Singlet–Triplet Fission of Carotenoid Excitation in Light-Harvesting LH2 Complexes of Purple Phototrophic Bacteria
I. B. Klenina*, Z. K. Makhneva, A. A. Moskalenko, N. D. Gudkov, M. A. Bolshakov, E. A. Pavlova, and I. I. Proskuryakov
Institute of Basic Biological Problems, Russian Academy of Sciences, ul. Institutskaya 2, 142290 Pushchino, Moscow Region, Russia; fax: 8 (4967) 330-532; E-mail: ikleonik@yandex.ru* To whom correspondence should be addressed.
Received November 13, 2013; Revision received December 9, 2013
The current generally accepted structure of light-harvesting LH2 complexes from purple phototrophic bacteria conflicts with the observation of singlet–triplet carotenoid excitation fission in these complexes. In LH2 complexes from the purple bacterium Allochromatium minutissimum, a drop in the efficiency of carotenoid triplet generation is demonstrated, which correlates with the extent of selective photooxidation of bacteriochlorophylls absorbing at ~850 nm. We conclude that singlet–triplet fission of carotenoid excitation proceeds with participation of these excitonically coupled bacteriochlorophylls. In the framework of the proposed mechanism, the contradiction between LH2 structure and photophysical properties of carotenoids is eliminated. The possibility of singlet–triplet excitation fission involving a third mediator molecule was not considered earlier.
KEY WORDS: purple phototrophic bacteria, light-harvesting complexes, structural organization, carotenoids, singlet–triplet excitation fissionDOI: 10.1134/S0006297914030092
Abbreviations: BChl, bacteriochlorophyll; Car, carotenoid; CD, circular dichroism; LH2, peripheral light-harvesting complex; RC, reaction center; TR EPR, time-resolved electron paramagnetic resonance; ZFS, zero field splitting.
Carotenoids (Car) are present in all photosynthetic organisms. Of their
two main functions in photosynthesis, one is light absorption in the
spectral region where the main pigments, (bacterio)chlorophylls
((B)Chls), do not absorb efficiently followed by transfer of excitation
energy to the main pigments, and the other is protection from singlet
oxygen, which may be generated when oxygen interacts with
(bacterio)chlorophyll triplet state [1, 2]. The second function is accomplished by quenching
the chlorophyll triplet states due to the presence in carotenoids of
low-lying triplet energy levels that quickly dissipate their triplet
state energy.
In this context, quite unexpected was the discovery of carotenoid triplet state population under their direct excitation [3]. This phenomenon was observed both in peripheral light-harvesting complexes LH2 of a number of purple bacteria [4] and in reaction-center (RC) containing LH1-RC complexes [3]. The high rate of 3Car generation, which competes with the process of energy transfer to BChls, indicates that population of this state results from singlet–triplet fission of carotenoid excitation [3], 1Car* → 3Car + 3Car. Excitation fission is well-studied in the case of molecular crystals [5] and other chemical systems [6]. The characteristic time of this process lies in the pico- and femtosecond range. For excitation fission to proceed, electron clouds of the participating molecules must overlap, i.e. separation between the two molecules should not be significantly greater than 10 Å. To estimate this distance between carotenoid molecules, one needs structural information on LH complexes, and this is available in only a limited number of cases.
In the case of LH2 complexes of Rhodoblastus acidophilus 10050, the X-ray structural data are not entirely unambiguous. According to the structure obtained with 2.0 Å resolution [7], these complexes are composed of nine identical polypeptide heterodimers arranged in a circle. Three BChl and two Car molecules are associated with each dimer. One of the BChl molecules weakly interacts with the rest of the bacteriochlorophylls of the complex and accounts for the optical absorption band with a peak at 800 nm. The other two molecules interact comparatively strongly with each other and similar BChls of the neighboring heterodimers, so that all 18 bacteriochlorophylls of this kind form a band shifted to 850 nm due to excitonic interaction [8-10]. Of the two Car molecules, one has a slightly distorted trans-configuration, while the other is strongly twisted. Raman scattering spectra of the R. acidophilus LH2 complexes [11] showed no indication of bands that could be attributed to Car molecules in such configuration. Consequently, it is now generally accepted that in the paper of Papiz et al. [7] the picture of electron density was interpreted erroneously, and the position of the strongly deformed Car is in fact occupied by a detergent molecule [12]. Within this assumption, the shortest distance between two Car molecules of the neighboring heterodimers will be 13-14 Å, which seems too large for the overlap of electron clouds and for efficient singlet–triplet excitation fission.
A contradiction thus arises between the generally accepted structural organization of LH2 complexes with a single Car molecule per heterodimer and the observation of excitation fission. To overcome this inconsistency, a peculiar proposal was made of the possibility of excitation fission within a single Car molecule [3]; this possibility was later disproved on the basis of the EPR properties of 3Car [13]. Carotenoids of LH2 complexes are located close enough to molecules of BChl 850 and BChl 800 (shortest contact 3.68 and 3.4 Å, correspondingly [14]). Interactions of Car with BChl are strong enough to cause a shift of BChl absorption bands when carotenoid triplet is populated [15], and this changes Car absorption when BChls are in the excited state [16]. It would be interesting to study the possibility of the participation of BChls in the process of fission of Car excitation. To approach this problem, we utilized earlier observed phenomenon of BChl 850 bleaching under light exciting Car in LH2 complexes from the purple bacterium Allochromatium minutissimum [17]. A similar effect in the case of R. acidophilus was not detected.
Here we used the technique of time-resolved EPR (TR EPR) for detecting the 3Car state. The TR EPR method provides information not only on the nature of triplet-bearing molecules, but often also on the mechanism of the triplet state population. Information on the molecular identity of a triplet molecule is given by the parameters of zero field splitting (ZFS) of its EPR spectrum [18]. The population mechanism affects the spin polarization pattern of the spectra, i.e. the distribution of spectral components detected in emission and enhanced absorption of microwave power [19]. Apart from this advantage, TR EPR is better suited in studies of short-lived (usually shorter than 10 μs) 3Car states. In our earlier works [13, 20] we reported the detection with TR EPR of carotenoid triplet states populated by excitation fission in LH2 complexes of A. minutissimum. An extensive investigation of the LH2 carotenoid triplet states was also carried out by Bittl et al. [21]. The spectra presented in that work were, however, erroneously interpreted as belonging to the 3Car populated due to T–T energy transfer from 3BChl, though spin polarization of EPR spectra unambiguously pointed to the process of excitation fission [13, 20].
In the present work we studied the dependence of the efficiency of 3Car state population in the process of excitation fission in light-harvesting complexes from A. minutissimum on the extent of BChl 850 photobleaching.
MATERIALS AND METHODS
The culture of the sulfur purple bacterium A. minutissimum from the collection of the Department of Microbiology, Biological Faculty, Moscow State University (Russia) was used. Cells of A. minutissimum were grown under anaerobic conditions at 1000 lx illumination and 25-27°C as described by Kondratieva [22]. The culture of non-sulfur purple bacterium R. acidophilus 10050 was obtained from the DSMZ collection (Germany). The cells of R. acidophilus were grown under the same conditions on a medium [23]. After 4-6 days, the cells in the stationary phase of growth were harvested, and biomass was used in experiments directly after storage at –18°C.
Pigment-containing membranes (chromatophores) were obtained by cell sonication followed by differential centrifugation. For chromatophore solubilization, they were treated with 2-2.5% w/v dodecyl maltoside. LH2 complexes were isolated using preparative electrophoresis in 7% polyacrylamide gel. Electrophoretic bands were cut out, and the gel was minced and eluted overnight with 10 mM Tris-HCl, pH 7.5, followed by centrifugation in Amicon Ultra 50k tubes (Millipore, USA). The procedure of LH2 isolation is described in more detail in the paper of Klenina et al. [20].
Optical absorption spectra were recorded on a Cary 50 spectrophotometer (Varian, USA). The purity of preparations was checked by their absorption spectrum, polypeptide composition, and by repeated electrophoresis.
CD spectra at room temperature were taken with a Jasco-600 spectropolarimeter (Jasco, Japan) at sample optical density of 0.5 at 850 nm.
Illumination of LH2 complexes, which led to bleaching of the 850-nm absorption band, was performed while pumping 1.5 ml of the preparation with optical density of 70 at 850 nm through a flat cell of 0.2 mm optical path. The full light of halogen lamp lightguide illuminator OVS-1 (LZOS, Russia) providing 100 mW/cm2 intensity was used. In the course of illumination, 50-μl volumes of the preparation were sampled, mixed with double volume of glycerol, and placed into standard 3 mm (i.d.) Suprasil tubes. The tubes were immediately frozen in the dark and stored in liquid N2 until EPR measurements. The maximum illumination time was 8 h. After recording the EPR spectra, the samples were thawed and diluted for spectrophotometric determination of BChl photobleaching.
The TR EPR measurements were performed using a home-built direct-detection EPR spectrometer. Magnetic field modulation was switched off, and the signal was taken from the preamplifier of the spectrometer microwave bridge (X-band spectrometer RE 1306; Russia). The preamplifier output signal was fed into an SR250 boxcar integrator (Stanford Research Systems, USA), where it was integrated for a preset time period. The front of the integration period was synchronized with excitation light flash and started at a preset delay after it. The boxcar output signal was recorded by a computer synchronously with the magnetic field sweep that was controlled by the same computer. To increase sensitivity, the sweep was multiply repeated, and the obtained spectra were summed. The excitation light flash was provided by a laser system (Solar Laser Systems, Belarus) comprising an LQ529B pump YAG laser and an LP603 parametric oscillator. The flash energy at the sample surface was ~2 mJ with pulse duration of 6 ns, wavelength 470 nm, and generation bandwidth < 1 nm. The sample temperature was stabilized at 120 K using an ESR900 cryostat (Oxford Instruments, UK).
RESULTS
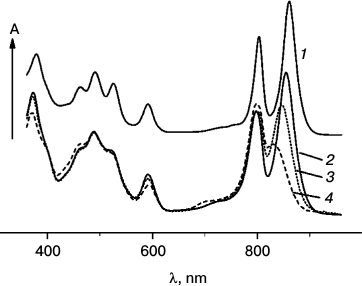
Figure 1 shows optical absorption spectra of LH2 complexes from R. acidophilus (1) and A. minutissimum (2) at room temperature and changes of the A. minutissimum spectrum caused by illumination for different periods of time (3, 4). The spectra of both complexes are composed of bacteriochlorophyll absorption (bands at 800 and 850-860 nm, a band common for the two BChl types at 590 nm, and in the region of 370 nm, the so-called Soret band) and carotenoid absorption (in the spectral region 420-550 nm). Illumination-induced spectral changes are limited to the bleaching and shift of the 850 nm band, to absorption decrease at 590 nm and in the Soret band, and appearance of a new band around 700 nm accompanied by absorption increase in the range 410-460 nm. The carotenoid absorption remains virtually unchanged.
Fig. 1. Optical absorption spectra of LH2 complexes measured at room temperature: 1) R. acidophilus; 2-4) A. minutissimum (2 – before illumination; 3 – 40 min illumination; 4 – 4 h illumination).
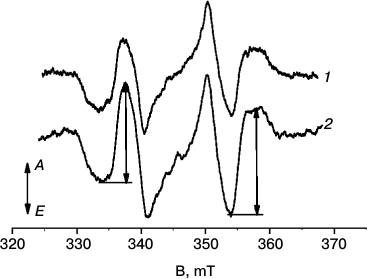
In Fig. 2, TR EPR spectra of LH2 complexes from R. acidophilus (1) and A. minutissimum (2) are shown. The spectra demonstrate the shown shape during 2-4 μs after the laser flash exciting carotenoids. Both spectra have the same ZFS parameters, |D| = 0.0279 cm–1 and |E| = 0.0026 cm–1, characteristic of rhodopin and rhodopin glucoside [24]. The spin polarization pattern (EAEAEA, E – emission, A – absorption of microwave power) corresponds to that observed earlier in LH2 complexes and films of isolated carotenoids [13, 20] and interpreted as resulting from singlet–triplet excitation fission. In samples prepared after varying periods of white-light illumination, the EPR signal amplitudes decrease with increased illumination period. In the following, the signal amplitude was determined as a sum of the two amplitudes shown in Fig. 2 with arrows. To increase the accuracy, the EPR signals were averaged for a few points around their extrema.
Fig. 2. TR EPR spectra of LH2 complexes of (1) R. acidophilus and (2) A. minutissimum before illumination measured in the period of 2-4 μs after an excitation laser flash. T = 120 K, λex = 470 nm. Letters A and E at the left-hand arrow indicate directions corresponding to absorption and emission of microwave power, correspondingly. The arrows on spectrum 2 show the measured amplitudes of the signal.
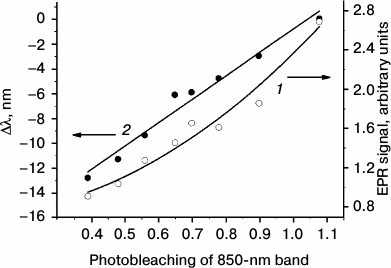
For quantitative analysis of overlapping IR bacteriochlorophyll absorption bands (determination of positions, amplitudes, and widths), spectral deconvolution was performed. It appeared that the BChl absorption spectrum in the 700-950 nm region was best reproduced by the sum of two bands, a Lorentzian band peaking near 800 nm and a Gaussian band with a maximum around 850 nm. The Lorentzian shape of the 800 nm band is probably related to its complex nature, which shows as a band splitting at low temperature [20]. Spectral deconvolution demonstrated that neither the position or width, nor the amplitude of the 800 nm band (more precisely, 798 nm), changed appreciably in the course of illumination. On the contrary, the BChl 850 band significantly (~3-fold) decreased in amplitude and shifted in the short-wavelength direction. Figure 3 shows the changes in the EPR signal amplitude and shift of the 850-nm band that accompany photobleaching of BChl 850. The decrease in BChl 850 absorption is presented in Fig. 3 as a drop in the ratio of its amplitude to that of the 800 nm band amplitude, which during photobleaching remains practically constant. Two sets of photobleaching experiments were performed, both giving qualitatively the same result. In Fig. 3 we give the results of the set where deeper changes in optical absorption were achieved.
Fig. 3. Amplitude of TR EPR signal (1) and shift of BChl 850 optical absorption band (2) at different photobleaching stages of this bacteriochlorophyll. Photobleaching is presented as the ratio of amplitudes of the BChl 850 band to BChl 800 band obtained from deconvolution of the absorption spectrum. The TR EPR signal amplitude is given in arbitrary units.
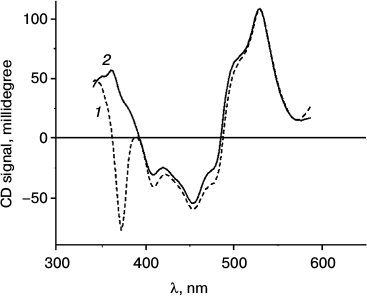
Figure 4 illustrates changes in the CD spectrum of A. minutissimum LH2 complexes in the region of Car absorption brought about by illumination. These changes are discussed in the following section.
Fig. 4. CD spectra of A. minutissimum LH2 complexes before (1) and after (2) illumination, which causes photobleaching of the BChl 850 absorption band.
DISCUSSION
Spectral changes caused by illumination of an A. minutissimum LH2 preparation (Fig. 1) are almost entirely restricted to absorption bands of BChl 850. Apart from these, absorption bands at 700 nm and the 410-460-nm region appear. The apparent growth of the BChl 800 band originates due to a shift of the overlapping 850-nm band, and absorption growth in the short-wavelength wing of the Car spectrum to the accumulation of the photoreaction product and its Soret band. The observed spectral changes generally correspond well to data obtained earlier for Car excitation in A. minutissimum whole cells [17]. There the appearing absorption bands were ascribed to generation of BChl oxidation product as the result of excitation into the carotenoid band (415-570 nm) or by white light in the presence of oxygen.
The circular dichroism spectra are often used to study interactions of carotenoids with their protein environment. Such possibility arises due to the absence of achiral carotenoid CD in solution of the pigments [2], so that the CD spectra observed in LH complexes are entirely determined by the changes in molecular geometry resulting from incorporation of Car into the polypeptide matrix. The CD spectra measured before (Fig. 4, 1) and after (Fig. 4, 2) illumination of A. minutissimum LH2 complexes show the only major difference, that is, disappearance of the band around 370 nm after illumination. The position of this band coincides with the maximum of the BChl Soret band. Earlier this band in the CD spectra of LH2 complexes has been attributed to BChl [25]. Now it may be more specifically identified as the band of excitonically interacting BChl 850. From the data of Fig. 4, it can be concluded that the procedure of illumination induces no major changes in the CD properties of the carotenoid.
The general conclusion can be drawn that illumination of A. minutissimum LH2 complexes modifies the properties of BChl 850 molecules leaving BChl 800 and carotenoids practically unaffected. The observed decrease in 3Car generation efficiency (Fig. 3) can thus be associated with changing properties of this excitonically coupled long-wavelength bacteriochlorophyll. For further analysis, it is important to develop a concept of the possible structural organization of carotenoids and bacteriochlorophylls in the A. minutissimum LH2.
There is no X-ray structural data on the A. minutissimum complexes. It is reasonable, however, to assume their close resemblance to the structure of R. acidophilus LH2 complexes. There are several experimentally observed properties that support this assumption. First, according to electron microscopy data [26], ring-like arrangement of LH2 complexes is observed in all studied purple bacteria, and is likely to be universal in this type of microorganisms. Then, the LH2 absorption spectra of R. acidophilus and A. minutissimum (Fig. 1) show only minor differences in the BChl absorption bands, and differ not much in the Car absorption region. Similarity of BChl spectral properties are an indication of close resemblance of interactions and consequently of structural organization of these molecules of the two complexes. Difference in Car absorption arises probably due to varying carotenoid composition of LH2 of the two bacteria. In R. acidophilus it is mainly rhodopin glucoside and rhodopin [7, 27], while in the A. minutissimum composition is more diversified and apart from rhodopin is represented also by didehydrorhodopin, spirilloxanthin, and a number of minor carotenoids [28]. Finally, the EPR spectral properties of 3Car observed under excitation into the Car absorption band in these two bacteria are identical (Fig. 2). These coincidences make it possible to study the role of BChl 850 in carotenoid singlet–triplet excitation fission in A. minutissimum LH2 relying on the structural data of R. acidophilus complexes.
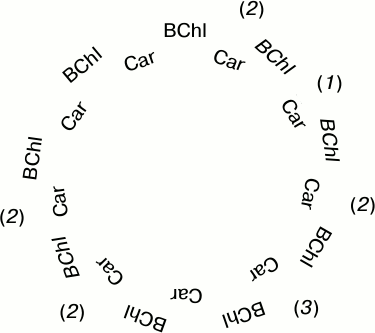
For quantitative description of the process, let us consider a simplified model in which bacteriochlorophylls and carotenoids are arranged in a circular structure as shown in Fig. 5. The BChl molecules with their properties modified by illumination are shown in Fig. 5 in italic. There are three possible variants developing in the course of illumination. Bacteriochlorophylls may get photobleached on both sides of a carotenoid (1), only one BChl molecule may be photobleached (2), or both neighboring bacteriochlorophylls may remain unchanged (3). BChl photobleaching leads to certain changes in its properties that influence the interaction with Car. Let us make the following simplifying assumptions:
– singlet–triplet fission of Car excitation develops with participation of BChl molecules and is completely terminated when both neighboring BChls are photobleached;
– excitation fission proceeds normally if an unaltered BChl molecule remains on at least one side of a carotenoid.
Fig. 5. Simplified structure of LH2 complexes used for developing the statistical model of BChl 850 photobleaching. The number of Car molecules (N = 9) corresponds to the generally accepted structure of R. acidophilus LH2 complexes.
One more simplification is in substituting dimeric BChl separating Car molecules with monomeric BChls. In practice, this means that for photobleaching of a BChl dimer, photooxidation of a single BChl molecule constituting the dimer is enough. The current state of knowledge on the properties of LH2 complexes is, unfortunately, insufficient for using in calculations the complete structure of the complexes.
Under these assumptions, the statistical problem of calculating the probability of 3Car generation dependence on the number of photobleached BChl molecules m can be solved for any initial number N of BChls (and Car). The probability of excitation fission with 3Car population appears to be:
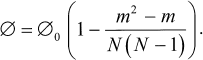
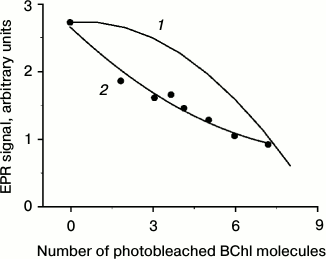
The observed amplitude of the TR EPR signal is calculated by replacing the initial probability of fission (0) by the initial amplitude of the 3Car signal. In Fig. 6 (1) this dependence is represented in the case of N = 9 (note that the appearance of the curve is qualitatively independent of the number N). In Fig. 6 (2) experimental points of Fig. 3 (1) are given, being recalculated to the number of photobleached BChl molecules. The calculated and experimental curves show apparent differences. The experimental curve undergoes steep decrease at the initial stage of photobleaching. The calculated dependence remains almost constant at low levels of photobleaching. This behavior has a simple physical interpretation. At low photobleaching levels the probability of a “double hit” modifying BChls on both sides of a carotenoid (case 1, Fig. 6) is low, and excitation fission remains efficient. Another difference between the two curves in Fig. 6 is that the experimental dependence shows a tendency for leveling-off at high photobleaching numbers, but not approaching zero.
Fig. 6. Theoretical (1) and experimental (2) dependence of the TR EPR signal amplitude (in arbitrary units) on the number of photobleached BChl 850 molecules in the course of illumination of A. minutissimum LH2 complexes. The number of photobleached molecules was calculated under the assumption m = 0 before illumination and m = 9 at complete bleaching of the BChl 850 band.
The reasons for the initial quick drop in 3Car population remain unclear. The preliminary hypothesis can be put forward that excitation fission proceeds not like described above, but with participation of all excitonically coupled molecules of BChl 850. This possibility is suggested by the pronounced short-wavelength shift of the 850-nm band appearing already at the initial stages of photobleaching (Fig. 3, 2). This shift most likely is related to the change in excitonic interaction between BChl 850 molecules. The same can be concluded from the disappearance of the excitonic band at 370 nm in the CD spectrum (Fig. 4). Generation of 3Car at high levels of BChl 850 photobleaching points at the existence of another, less efficient channel of excitation fission. Possibly it is a process developing with the participation of BChl 800. These assumptions require further investigation.
The possibility of singlet–triplet excitation fission with participation of a third molecule (the more so, of a group of excitonically coupled molecules) has not been discussed in the literature before. The physical mechanism of such a process remains somewhat unclear. An important experimental fact in this respect is the absence of detection of 3BChl, which would accompany 3Car generation, even in high-resolution (picosecond) measurements [3]. This is reminiscent of the processes developing via the so-called superexchange mechanism, when participation of intermediary molecules is only virtual. For example, in the work of Eng et al. [29] a related to excitation fission process of triplet–triplet energy transfer was studied between two porphyrin molecules separated by a covalently linked bridge of 2-5 molecules of different chemical nature with triplet energy levels that are inaccessible for porphyrin. An efficient energy transfer between porphyrins proceeding via the superexchange mechanism was observed involving bridge molecules but without population of their triplet states. The T–T energy transfer was detected at inter-porphyrin distances exceeding 30 Å. Probably within the same category falls the process of triplet excitation transfer from the RC primary donor to Car, which proceeds with participation of the monomeric BChl molecule [30, 31], where the population of the 3BChl state was also never detected.
It can be concluded that the effect of BChl 850 photooxidation on the generation of 3Car observed in this work brings in compliance X-ray structural data showing the presence of a single Car molecule per polypeptide heterodimer and observation of singlet–triplet fission of carotenoid excitation.
The authors thank B. S. Melnik (Institute of Protein Research, Russian Academy of Sciences) for help with measuring CD spectra and Z. A. Zhuravleva for growing A. minutissimum and R. acidophilus cultures.
Financial support of the Russian Foundation for Basic Research (grants 12-04-00412-a and 13-04-01184-a) and the MCB Program of the Russian Academy of Sciences Presidium is gratefully acknowledged.
REFERENCES
1.Krasnovsky, A. A., Jr. (1977) Photochem.
Photobiol., 29, 29-36.
2.Frank, H. A., and Cogdell, R. J. (1993) in
Carotenoids in Photosynthesis (Young, A., and Britton, G., eds.)
Chapman and Hall, London, pp. 252-326.
3.Gradinaru, C. C., Kennis, J. T. M., Papagiannakis,
E., van Stokkum, I. H. M., Cogdell, R. J., Fleming, G. R., Niederman,
R. A., and van Grondelle, R. (2001) Proc. Natl. Acad. Sci. USA,
98, 2364-2369.
4.Papagiannakis, E., Kennis, J. T. M., van Stokkum,
I. H. M., Cogdell, R. J., and van Grondelle, R. (2002) Proc. Natl.
Acad. Sci. USA, 99, 6017-6022.
5.Swenberg, C. E., and Geacintov, N. E. (1973) in
Organic Molecular Photophysics, Vol. 1 (Birks, J. B., ed.) John
Wiley and Sons, London, pp. 489-564.
6.Smith, M. B., and Michl, J. (2010) Chem.
Rev., 110, 6891-6936.
7.Papiz, M. Z., Prince, S. M., Howard, T., Cogdell,
R. J., and Isaacs, N. W. (2003) J. Mol. Biol., 326,
1523-1538.
8.Cogdell, R. J., Howard, T. D., Isaacs, N. W.,
McLuskey, K., and Gardiner, A. T. (2002) Photosynth. Res.,
74, 135-141.
9.Renger, T., May, V., and Kuhn, O. (2001) Phys.
Rep., 343, 137-254.
10.Dracheva, T. V., Novoderezhkin, V. I., and
Razjivin, A. P. (1996) FEBS Lett., 387, 81-84.
11.Gall, A., Gardiner, A. T., Cogdell, R. J., and
Robert, B. (2006) FEBS Lett., 580, 3841-3844.
12.Cherezov, V., Clogston, J., Papiz, M. Z., and
Caffrey, M. (2006) J. Mol. Biol., 357, 1605-1618.
13.Klenina, I. B., Makhneva, Z. K., Moskalenko, A.
A., and Proskuryakov, I. I. (2011) Dokl. Biochem. Biophys.,
441, 290-293.
14.Freer, A., Prince, S., Sauer, K., Papiz, M.,
Hawthornthwaite-Lawless, A., McDermott, G., Cogdell, R., and Isaacs, N.
W. (1996) Structure, 4, 449-462.
15.Angerhofer, A., Bornhauser, F., Gall, A., and
Cogdell, R. J. (1996) Chem. Phys., 194, 259-274.
16.Herek, J. L., Polivka, T., Pullerits, T., Fowler,
G. J. S., Hunter, C. N., and Sundstrom, V. (1998) Biochemistry,
37, 7057-7061.
17.Makhneva, Z. K., Erokhin, Yu. E., and Moskalenko,
A. A. (2007) Dokl. Biochem. Biophys., 416,
256-259.
18.Wertz, J. E., and Bolton, J. R. (1972)
Electron Spin Resonance. Elementary Theory and Practical
Applications, McGraw-Hill Book Company, N. Y.
19.Klenina, I. B., Kuzmin, A. N., Fufina, T. Yu.,
Gudkov, N. D., and Proskuryakov, I. I. (2011) Biophysics
(Moscow), 56, 429-435.
20.Klenina, I. B., Makhneva, Z. K., Moskalenko, A.
A., Kuzmin, A. N., and Proskuryakov, I. I. (2013) Biophysics
(Moscow), 58, 43-50.
21.Bittl, R., Schlodder, E., Geisenheimer, I.,
Lubitz, W., and Cogdell, R. J. (2001) J. Phys. Chem. B,
105, 5525-5535.
22.Kondratieva, E. N. (1963) The Photosynthetic
Bacteria [in Russian], USSR Academy of Sciences Publishers,
Moscow.
23.Pfennig, N. (1969) J. Bacteriol.,
99, 597-602.
24.Frank, H. A., Chadwick, B. W., Oh, J. J., Gust,
D., Moore, T. A., Liddell, P. A., Moore, A. L., Makings, L. R., and
Cogdell, R. J. (1987) Biochim. Biophys. Acta, 892,
253-263.
25.Cogdell, R. J., and Scheer, H. (1985)
Photochem. Photobiol., 42, 669-678.
26.Sturgis, J. N., and Niederman, R. A. (2009) in
Advances in Photosynthesis and Respiration, Vol. 28 (Hunter, C.
N., Daldal, F., Thurnauer, M. C., and Beatty, J. T., eds.) Springer,
Dordrecht, pp. 253-273.
27.Cogdell, R. J., Fyfe, P. K., Howard, T. D.,
Fraser, N., Isaacs, N. W., Freer, A. A., McKluskey, K, and Prince, S.
M. (1999) in Advances in Photosynthesis, Vol. 8 (Frank, H. A.,
Young, A. J., Britton, G., and Cogdell, R. J., eds.) Kluwer Acad.
Publishers, N. Y., pp. 71-80.
28.Makhneva, Z., Bolshakov, M., and Moskalenko, A.
(2008) Photosynth. Res., 98, 633-641.
29.Eng, M. P., Ljungdahl, T., Martensson, E., and
Albinsson, B. (2006) J. Phys. Chem. B, 110,
6483-6491.
30.Frank, H. A., Chynwat, V., Posteraro, A.,
Hartwich, G., Simonin, I., and Scheer, H. (1996) Photochem.
Photobiol., 64, 823-831.
31.Angerhofer, A., Bornhauser, F., Aust, V.,
Hartwich, G., and Scheer, H. (1998) Biochim. Biophys. Acta,
1365, 404-420.