized similarly and differ mainly in the contents and spectral characteristics of the LWCs. Similarly to P700, the LWCs act as a terminal trap of energy and therefore can significantly determine the specificity of energy migration within the antenna and its being trapped by the reaction center. These chlorophylls increase energy trapping and decrease the quantum yield of the charge separation. However, the decrease in the quantum yield of the photochemical reaction is compensated by an increase in the absorption cross section and also by an effective uphill energy transfer from LWC onto the bulk chlorophyll. Depending on their localization, LWCs decrease the number of energy transfers between bulk chlorophylls and thus promote energy concentration on the reaction center [36], increase the absorption of long-wavelength light under conditions of cyanobacterial cultivation at low light intensities [13, 37], or protect the PSI reaction center against energy excess when the reaction center is inactive [38-41].
Spectral characteristics of long-wavelength forms. The antennas of the PSI complexes in the cyanobacteria Synechocystis PCC 6803, Th. elongatus, and A. platensis contain different numbers of LWCs (Table 1) with species-specific spectral characteristics [14, 23, 42]; the PSI trimers of the cyanobacterium Gloeobacter violaceus do not have LWCs at all [43]. Both bulk chlorophylls and LWCs are spectrally heterogeneous. The low-temperature absorption spectra of the PSI complexes of cyanobacteria were decomposed into bands to evaluate the contents of chlorophyll molecules in each long-wavelength form. The PSI trimers and monomers of Synechocystis PCC 6803 are supposed to be characterized by a pigment form with absorption maximum at 708 nm (Chl708): the PSI trimers contain 3-4 LWCs and the monomers have 30% less [44]. The PSI monomers of Synechocystis contain only Chl708, and the trimers contain the forms Chl706 and Chl714, which are chlorophyll dimers [45, 46]. The PSI trimers and monomers of Th. elongatus contain the same forms of LWCs, Chl708 and Ch719 (Table 1). In the trimers and monomers, Chl708 includes five chlorophyll molecules, whereas Chl719 in the trimers contains four chlorophyll molecules and in the monomers only two molecules [47]. The decomposition of the absorption spectrum of the A. platensis PSI trimers at 6 K revealed Chl708 and Chl740 forms that contained seven and three chlorophyll molecules, respectively, whereas the monomeric complex contained only seven chlorophyll molecules [48]. An appearance of the absorption band at 740 nm in the A. platensis PSI trimers is accompanied by a decrease in the absorption band at 710-712 nm [49]. The half-width of the absorption and fluorescence bands of the red-most forms of the A. platensis PSI trimers is 20-25 nm [11, 23].
Table 1. Spectral characteristics of LWCs in
trimeric and monomeric PSI complexes of cyanobacteria A.
platensis and Th. elongatus
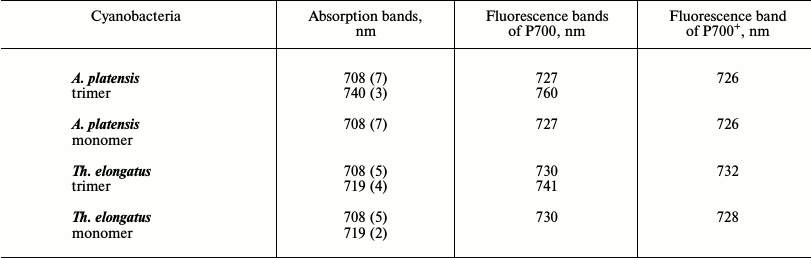
Note: Positions of the bands are indicated for 5 K; the number of
chlorophyll molecules forming the LWCs is presented in parentheses [23].
The LWCs significantly influence the PSI fluorescence spectrum at room temperature. In the PSI complexes of Synechocystis containing two LWCs, the major maximum in the fluorescence spectrum is at 685 nm (F685) with a shoulder at 712 nm [50]. In the A. platensis PSI trimers, the main fluorescence is characterized by a band at 730-735 nm that is more intense than the F685 band [48]. Differences in fluorescence of the PSI from different cyanobacteria are especially evident at low temperatures. Decrease in temperature significantly increases the quantum yield of fluorescence of the long-wavelength forms of PSI chlorophyll because the greater part of the excitation energy unused by the reaction center is trapped by the LWCs [51, 52]. The major emission peak of Synechocystis is at 720-724 nm [44, 53, 54], whereas for the Th. elongatus trimers the peak is at 730 nm [48]. In the A. platensis PSI trimers at 77 K, in addition to a peak at 685 nm, there are also emission bands F730 and F760; when P700 and the PSI acceptor cofactors are reduced before the measurement, F760 is more than an order of magnitude more intense than F730 [48, 49].
Nature of long-wavelength chlorophyll forms and their possible localization. LWCs with absorption at wavelengths longer than 700 nm were observed also in concentrated solutions and films [18, 19]. Chlorophyll films prepared by evaporation of an alcoholic extract displayed spectral characteristics similar to those of intracellular chlorophyll. The fluorescence yield of chlorophyll films, similarly to that of chlorophylls in vivo, is strongly quenched. Keeping chlorophyll films in water vapors resulted in an additional red shift of the emission to 740 nm, which suggests a role of water molecules in the shift. The localization of water molecules near chlorophyll dimers and trimers within the PSI antenna [2] can be of help in determining the possible role of water in the generation of LWCs. Two types of chlorophyll aggregations in films have been supposed: 1) direct chlorophyll–chlorophyll contacts via the interaction of keto groups and magnesium; 2) coordination interactions of keto groups with magnesium of another molecule leading to generation of aggregates with different optical and magnetic characteristics [19, 55].
The long-wavelength fluorescence at 730-735 nm of the A. platensis PSI trimers at physiological temperatures with intensity higher than that of F685 indicates that these forms are not results of the temperature decrease [49]. The presence of LWCs at physiological temperatures has been shown also for other cyanobacteria [56, 57]. Consequently, LWC that are recorded more distinctly at low temperatures are not a result of the temperature decrease. The long-wavelength absorption shift of these forms is ascribed to chlorophyll aggregates. The generation of the PSI trimer is accompanied by changes in the state of the antenna chlorophylls localized on the periphery of the monomeric complex [58]. Chlorophyll aggregates appear on pigment–pigment or pigment–protein interactions. The absence of long-wavelength bands in the light-induced circular dichroism spectrum indicates that the LWCs are localized at a great distance from P700 [33, 34]. Electric field influences differently spectral forms of LWCs [59]. According to polarization spectra of the A. platensis PSI trimer measured at 1.4 K, chlorophylls in Chl740 are polarized similarly, whereas the seven chlorophylls included into three different aggregates of the PSI antenna and forming Chl708 are polarized differently [60]. Reconstruction of the A. platensis PSI trimer on incubation in liposomes of PSI monomeric complexes from A. platensis has shown that Chl740 is produced as a result of pigment–pigment interaction of chlorophylls localized on different monomeric complexes of the trimer and confirmed the peripheral localization of LWCs [58]. The generation of dimers from chlorophylls localized on different monomeric complexes in the PSI trimer has been detected also in the Th. elongatus PSI trimer [61].
Although both spectral characteristics of LWCs and their number are known, as well as the position of chlorophyll aggregates within the PSI complex of Th. elongatus, it is still difficult to judge unambiguously about the localization of these spectral forms of chlorophyll within the antenna. Linker chlorophylls A40 and B39 (using the terms of Jordan et al. [2]) or closely localized Chl708 dimers [62-64] capable of focusing the excitation energy on P700 are supposed to act as LWCs. However, as noted above, the light-induced circular dichroism spectrum of the A. platensis PSI does not confirm exciton interaction of P700 and LWCs. The PSI antenna of the cyanobacterium Th. elongatus contains many chlorophyll aggregates, but only one trimer and three dimers can be considered as candidates for LWCs [2, 30].
There was an attempt to localize spectral forms of LWC within the Th. elongatus PSI antenna. The trimer B31/B32/B33 on the luminal side of the complex and the dimers A38/A39 and B37/B38 (termed according to Jordan et al. [2]) were considered as LWCs. Based on the totality of spectral data, it was supposed that Chl708 of the Th. elongatus PSI should be a chlorophyll trimer B31/B32/B33 and Chl719 a dimer B37/B38 [65]. Because the Chl708 form in the PSI trimers and monomers includes five chlorophyll molecules (Table 1), it is reasonable to suppose that a chlorophyll dimer with absorption at 708 nm should exist. Because Chl708 in the trimer and monomer of the A. platensis PSI contains seven chlorophyll molecules, it is supposed that the PSI antenna of this cyanobacterium should contain one more chlorophyll dimer in addition to the chlorophyll trimer and dimer in the Th. elongatus PSI. It is more difficult to localize within the antenna the red-most chlorophylls: Chl719 in Th. elongatus PSI and Chl740 in A. platensis PSI. Although the antenna of PSI in cyanobacteria consists of spectrally different LWCs, it can be supposed that the generation of the red-most forms should be the same for PSI of both of these cyanobacteria. These LWCs are present only in trimeric complexes, and they therefore can be generated on trimerization as a result of pigment–pigment interaction of chlorophyll molecules localized on different monomeric complexes. We have shown this possibility in the case of Chl740 in A. platensis PSI containing three chlorophyll molecules [38, 58]. It seems that Chl740 is generated as a result of the interaction of peripherally localized chlorophyll dimer from one monomeric complex with chlorophyll of another monomeric complex. And both the chlorophyll dimer and monomer in each monomeric complex are localized on opposite sides of the complex, which can be established only upon the determination of the molecular organization of the A. platensis PSI trimer. The Th. elongatus PSI trimer contains four chlorophyll molecules in Chl719, and the monomer contains only two chlorophylls in Chl719 (Table 1). Consequently, another Chl719 dimer is generated on interaction of peripherally localized chlorophylls on trimerization of monomeric complexes, similarly to Chl740 in A. platensis.
Dependence of fluorescence yield of PSI long-wavelength chlorophylls on P700 redox-state. We have detected a changing fluorescence of PSI at 295 K: a light-induced increase in the fluorescence yield of isolated complex of plant PSI was observed on accumulation of both P700+ [66, 67] and reduced P700 and cofactors of the PSI acceptor part [68]. The accumulation of P700+ at 77 K leads not to an increase in fluorescence but to its quenching, which has been confirmed for PSI of cyanobacteria: the quenching effect of P700+ increases with decrease in temperature [48].
The fluorescence quantum yield of PSI chlorophylls is low at room temperature, whereas the quantum yield of charge separation is above 95% [36]. The intensity of F760 at 77 K of the PSI trimers of the cyanobacterium A. platensis strongly depends on the redox-state of P700: it is maximal in the reduced state and is more than an order quenched at the oxidized state. Similar kinetics of P700+ regeneration and F760 fluorescence quenching at 77 K indicates that just P700+ is the quencher [48]. In plants the fluorescence yield at 735 nm (77 K) does not depend on the redox-state of P700, which is due to the greater distance (about 60 Å) between P700 and LWC molecules in the LHC1 complexes [69]. PSI long-wavelength chlorophylls in plants are localized mainly in polypeptides Lhca1/Lhca4 [70], and the energy absorbed by them can oxidize P700 at physiological temperatures [13, 71].
MIGRATION OF EXCITATION ENERGY AND ENERGY DISSIPATION IN PSI OF
CYANOBACTERIA
The highly efficient energy migration within the antenna and its transformation in PSI reaction centers are due to a particular spatial organization of the antenna and conjunction of the reaction center chlorophyll with the antenna and the primary acceptor [2, 30, 33, 36]. Energy absorbed by bulk chlorophylls migrates to P700 with nearly 100% efficiency [36], whereas in the A. platensis PSI trimers the energy migration efficiency from bulk chlorophylls to LWCs depends on the redox-state of the reaction center; it is 30% when P700 and the PSI acceptors are reduced, and it is much higher when P700 is oxidized [48].
The sequence of excitation energy migration between chlorophyll pools within the PSI antenna was evaluated by the measurement of fluorescence spectra by kinetics of fluorescence intensity decrease [20] or quenching [72] in the subpicosecond time interval. The studies on these spectra in picosecond and femtosecond intervals at 295 K did not reveal a dependence of the excitation energy migration kinetics on the redox-state of P700 [20, 49]. We were the first to resolve in these spectra processes of energy migration between different pools of long-wavelength chlorophylls for the A. platensis PSI trimers due to narrowing of the chlorophyll emission bands at 77 K and slowing of energy migration and its trapping by the reaction center [68, 73]. Later, energy migration between different pools of long-wavelength chlorophyll was detected at room temperature using short (30-60 fs) light pulses [20, 52].
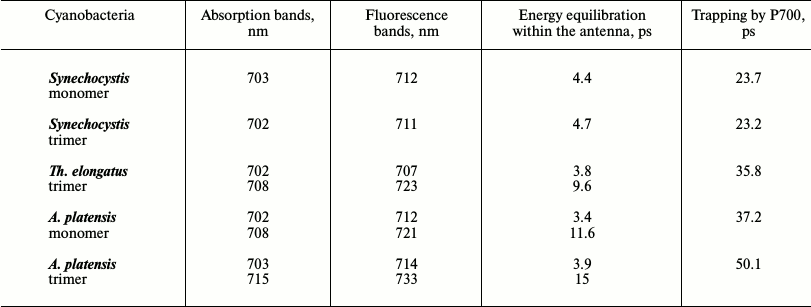
Correlation of the number and spectral characteristics of LWCs with the excitation energy dynamics can give information about their functional role. According to Table 2 (measurements were performed at room temperature), the Synechocystis PSI trimers and monomers, which contain LWCs with absorption at 702-703 nm, have 4.4-4.7 ps and approximately 23.5 ps components that are caused by energy equilibration and energy trapping by P700, respectively. In the case of the Th. elongatus PSI trimer with the antenna containing LWCs with absorption at 702 and 708 nm, there are 9.6 and 35.8 ps components reflecting the same processes. In the A. platensis PSI trimers containing LWCs with absorption at 703 and 715 nm, the rate characteristics are slower, 15 and 50.1 ps, respectively. Consequently, LWCs poorly influence the rate of establishment of equilibrium in the heterogeneous antenna but significantly slow energy trapping by the reaction center [20].
Table 2. Influence of LWCs on energy
equilibration in the antenna and on energy trapping by P700 in
cyanobacterial PSI trimers and monomers at 293 K [20]
For effective transfer of excitation energy to the reaction center, it is sufficient to have a small overlapping between the emission bands of long-wavelength pigments and the absorption of the primary electron donor. The fluorescence of the A. platensis PSI trimers (F760) is effectively quenched by P700+ because the Forster integral overlap between the bands of long-wavelength fluorescence F760 and P700+ absorption in the PSI trimers is 150 times higher than it is for P700 [68]. The distance between P700 and the long-wavelength chlorophyll form characterized by the emission maximum at 760 nm is 32 Å [48]. Values of the integral overlap between the band F730 and P700/P700+ absorption bands of the A. platensis PSI trimer are approximately the same and close to the values for the PSI monomeric complexes of higher plants and cyanobacteria. Although the long-wavelength chlorophyll form with absorption maximum at 735 nm is peripherally localized on PsaA/PsaB, its remoteness from P700 is fully compensated by the high value of the overlap interval, which at 77 K provides rapid (~150 ps) transfer of the excitation energy from this chlorophyll form to P700+ [69].
The excitation energy unused in the primary photoact is emitted as fluorescence or dissipates as heat; a part of the energy is spent for transition of the antenna chlorophylls or P700 to the triplet state. The excess energy dissipation is necessary for preventing photodestruction of the complex. The efficiency of photosynthesis is determined not only by activity of the electron transfer processes, but also by stability of the photosynthetic apparatus. It seems that during the outset of life primary heterotrophs had to develop a corresponding strategy to protect against extreme ultraviolet and intense sunlight.
Because of compact packing of the reaction centers in the photosynthetic membrane, a question always arose of whether electronic excitation exchange is possible between the antennas of the complexes. Excitation energy migration from a complex with closed center to a complex with active center in the photosystem of purple bacteria and in PSII of higher plants provides for its use in the primary photoact. However, no such interaction has been detected in the PSI trimers of cyanobacteria at 295 K, which is indicated by the linear dependence between P700 photooxidation rate and the P700+ concentration [13]. The peripheral localization of Chl740 makes such exchange possible in the A. platensis PSI trimer [38], which is indicated by the hyperbolic dependence of the fluorescence quenching rate at 760 nm on the relative content of P700+ (P700+/(P700 + P700+)). The excitation energy exchange between molecules of antenna chlorophyll of monomers in PSI trimer promotes emission-caused deactivation of the excess energy of the electronic excitation and thus protects the complex against photodestruction. Oxidized P700 in the PSI trimer of the cyanobacterium A. platensis is an effective quencher of fluorescence at 760 nm. If P700 of a monomeric complex is in the oxidized state, the excitation energy from the antenna of the complex with P700 migrates through the long-wavelength chlorophyll Chl740 to the antenna of the complex with P700+ and is efficiently quenched [38]. Intense illumination leads to bleaching of the chlorophyll form with absorption maximum at 709 nm [74, 75]; the antenna of the PSI trimers is much more resistant to photodestruction than the antenna of the monomers [14]. Compared to bulk chlorophyll molecules, the P700 molecule is exclusively stable in both trimers and monomers of PSI [76], which can determine its photoprotective effect against excess absorbed energy.
If the PSI reaction center is closed (P+A1 or PA1–), chlorophylls of the antenna or P700 become triplets under intense illumination [77, 78]. When on the oxidized P700 the primary photochemistry is blocked, intense illumination of Th. elongatus PSI trimers leads to generation of antenna triplet chlorophyll, which is recorded by an increase in the absorption at 830 nm [78]. The dissipation of the chlorophyll triplet is accompanied by generation of a triplet of carotenoids (absorption at 532 nm increases). Identical kinetics of chlorophyll triplet decay and generation of the carotenoid triplet suggest direct triplet–triplet exchange. When P700 and the PSI acceptors (A1, FX, and FA/FB) are preliminarily reduced, at 5 K in the light charges are separated in the reaction center between P700 and A0, and the recombination of charges generates the P700 triplet state [78, 79]. The “triplet-minus-singlet” spectrum of Th. elongatus PSI trimers is similar to the photooxidized P700 spectrum: bleaching at 701 nm is caused by the disappearance of the P700 energy-poor exciton band, whereas bands at 687, 674, and 683 nm can reflect the loss of excitonic interaction with the neighbor chlorophylls on arising of the P700 triplet [33, 34]. Generation of P700 triplet with lifetime of 1.1 ms was found also for A. platensis PSI trimers (5 K). The similarity of the P700 decay kinetics and the quenching of the F760 fluorescence indicates that just the P700 triplet is responsible for quenching of the long-wavelength fluorescence [48].
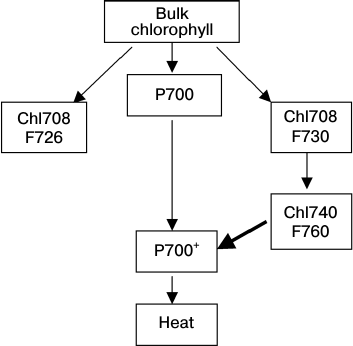
According to the kinetic theory of energy migration in the A. platensis PSI trimers (see Scheme), the energy from bulk chlorophyll to LWCs characterized by the fluorescence F726 and F730 is transferred in parallel. The F726 signal can be recorded even at 1.4 K, which evidences the localization of LWC with such fluorescence far from Chl740 or its difference in polarization [60]. LWCs with F730 can pass the excitation energy to Chl740 (F760). The cation radical P700 effectively quenches F760 but can also quench F730 of the PSI monomer with significantly lower efficiency [80]. Pathways of excitation energy transfer between LWC molecules and then to P700 can be identical with both oxidized and reduced P700, but they will be different in the transfer rates.
Scheme of energy migration between LWCs within the antenna of the
cyanobacterium A. platensis PSI trimer; chlorophyll forms are
designated by absorption band maximums at 5 K (Chl708, Chl740) and by
fluorescence maximums at 77 K (F760, F730); the bold arrow indicates
the F760 quenching by P700+
This work was supported by the program “Molecular and Cell Biology” of the Russian Academy of Sciences Presidium and by the Russian Foundation for Basic Research (projects 11-04-00247a and 14-04-00148a).
REFERENCES
1.Golbeck, J. H., and Bryant, D. A. (1991) Curr.
Top. Bioenerg., 16, 83-177.
2.Jordan, P., Fromme, P., Klukas, O., Witt, H. T.,
Saenger, W., and Krauss, N. (2001) Nature, 411,
909-917.
3.Ben-Shem, A., Frolov, F., and Nelson, N. (2003)
Science, 426, 630-635.
4.Boekema, E. J., Dekker, J. P., van Heel, M. G.,
Roegner, M., Saenger, W., Witt, I., and Witt, H. T. (1987) FEBS
Lett., 217, 283-286.
5.Roegner, M., Muehlenhoff, U., Boekema, E. J., and
Witt, H. T. (1990) Biochim. Biophys. Acta, 1015,
415-424.
6.Shubin, V. V., Tsuprun, V. L., Bezsmertnaya, I. N.,
and Karapetyan, N. V. (1993) FEBS Lett., 334, 79-82.
7.Karapetyan, N. V., Holzwarth, A. R., and Roegner,
M. (1999) FEBS Lett., 460, 395-400.
8.Boekema, E. J., Jensen, P. E., Schlodder, E., van
Breemen, J. F. L., van Roon, H., Scheller, H. V., and Dekker, J. P.
(2001) Biochemistry, 40, 1029-1036.
9.Croce, R., and van Amerongen, H. (2013)
Photosynth. Res., 116, 153-166.
10.Rakhimberdieva, M. G., Boichenko, V. A.,
Karapetyan, N. V., and Stadnichuk, I. N. (2001) Biochemistry,
40, 15780-15788.
11.Shubin, V. V., Murthy, S. D. S., Karapetyan, N.
V., and Mohanty, P. (1991) Biochim. Biophys. Acta, 1060,
28-36.
12.Shubin, V. V., Bezsmertnaya, I. N., and
Karapetyan, N. V. (1992) FEBS Lett., 309, 340-342.
13.Shubin, V. V., Bezsmertnaya, I. N., and
Karapetyan, N. V. (1995) J. Photochem. Photobiol., 30B,
153-160.
14.Karapetyan, N. V. (1998) Biol. Membr.
(Moscow), 15, 461-471.
15.Litvin, F. F., and Krasnovsky, A. A. (1957)
Dokl. AN SSSR, 117, 106-109.
16.Brody, S. S. (1958) Science, 128,
838-839.
17.Krasnovsky, A. A. (1960) Ann. Rev. Plant
Physiol., 11, 363-410.
18.Krasnovsky, A. A. (1992) Photosynth. Res.,
33, 177-193.
19.Katz, J. J., Bowman, M. K., Michalski, T. J., and
Worchester, D. L. (1991) in Chlorophylls (Scheer, H., ed.) CRC
Press, Boca Press, pp. 211-235.
20.Gobets, B., van Stokkum, I. H. M., Roegner, M.,
Kruip, J., Schlodder, E., Karapetyan, N. V., Dekker, J. P., and van
Grondelle, R. (2001) Biophys. J., 81, 407-424.
21.Gobets, B., and van Grondelle, R. (2001)
Biochim. Biophys. Acta, 1507, 80-99.
22.Brettel, K. (1997) Biochim. Biophys. Acta,
1318, 322-373.
23.Karapetyan, N. V., Schlodder, E., van Grondelle,
R., and Dekker, J. P. (2006) in Photosystem I: The Light-Driven
Plastocyanine: Ferredoxin Oxidoreductase (Golbeck, J. H., ed.) in
Series Advances in Photosynthesis and Respiration, Springer,
Dordrecht, Vol. 24, pp. 177-192.
24.Melkozernov, A. N. (2001) Photosynth.
Res., 70, 129-153.
25.Nelson, N., and Yokum, C. F. (2006) Ann. Rev.
Plant Biol., 57, 521-565.
26.Fromme, P., Jordan, P., and Krauss, N. (2001)
Biochim. Biophys. Acta, 1507, 5-31.
27.Schubert, W.-D., Klukas, O., Krauss, N., Saenger,
W., Fromme, P., and Witt, H. T. (1997) J. Mol. Biol.,
272, 741-769.
28.Krauss, N., Schubert, W.-D., Klukas, O., Fromme,
P., Witt, Y. T., and Saenger, W. (1996) Nature Struct. Biol.,
3, 965-973.
29.Jekow, P., Schubert, W.-D., Fromme, P., Kruip,
J., Chitnis, P. R., Roegner, M., and Saenger, W. (1996)
Naturforsch., 51B, 195-199.
30.Byrdin, M., Jordan, P., Krauss, N., Fromme, P.,
Stehlik, D., and Schlodder, E. (2002) Biophys. J., 83,
433-457.
31.Shubin, V. V., Karapetyan, N. V., and Krasnovsky,
A. A. (1986) Photosynth. Res., 9, 3-12.
32.Shubin, V. V., and Karapetyan, N. V. (1986)
Biofizika, 31, 21-26.
33.Schlodder, E., Shubin, V. V., El-Mohsnawy, E.,
Roegner, M., and Karapetyan, N. V. (2007) Biochim. Biophys.
Acta, 1767, 732-741.
34.Shubin, V. V., Roegner, M., El-Mohsnawy, E.,
Terekhova, I. V., Shlodder, E., and Karapetyan, N. V. (2010) Prikl.
Biokhim. Mikrobiol., 46, 299-307.
35.Moskalenko, A. A., and Karapetyan, N. V. (1996)
Z. Naturforsch., 51C, 763-771.
36.Van Grondelle, R., Dekker, J. P., Gilbro, T., and
Sundstroem, V. (1994) Biochim. Biophys. Acta, 1187,
1-65.
37.Trissl, H.-W. (2001) Photosynth. Res.,
35, 247-263.
38.Karapetyan, N. V., Shubin, V. V., and Strasser,
R. J. (1999) Photosynth. Res., 61, 291-301.
39.Karapetyan, N. V. (2007) Biochemistry
(Moscow), 72, 1127-1135.
40.Karapetyan, N. V. (2008) Photosynth. Res.,
97, 195-204.
41.Karapetyan, N. V. (2004) Biochemistry
(Moscow), 69, 1299-1304.
42.Witt, H., Bordignon, E., Carbonera, D., Dekker,
J. P., Karapetyan, N. V., Teutloff, C., Webber, A., Lubitz, W., and
Schlodder, E. (2003) J. Biol. Chem., 278,
46760-46771.
43.Mangels, D., Kruip, J., Berry, S., Roegner, M.,
Boekema, E. J., and Koenig, F. (2002) Photosynth. Res.,
72, 307-319.
44.Gobets, B., van Amerongen, H., Monshower, R.,
Kruip, J., Roegner, M., van Grondelle, R., and Dekker, J. P. (1994)
Biochim. Biophys. Acta, 1188, 75-85.
45.Raetsep, M., Johnson, T. W., Chitnis, P. R., and
Small, G. J. (2000) J. Phys. Chem., 104B, 836-847.
46.Hayes, J. M., Matsuzaki, S., Raetsep, M., and
Small, G. J. (2000) J. Phys. Chem., 104B, 5625-5633.
47.Palsson, L.-O., Dekker, J. P., Schlodder, E.,
Monshouwer, R., and van Grondelle, R. (1996) Photosynth. Res.,
48, 239-246.
48.Schlodder, E., Cetin, M., Byrdin, M., Terekhova,
I. N., and Karapetyan, N. V. (2005) Biochim. Biophys. Acta,
1706, 53-67.
49.Karapetyan, N. V., Dorra, D., Schweitzer, G.,
Bezsmertnaya, I. N., and Holzwarth, A. R. (1997) Biochemistry,
36, 13830-13837.
50.Wittmershaus, B. P., Wolf, V. M., and Vermaas, W.
F. J. (1992) Photosynth. Res., 31, 75-87.
51.Palsson, L.-O. Flemming, C., Gobets, B., van
Grondelle, R., Dekker, J. P., and Schlodder, E. (1998) Biophys.
J., 74, 2611-2622.
52.Byrdin, M., Rimke, I., Schlodder, E., Stehlik,
D., and Roelofs, T. A. (2000) Biophys. J., 79,
992-1007.
53.Van der Lee, J., Bald, D., Kwa, S. L. S., van
Grondelle, R., Roegner, M., and Dekker, J. P. (1993) Photosynth.
Res., 35, 311-321.
54.Woolf, V. M., Wittmershaus, B. P., Vermaas, W.
J., and Tran, T. D. (1994) Photosynth. Res., 40,
21-34.
55.Krasnovsky, A. A., and Bystrova, M. I. (1986)
Biosystems, 7, 181-194.
56.Koehne, B., and Trissl, H.-W. (1998)
Biochemistry, 37, 5494-5500.
57.Gill, E. M., and Wittmershaus, B. P. (1999)
Photosynth. Res., 61, 53-64.
58.Kruip, J., Karapetyan, N. V., Terekhova, I. V.,
and Roegner, M. (1999) J. Biol. Chem., 274,
18181-18188.
59.Frese, R. N., Palacios, M. A., Azzizi, A., van
Stokkum, I. H. M., Kruip, J., Roegner, M., Karapetyan, N. V.,
Schlodder, E., van Grondelle, R., and Dekker, J. P. (2002) Biochim.
Biophys. Acta, 1554, 180-191.
60.Brecht, M., Hussels, M., Schlodder, E., and
Karapetyan, N. V. (2012) Biochim. Biophys. Acta, 1817,
445-452.
61.El-Mohsnawy, E., Kopczak, M. J., Schlodder, E.,
Nowaczyk, M., Meyer, H. E., Warscheid, B., Karapetyan, N. V., and
Roegner, M. (2010) Biochemistry, 49, 4740-4751.
62.Melkozernov, A. N., Lin, S., and Blankenship, R.
E. (2000) J. Phys. Chem., 104, 1651-1656.
63.Savikin, S., Xu, W., Martinsson, P., Chitnis, P.
R., and Struve, W. S. (2001) Biochemistry, 40,
9282-9290.
64.Savikin, S., Xu, W., Chitnis, R. P., and Struve,
W. (2000) Biophys. J., 79, 1573-1586.
65.Zazubovich, V., Matsuzaki, S., Johnson, T. W.,
Hayes, J. M., Chitnis, P. R., and Small, G. J. (2002) Chem.
Phys., 275, 47-59.
66.Karapetyan, N. V., Klimov, V. V., and Krasnovsky,
A. A. (1973) Photosynthetica, 7, 330-337.
67.Karapetyan, N. V., Klimov, V. V., and Krasnovsky,
A. A. (1973) Dokl. Akad. Nauk SSSR, 211, 729-732.
68.Klimov, V. V., Krakhmaleva, I. N., Shuvalov, V.
A., Karapetyan, N. V., and Krasnovsky, A. A. (1975) Dokl. Akad. Nauk
SSSR, 221, 1207-1210.
69.Dorra, D., Fromme, P., Karapetyan, N. V., and
Holzwarth, A. R. (1998) in Photosynthesis: Mechanisms and
Effects (Garab, G., ed.) Kluwer, Dordrecht, Vol. 1, pp.
587-590.
70.Wientjest, E., van Stokkum, I. H. M., van
Amerongen, H., and Croce, R. (2011) Biophys. J., 101,
745-754.
71.Sener, M. K., Jolley, C., Ben-Shem, A., Fromme,
P., Nelson, N., Croce, R., and Schulten, K. (2005) Biophys. J.,
89, 1630-1642.
72.Shelaev, I. V., Gostev, F. E., Mamedov, M. D.,
Sarkisov, O. M., Nadtochenko, V. A., Shuvalov, V. A., and Semenov, A.
Y. (2010) Biochim. Biophys. Acta, 1797, 1410-1420.
73.Karapetyan, N. V. (2004) Biofizika,
49, 212-226.
74.Cometta, A., Zucchelli, G., Karapetyan, N. V.,
Engelman, E., Garlashi, F. M., and Jennings, R. (2000) Biophys.
J., 79, 3235-3243.
75.Engelman, E., Tagliaube, T., Karapetyan, N. V.,
Garlaschi, F. M., Zucchelli, Z., and Jennings, R. C. (2001) FEBS
Lett., 499, 112-115.
76.Shubin, V. V., Terekhova, I. N., Kirillov, B. A.,
and Karapetyan, N. V. (2008) Photochem. Photobiol.
Sci., 7, 956-962.
77.Polm, M., and Brettel, K. (1998) Biophys.
J., 74, 3173-3181.
78.Schlodder, E., Paul, A., and Cetin, M. (2001)
Proc. 12th Photosynthesis Congr., CSIRO Publishing, Melbourne,
S6-015.
79.Schlodder, E., Falkenberg, K., Gergeleit, M., and
Brettel, K. (1998) Biochemistry, 37, 9466-9476.
80.Schlodder, E., Hussels, M., Cetin, M.,
Karapetyan, N. V., and Brecht, M. (2011) Biochim. Biophys. Acta,
1807, 1423-1431.