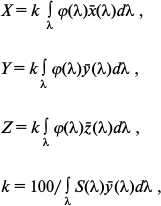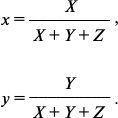Chlorophyll Fluorescence Induction, Chlorophyll Content, and Chromaticity Characteristics of Leaves as Indicators of Photosynthetic Apparatus Senescence in Arboreous Plants
V. V. Ptushenko1,2*, O. S. Ptushenko3, and A. N. Tikhonov2,4
1Lomonosov Moscow State University, Belozersky Institute of Physico-Chemical Biology, 119992 Moscow, Russia; fax: +7 (495) 939-0338; E-mail: fxb@genebee.msu.su; ptush@belozersky.msu.ru2Emanuel Institute of Biochemical Physics, Russian Academy of Sciences, ul. Kosygina 4, 119334 Moscow, Russia; fax: +7 (499) 137-4101; E-mail: ibcp@sky.chph.ras.ru
3Moscow Secondary School No. 261, ul. Kastanaevskaya 29/1, 121108 Moscow, Russia; E-mail: sch261@sch261.edu.ru
4Lomonosov Moscow State University, Faculty of Physics, 119992 Moscow, Russia; fax: +7 (495) 932-8820; E-mail: info@physics.msu.ru
* To whom correspondence should be addressed.
Received November 27, 2013; Revision received December 11, 2013
Parameters of chlorophyll fluorescence induction (CFI) are widely used for assessment of the physiological state of higher plant leaves in biochemical, physiological, and ecological studies and in agricultural applications. In this work we have analyzed data on variability of some CFI parameters – ΦPSIImax = Fv/Fm (relative value of variable fluorescence), qNPQ (non-photochemical quenching coefficient), RFd (“vitality index”) – in autumnal leaves of ten arboreous plant species of the temperate climatic zone. The correlation between the chlorophyll content in the leaves and fluorescence parameters characterizing photosynthetic activity is shown for two representative species, the small-leaved linden Tilia cordata and the rowan tree Sorbus aucuparia. During the period of mass yellowing of the leaves, the ΦPSIImax value can be used as an adequate characteristic of their photochemical activity, while in summer the qNPQ or RFd values are more informative. We have established a correlation between the ΦPSIImax value, which characterizes the maximal photochemical activity of the photosystem II, and “chromaticity coordinates” of a leaf characterizing its color features. The chromaticity coordinates determined from the optical reflection spectra of the leaves serve as a quantitative measure of their hues, and this creates certain prerequisites for a visual expert assessment of the physiological state of the leaves.
KEY WORDS: photosynthesis, chlorophyll fluorescence, chromaticity characteristics of leavesDOI: 10.1134/S0006297914030122
Abbreviations: Car, carotenoids; CFI, chlorophyll fluorescence induction; Chl, chlorophyll; CIE, International Commission on Illumination (fr. Commission internationale de l'éclairage); NPQ, non-photochemical quenching; PSI, photosystem I; PSII, photosystem II; PSA, photosynthetic apparatus; qNPQ, non-photochemical fluorescence quenching coefficient, qNPQ = (Fm - Fm′)/Fm′; RFd, “vitality index”, RFd = (FM - FT)/FT; ΦPSII, efficiency of PSII photochemical activity, ΦPSII = (Fm′ - F ′)/Fm′; ΦPSIIst.st., steady-state value of the PSII photochemical activity; ΦPSIImax, maximal efficiency of PSII photochemical activity, ΦPSIImax = Fv/Fm.
An approach of chlorophyll fluorescence induction (CFI) is widely used
for assessment of the physiological state of the photosynthetic
apparatus (PSA) of plants [1-11]. This method is used for solution of fundamental
problems in biochemistry and biophysics of photosynthesis, plant
physiology, and also in various ecological and agricultural studies,
etc. The main advantages of fluorescence studies on the PSA are
noninvasiveness, the possibility of performing express analysis under
field conditions, and the sensitivity of parameters determined by this
method to some practically important stress factors of the environment
[1, 7, 12,
13]. The first works using fluorescence methods of
PSA analysis were performed more than 80 years ago by Kautsky and
Hirsch [14]. They detected non-monotonic changes
in the fluorescence intensity of leaves of different plants. The slow
phase of the fluorescence induction curve correlated with the rate of
CO2 assimilation, was suppressed at low temperatures, and
was inhibited with cyanide. It is now known that chlorophyll a
(Chl a) of photosystem II (PSII) is the major source of
fluorescence of green plant leaves; the contribution of photosystem I
(PSI) to the gross fluorescence of the PSA at physiological
temperatures is insignificant, but it increases at low temperatures
(4-120 K) [6]. From the mid-1980s a method of pulse
amplitude modulation fluorimetry (PAM-fluorimetry) has received wide
application [15]. This method is based on the
measurement of fluorescence when a plant is illuminated by a continuous
“actinic” light along with short pulses of light with
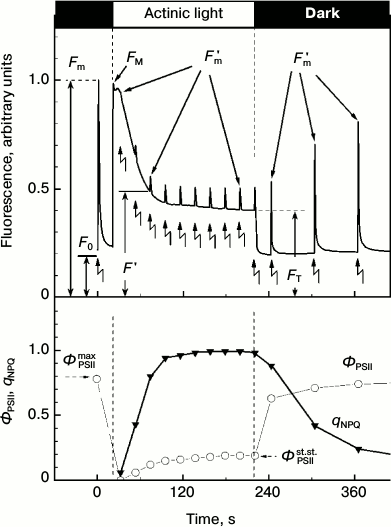
saturating intensity. Figure 1 presents a typical
picture of changes in the Chl fluorescence intensity in higher plant
leaves obtained according to the routine protocol of measurements.
Zigzag-like arrows indicate the application of saturating intensity
light flashes given in the dark or on the background of continuous
illumination. The initial fluorescence level F0
corresponds to luminescence under the influence of weak pulses of
measuring light. In response to the first flash of the saturating light
given before the switching on the continuous light, the fluorescence
increases from the initial level F0 to the maximal
level Fm. After the abatement of fluorescence in the
dark, continuous light was added. The fluorescence intensity measured
during the continuous light changes non-monotonically. Figure 1 shows that immediately after the addition of the
continuous light the fluorescence intensity first rapidly increases and
then decreases relatively slowly. The decrease can be caused, first, by
activation in the light of the Calvin cycle reactions and, second, by
strengthening of the thermal deactivation of Chl excited states within
the light-harvesting antenna. The illumination of chloroplasts leads to
a fast reduction of the plastoquinone pool that manifests itself by a
relatively fast initial increase in fluorescence intensity. The
light-induced activation of the Calvin cycle reactions accelerates
electron outflow from PSI. This results in re-oxidation of the
plastoquinone pool, which reveals itself as a decrease in the Chl
fluorescence (photochemical fluorescence quenching) [3, 9, 16, 17].
Fig. 1. Kinetics of chlorophyll fluorescence induction of a small-leaved linden (T. cordata) leaf recorded using the PAM-protocol. Moments of addition and turning-off of the continuous actinic light are shown by vertical lines, and moments of saturating intensity light flashes are shown by zigzag arrows. Changes in fluorescence on leaf illumination and in the ΦPSII and qNPQ parameters are shown on the upper and lower panels, respectively.
The abatement of the fluorescence under the action of the continuous light is caused also by strengthening of the thermal deactivation of Chl excited states within the light-harvesting antenna, so-called non-photochemical quenching (NPQ) of Chl fluorescence [7, 9, 18]. The non-photochemical quenching decreases the probability of generation of singlet oxygen, which can initiate uncontrolled oxidative reactions in the cell [19-21]. The increase in NPQ can be recorded by a decrease in the fluorescence intensity measured on the imposition of saturating light flashes given on the background of the continuous light (Fig. 1). The increase in NPQ of Chl fluorescence within the light-harvesting antenna is known [22-25] to reflect the protective reaction of the plant PSA in response to excess illumination.
The efficiency of NPQ can be characterized by the parameter qNPQ = (Fm - Fm′)/Fm′ (see determinations in Fig. 1), i.e. by the coefficient of non-photochemical fluorescence quenching. Photochemical activity of PSII is often characterized by the parameter ΦPSIImax = Fv/Fm (Fv = Fm - F0) measured in response to a saturating light flash given after a sufficiently long-term adaptation of the leaf to darkness (from dozens of minutes to some hours [10, 22]). The parameter ΦPSIImax characterizes the maximal efficiency of PSII photochemical activity. The operating efficiency of PSII, ΦPSII = (Fm′ - F ′)/Fm′, measured during illumination of the leaf (or after insufficiently long-term dark adaptation) is usually lower than ΦPSIImax, and its value decreases with increase in the light intensity [10]. The value of ΦPSII gradually increases along with adaptation to illumination and reaches the steady-state level ΦPSIIst.st. (Fig. 1). The intensity of the light-dependent electron transport through PSII at the given illumination is proportional to the parameter ΦPSII measured at a given moment under given conditions. Along with these coefficients, the coefficients RFd = (FM - FT)/FT [26, 27] and FM/FT = RFd + 1 [28, 29] are used also. Depending on the particular task, other CFI parameters measured over a wide time interval (from milliseconds to dozens of minutes) are also used [8, 9, 30, 31].
It is still actively discussed which CFI parameters are the most useful for analysis of photosynthetic activity of the PSA and its protective reactions against the excess light. In particular, the coefficient of non-photochemical quenching of Chl fluorescence qNPQ is used rather frequently as a parameter characterizing the weakening of PSII activity associated with the activation of mechanisms for PSA protection against light-induced damage [18, 23, 24]. On the other hand, Demmig-Adams et al. [22] have criticized the use of this parameter for assessment of physiological state of plants under conditions of long-term (e.g. seasonal) suppression of photosynthetic activity. To assess the stress-induced suppression of photosynthetic activity, they proposed the use of the parameter ΦPSIImax.
In the present work, we have compared the possibilities of using some parameters of CFI for assessment of the physiological state of leaves of arboreous plants of the temperate climatic zone in summer and in autumn. The wide use of the parameter ΦPSII and other above-listed parameters for assessment of the plant physiological state requires studies on their natural variability. In this paper we present data on variability of some CFI parameters for leaves of arboreous plants of the temperate climatic zone under conditions of their natural growth and on its correlation with contents of pigments in the leaves. According to our data, during the period of mass yellowing of leaves, their photochemical activity can be adequately characterized with the parameter ΦPSIImax, whereas in summer such above-presented steady-state characteristics as ΦPSIIst.st., qNPQ, or RFd are preferable. We have analyzed also the possibility of using a visual expert assessment on choosing a homogenous (in functional characteristics of the photosynthetic activity) material for photosynthetic studies. In this paper we consider the expert assessment as a visual (non-instrumental) assessment of leaf parameters characterizing its photosynthetic activity. Such assessment is principally underlain by a correlation in the decrease in PSA activity of arboreous plant leaves in autumn with changes in their pigment composition [32-34]. Besides, such assessment, though very rough, is necessary on dealing with a real plant material. Moreover, in some works spectral features of leaves and chlorophyll-containing fruits were studied in detail, and the possibility was shown of using instrumentally-measured reflection spectra for determination of pigment contents in plant tissues [34-37]. However, according to our knowledge, the possibility of quantitative visual assessment of PSA characteristics has never been considered. In the present work, based on our data on the correlations between characteristics of the PSA of autumnal leaves (ΦPSIImax, ΦPSIIst.st., qNPQ, and RFd) and the Chl content, and using the literature data on the threshold color discrimination by the human eye, we have attempted to answer the question about characteristic differences of ΦPSIImax in the leaves of arboreous plants in autumn that can be visually assessed by a researcher.
MATERIALS AND METHODS
Objects of the study. The study was performed on leaves of some arboreous plants of the temperate climatic zone: small-leaved linden (Tilia cordata Mill.), rowan tree (Sorbus aucuparia L.), stag’s-head (Salix fragilis L.), Norway maple (Acer platanoides L.), hawthorn (Crataegus sp. L.), Norway spruce (Picea abies (L.) H. Karst), blue spruce (Picea pungens Engelm.), Archangel’ fir (Pinus sylvestris L.), Siberian larch (Larix sibirica Ledeb.), and the semi-shrub with partially lignified sprouts – the red raspberry (Rubus idaeus L.). The measurements were performed in August and in October 2009-2013.
Measurements of chlorophyll fluorescence induction. Induction curves of fluorescence of leaves were measured with a portable FluorPen FP100 fluorimeter (Photon System Instruments, Czechia). The CFI parameters ΦPSIImax = Fv/Fm, ΦPSII = (Fm′ - F ′)/Fm′, qNPQ = (Fm - Fm′)/Fm′, and RFd = (FM - FT)/FT were determined according to published works [8, 9, 27] (see Fig. 1 for determination of the fluorescence parameters). To study correlations of the CFI parameters and Chl content in the leaves, the measurements were performed on cuttings from dark-adapted leaves. In all other cases the measurements were performed on intact leaves that had not been separated from the plant in the evening (not earlier than 2 h after sunset, i.e. on dark-adapted leaves). The fluorescence was excited and measured from the ventral (abaxial) side of the leaf.
Measurement of reflection spectra of leaves and calculation of chromaticity coordinates. Reflection spectra of leaves, r(λ), were measured with a portable PolyPen RP 400 spectrophotometer (Photon System Instruments) in the range 380-780 nm. The measurements were performed on the same cuttings of the leaves immediately after the CFI determination.
To determine chromaticity coordinates of the leaf cutting, the reflected light spectrum φ(λ) was calculated on illumination of the specimen with daylight. The energy distribution S(λ) in the daylight spectrum was assumed according to the International Commission on Illumination (CIE) standard D65 [38]. The energy distribution in the light reflected from the leaf in this case was determined by a simple ratio: φ(λ) = r(λ) S(λ).
Then, to describe the visual perception of the surface color with the given spectrum of the reflected light by an average human eye (according to the CIE nomenclature it is termed “CIE 1931 standard colorimetric observer”), we calculated the coordinates in three-dimensional CIE XYZ color space (so-called CIE tristimulus values) – X, Y, and Z of the given color, which approximately corresponded to the yellow-green, green, and blue-green spectral components of the analyzed light. The CIE tristimulus values were calculated by the formulas:
where x̅(λ), y̅(λ), and z̅(λ) are the so-called CIE color-matching functions characterizing the color perception of “CIE 1931 standard colorimetric observer”. Table values of these functions asserted by the CIE are taken from the work [38]. Finally, from the X, Y, and Z coordinates describing the perceptible color in the CIE XYZ color space, the so-called two-dimensional CIE chromaticity coordinates x and y were calculated, which characterized the object color (as differentiated from the color coordinates X, Y, and Z, which in addition to the color also characterize the brightness [38]):
We used the x and y values for further analysis of the data.
Measurement of chlorophyll content in leaves. The pigments were extracted with 80% acetone from the leaf cutting after the CFI and reflection spectrum were measured for it. The optical spectrum of the extract absorption was measured with a Cary-50 spectrophotometer (Varian, USA). The concentrations of pigments in the extract were determined as described in [39]. Contents of Chl and carotenoids (Car) in the leaf were calculated per unit of leaf surface (nmol/cm2).
RESULTS AND DISCUSSION
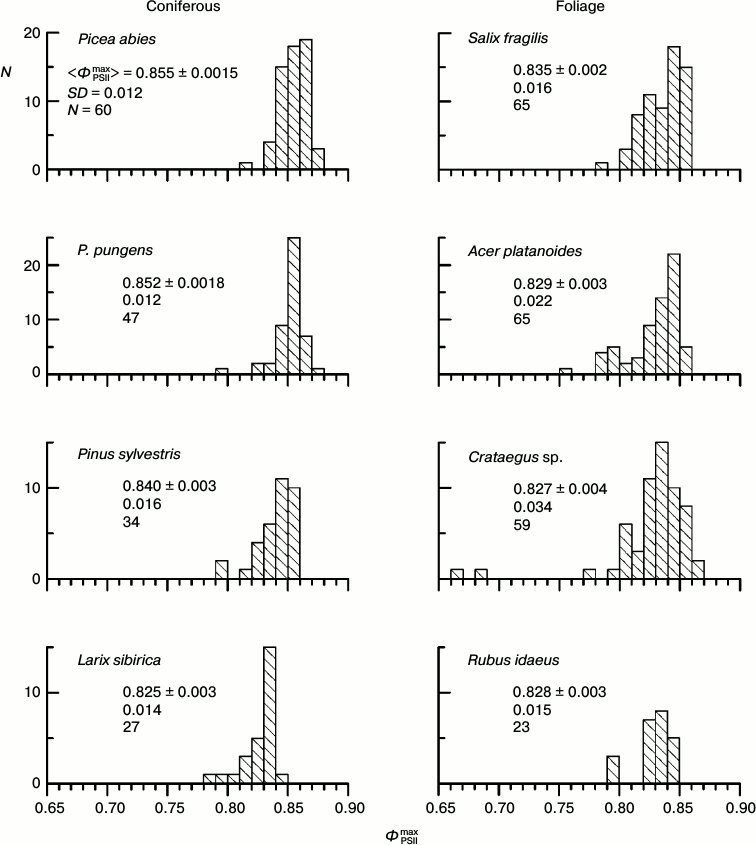
Histograms of distribution of parameter ΦPSIImax for leaves of some arboreous plants in summer. The physiological state of the leaf PSA and fluorescent parameters of the leaves depend on conditions of the plant growth and of the CFI measurements: temperature, illumination, humidity, etc. The parameter ΦPSIImax is one of the most methodically suitable and stable parameters of the CFI. It is thought that in the majority of higher plants the ΦPSIImax value is within a narrow range close to 0.83 (0.78-0.87 for different species) [40, 41]. Histograms of the ΦPSIImax distribution in the leaves of some arboreous plants of the temperate climatic zone of Russia in summer (August 2009-2013) are presented in Fig. 2. In all the studied plants, the distribution maximum occurs within this range, but the distribution of the ΦPSIImax value always occupies a rather wide region, approximately from 0.79 to 0.86. Note that the results of measurements that are presented in each diagram in Fig. 2 were performed on neighboring leaves of the same plant, i.e. situated under virtually the same conditions of illumination, temperature, humidity, etc. In other words, the ΦPSIImax variability noticeable in Fig. 2 is not associated with differences between the illuminated and shadowed leaves, age of the sprouts, their positions in the crown, etc.
Fig. 2. Histograms of distribution of parameter ΦPSIImax in leaves of coniferous and foliage plants of the temperate climatic zone from results of measurements in August. In each histogram the mean value of ΦPSIImax and its standard error, the standard deviation of the ΦPSIImax value, and the number of measurements are given.
What brings about the variability of parameter ΦPSIImax, and which factor could be responsible for this variability? The detection of such a factor would be very favorable for increasing the uniformity of the material chosen for investigation. As a working hypothesis, we supposed that in the case of the absence of significant differences in conditions of development and functioning of the PSA of different leaves, their different photosynthetic activity should be caused by a non-uniform degradation of the PSA with age in different leaves. Therefore, we chose for studies preferentially two groups of plants: the first group consisted of plants in late autumn (October 2013) when the PSA degradation is expressed in the majority of leaves, and its variations are the strongest in different leaves, whereas the second group was represented by plants in late summer (August – early September 2009-2013) when the autumnal degradation of the PSA is only beginning in the majority of the leaves.
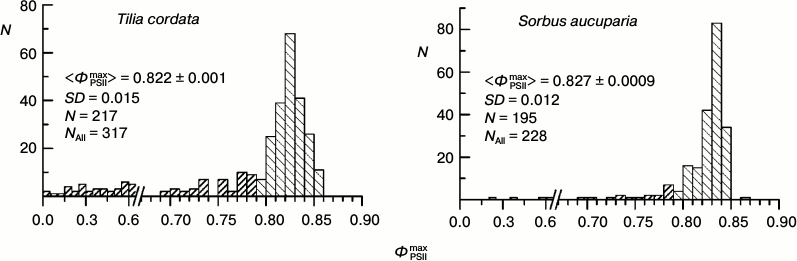
Our main investigations were performed on leaves of the rowan tree (Sorbus aucuparia L.) and the small-leaved linden (Tilia cordata Mill.). We studied characteristics of the CFI and some other biophysical parameters of these plants and their reaction to pollution of the environment earlier [29]. Figure 3 presents histograms of the ΦPSIImax value distribution for these two species measured in August – early September. As differentiated from Fig. 2, in Fig. 3 the data for the wider time interval are presented, which include leaves already beginning to turn yellow. The yellow leaves are characterized by relatively low ΦPSIImax values, which is clearly noticeable by presence of a “tail” from the side of low ΦPSIImax values in the histograms in Fig. 3. However, the leaves without yellowing even during this autumnal period displayed virtually the same values of ΦPSIImax as in summer (compare with Fig. 2).
Fig. 3. Histograms of distribution of maximal PSII efficiency, ΦPSIImax, in leaves of arboreous plants of the temperate climatic zone based on data of measurements during August–September: small-leaved linden (Tilia cordata Mill.) and rowan tree (Sorbus aucuparia L.). Columns with dense hatching show data for leaves beginning to turn yellow. In each histogram the mean value of ΦPSIImax and its standard error, the standard deviation of the ΦPSIImax value, the number of measurements on green leaves (N), and the total number of measurements (NAll) are given.
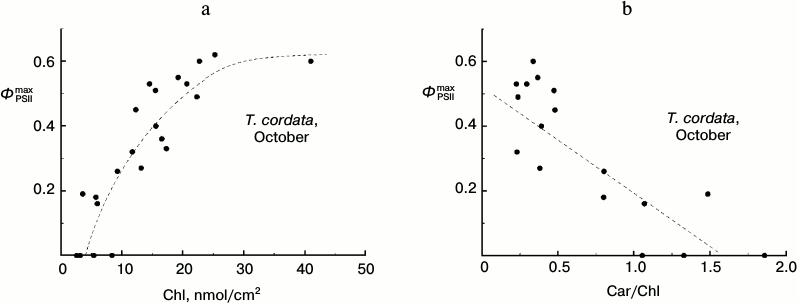
Dependence of ΦPSIImax on chlorophyll content in T. cordata leaves in autumn. Because the degradation of Chl is the main easily observable manifestation of the autumnal senescence of leaves, we studied the correlations of some parameters of CFI and Chl content in the leaves. We found, in particular, that in late summer leaves of the small-leaved linden (T. cordata) the parameter ΦPSIImax decreased with the decrease in Chl content in the leaf and, vice versa, this decrease in ΦPSIImax value corresponded with the relative growth of the Car content calculated per Chl (Fig. 4).
Fig. 4. Dependence of parameter ΦPSIImax on Chl content (a) and Car/Chl relation (b) in leaves of small-leaved linden based on data of measurements during the period of the mass yellowing of leaves (October 2013).
Parameter ΦPSIImax and color of T. cordata leaves in autumn. Based on the above-presented data, we supposed that during the late autumn the degree of Chl degradation in the leaves and the decrease in their PSII efficiency (decrease in parameter ΦPSIImax) should equally reflect both leaf senescence and accompanying suppression of photosynthetic reactions. This result can be practically important, because Chl degradation in a leaf and accompanying growth of Car/Chl ratio lead to changes in the directly (visually) observable optical properties of the leaf, first of all its color. Therefore, a researcher can preliminarily choose leaves at different senescence stages and possessing different activities of PSA using a simple visual assessment.
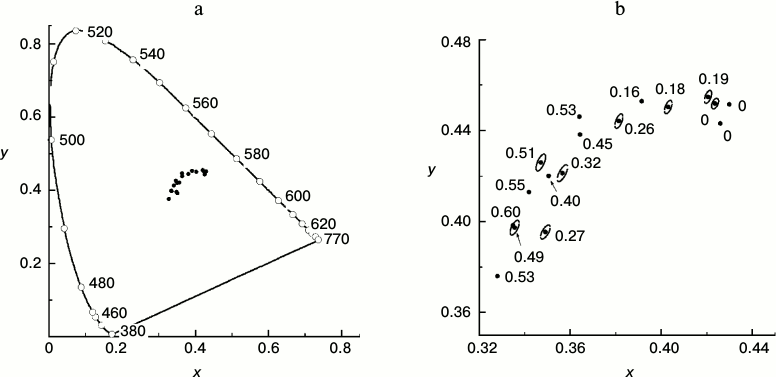
To what extent can expert visual assessment serve a quantitative estimation of leaf state? To answer this question, we measured the reflection spectra of leaves with different values of the ΦPSIImax parameter. We quantitatively described the hues of the leaves using the two-dimensional color scale proposed by the CIE in 1931. In other words, each color is represented by two numbers called chromaticity coordinates (x, y) that change within the range 0-1. The so-called CIE 1931 color space chromaticity diagram (Fig. 5) represents a two-dimensional color space where each color characterized by its color tone and saturation is shown by a single point. The most saturated spectral colors (i.e. colors created by monochromatic light of the visible spectrum) form a horseshoe-shaped arc. All non-monochromatic colors with saturation lower than unity are shown by points inside the “horseshoe” of spectral colors. Coordinates of the leaf chromaticity calculated basing on results of our measurements are shown in the CIE chromaticity diagram (Fig. 5).
Fig. 5. a) Points characterizing the color of a series of yellowing leaves of small-leaved linden in the CIE 1931 chromaticity diagram based on data of measurements during the period of mass yellowing of the leaves (October 2013). The continuous line shows the chromaticity line of spectral and purple colors. The numbers near the points on the line show the monochromatic light wavelength inducing the color sensation characterized by the chromaticity coordinates of the corresponding point in the plot for a standard colorimetric observer. b) Magnified fragment of the CIE chromaticity diagram representing the points characterizing the color of a series of yellowing leaves of small-leaved linden. Near each point the ΦPSIImax value is shown for the given leaf. Ellipses around some points show thresholds of the color discrimination for an average observer by MacAdam’s data [43].
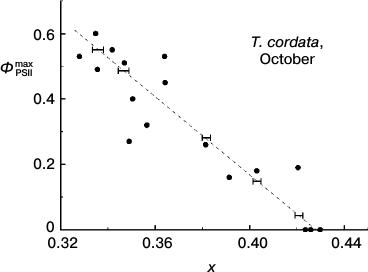
Quantitative description of hues of T. cordata leaves. Figure 5a shows positions of the chromaticity coordinates of leaves in the full-range chromaticity diagram relative to the line of pure spectral colors, and in Fig. 5b a magnified region of their compact localization is shown. The points in the lower left angle of the field presented in Fig. 5b correspond to greener nuances, the points in the upper right angle correspond to yellow nuances, and the whole diagonal through the chromaticity points of the leaves corresponds to yellow–green tones. Thus, each point corresponds to one leaf, its coordinates (x, y) in the chromaticity space characterize the hue of the leaf, and the number near each point indicates the ΦPSIImax value in the leaf. The efficiency of PSII of the leaf, ΦPSIImax, noticeably decreases with increase in the yellow hue. The next figure, Fig. 6, demonstrates that the ΦPSIImax value falls virtually linearly on movement along the diagonal in Fig. 5b from the green leaves to the yellow ones (that corresponds to increase in chromaticity coordinate x). Moreover, Fig. 7 also shows why the choice of the visually assessed color coordinate x (and also of color characteristic in general) of a leaf is a reasonable measure of the ratio between the contents of yellow carotenoids and green chlorophylls: the increase in the Car/Chl ratio also leads to nearly linear growth of the color coordinate x. However, this tendency obviously is observed only as a trend, whereas the greater values of ΦPSIImax sometimes correspond to yellower leaves than their “neighbors” in the chromaticity diagram with lower ΦPSIImax values. Moreover, points with noticeably different ΦPSIImax values occur not far from one another, e.g. ΦPSIImax = 60 and 49 or 51, 40, 32. Is it possible to discriminate these colors “by eye” and, consequently, conclude about the difference in the photosynthetic activity of these leaves?
Fig. 6. Correlation of maximal efficiency of PSII, ΦPSIImax, and small-leaved linden leaf chromaticity (coordinate x in the two-dimensional CIE 1931 color space) based on data of measurements during the period of mass yellowing of leaves (October 2013). Horizontal strokes indicate characteristic thresholds of color discrimination (correspond to dimensions of ellipses in Fig. 5b along the x axis).
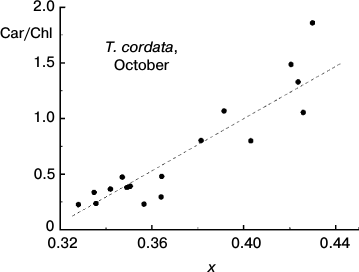
Fig. 7. Correlation between the Car/Chl ratio and small-leaved linden leaf chromaticity (coordinate x in the two-dimensional CIE 1931 color space) based on data of measurements during the period of mass yellowing of leaves (October 2013).
Possibility of visual expert assessment of leaf photosynthetic activity. The question about the threshold discrimination of colors throughout the whole region of non-spectral colors was studied in detail in works of MacAdam [42, 43]. Regions of color space virtually undistinguishable from the given color for an average observer according to MacAdam are shown in Fig. 5b as ellipses around the points. The eye-perceptible threshold changes in color are different for different color tones or saturations (this is shown by different dimensions and shapes of the elliptic “undistinguishable areas” in different regions of the chromaticity plot). In this case it is determined as a non-uniformity of the CIE chromaticity space metric. However, it is not very great in the region of our interest. Therefore, in order to avoid the figure overloading, the elliptic “undistinguishable areas” are shown only around some points in Fig. 5b. This figure shows that the typical size of the ellipse along the x axis is about 0.004, i.e. the minimal difference perceptible by the average observer between the colors in this region (located mainly along the dotted line) is Δx = 0.002. The plot in Fig. 6 shows that on the average abatement of ΦPSIImax observed with a decrease in the color coordinate x, this minimal distance between the distinguishable points is ΦPSIImax ≈ 0.017. The change in ΦPSIImax by 0.017 is rather small (thus, from the histograms of ΦPSIImax value distributions for leaves of different plant species in summer presented in Fig. 2, i.e. for the most uniform specimens, the characteristic range of changes in ΦPSIImax is about 0.05, i.e. virtually threefold higher than 0.017). This indicates that the visual diagnosis of differences in ΦPSIImax value of leaves during the autumnal degradation of Chl and the loss of PSA activity is realizable.
On the other hand, Fig. 5b also shows that leaves with rather pronounced differences in ΦPSIImax value (~0.1) also may have undistinguishable colors. In other words, the “noise” in Figs. 4 and 6 (i.e scattering of points around the smooth curves) limits possibilities of visual assessment of the photosynthetic activity in the series of leaves.
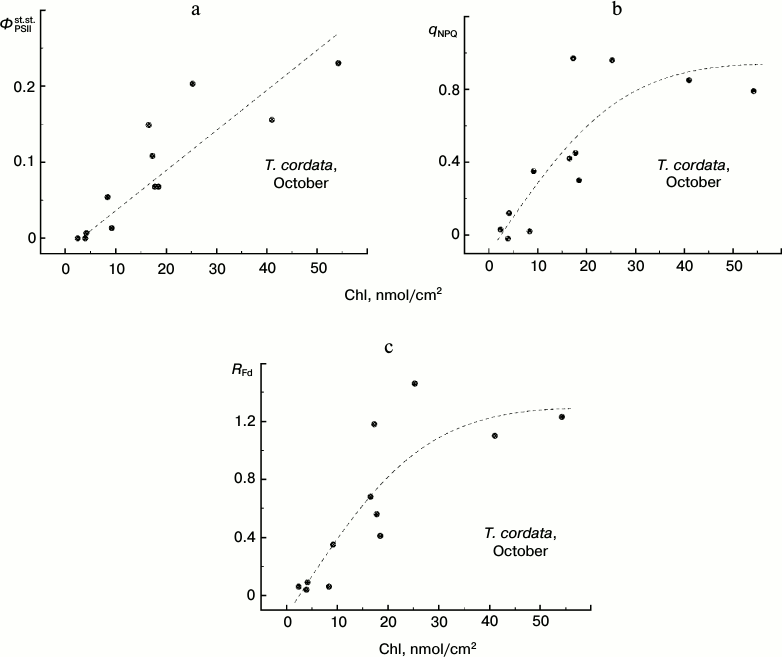
Chlorophyll content and steady-state characteristics of CFI of T. cordata leaves in autumn. Up to now we have considered only the maximal efficiency of the photochemical reactions in PSII, ΦPSIImax. This characteristic of photosynthetic activity of a leaf represents the most methodically convenient approach to express assessment of the PSA functioning (because for such assessment no plant adaptation is required for the strictly determined conditions of illumination). However, in some cases the CFI parameters reflecting the steady-state conditions of the leaves under illumination are more informative characteristics of the PSA. Such parameters frequently used in the literature [12, 18, 23, 26, 27, 29, 41, 44-45] include the PSII steady-state operating efficiency ΦPSIIst.st., the non-photochemical quenching coefficient qNPQ, and the vitality index RFd. In Fig. 8 we present the dependences of these parameters that we obtained under steady-state conditions of illumination of leaves at 100 µE⋅m-2⋅s-1 on the Chl content in the leaves.
Fig. 8. Dependence of steady-state (under conditions of light adaptation) CFI parameters (ΦPSIIst.st. (a), qNPQ (b), and RFd (c)) on the chlorophyll content in leaves of small-leaved linden by data of measurements during the period of mass yellowing of leaves (October 2013).
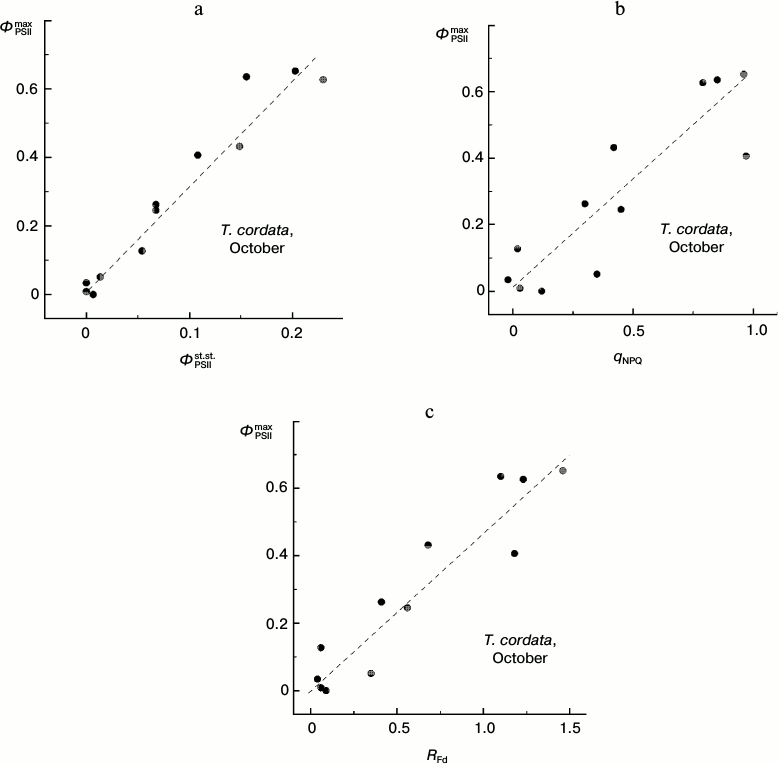
Attention should be paid to the character of correlations between changes in different CFI parameters associated with the degradation of Chl in autumnal leaves (Fig. 9). Despite a significant scattering of individual points, it is obvious that virtually any change in the steady-state parameters corresponds with a noticeable change in the ΦPSIImax value (i.e. there is no pronounced horizontal region in the plots presented in Fig. 9). This feature discriminates the autumnal leaves from observations on the summer leaves (see below).
Fig. 9. Correlation of the PSII maximal efficiency value, ΦPSIImax, and the steady-state (under conditions of light adaptation) CFI parameters (ΦPSIIst.st. (a), qNPQ (b), and RFd (c)) in leaves of small-leaved linden by data of measurements during the period of mass yellowing of leaves (October 2013).
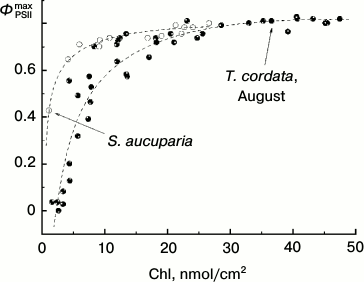
Dependence of ΦPSIImax on chlorophyll content in T. cordata leaves during late summer. Yellowing leaves can be found in many plants even in summer. However, such yellowing is generated under quite other conditions. Thus, by October the leaves retained on the plant have usually undergone a number of pronounced temperature minima (that usually does not occur in summer). This is associated with changes in enzymes of the violaxanthin cycle resulting in its “sticking” in the so-called “sustained NPQ” state [22]. Figure 10 shows the dependence of ΦPSIImax on the Chl content in the late summer leaves of linden and rowan tree. This dependence is obviously different from the similar dependence for the leaves collected in late autumn (in October) by a somewhat wider range of Chl concentrations when the ΦPSIImax value still remains virtually unchanged. A sharp drop in ΦPSIImax starts in linden at Chl concentrations lower than 17-20 nmol/cm2 and in rowan tree at still lower concentrations (lower than 5-7 nmol/cm2). These results are in agreement with data of other authors for other arboreous plants. Thus, in the western plane (Platanus occidentalis L.) the drop in ΦPSIImax = Fv/Fm begins with decrease in Chl content in the leaf below 10-15 nmol/cm2 [34].
Fig. 10. Dependence of parameter ΦPSIImax on chlorophyll content in leaves of small-leaved linden (black circles) and rowan tree (white circles) by data of measurements before the beginning of mass yellowing of leaves (August 2009-2013).
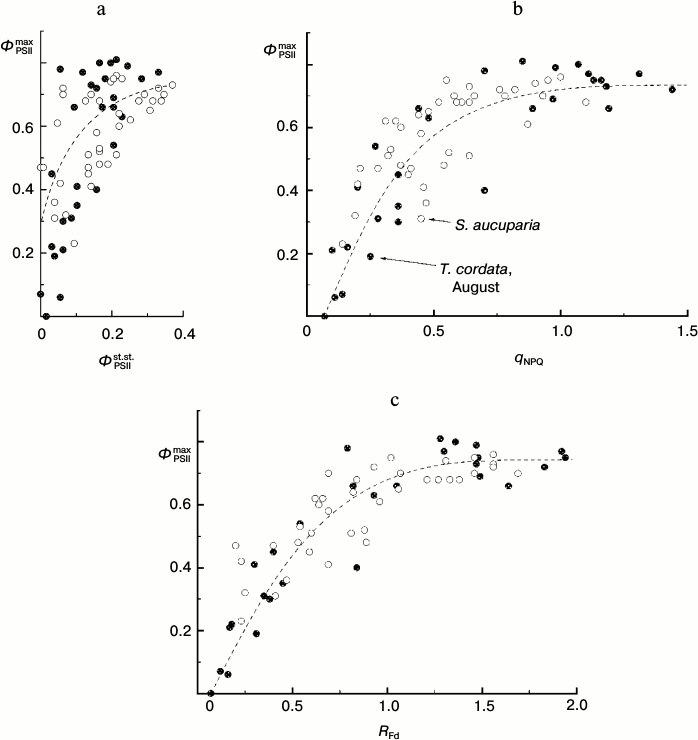
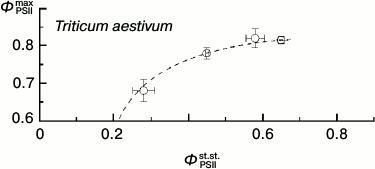
Thus, under these conditions the ΦPSIImax value is virtually insensible to destructive changes in the PSA of the leaves. And its drop only insignificantly reflects the drop in steady-state (light-adapted) characteristics of the PSA (Fig. 11). This is because ΦPSIImax reflects mainly the features of PSII itself and the antenna Chl-containing complex [46]. However, the CFI parameters characterizing the light-adaptation of the photosynthetic apparatus of the leaf change with leaf senescence significantly more noticeably. Thus, the ΦPSIIst.st. value corresponding to the mean quantum yield of the photochemical reaction in chloroplasts of the light-adapted leaf determines, first of all, the efficiency of using the reducing equivalent and ATP in reactions of carbon dioxide fixation. This value has to be significantly more sensitive to the leaf senescence [6, 32, 33]. Figure 11 shows the interrelations between changes in the ΦPSIImax and ΦPSIIst.st. values accompanying leaf senescence. It is obvious that even on a significant decrease in ΦPSIIst.st., the ΦPSIImax value still remains, on average, higher than 0.5-0.6. It is interesting to note that on decrease in the photosynthetic activity of the leaf (corresponding to decrease in ΦPSIIst.st. value), not only the mean value of ΦPSIImax decreases, but first of all its scattering significantly increases. This seems to indicate that the final “disassembling” of pigment–protein complexes of the photosynthetic apparatus of chloroplasts has different dynamics varying from leaf to leaf. A noticeable (up to twofold) decrease in other steady-state characteristics (non-photochemical quenching coefficient qNPQ, coefficient RFd) also is virtually not accompanied by a decrease in the ΦPSIImax value, and the ΦPSIImax value begins to fall only on more significant decrease in these steady-state parameters. This result is in accordance with the literature data obtained for both dicotyledonous (e.g. grape Parthenocissus tricuspidata Veitchii) and monocotyledonous plants (e.g. common wheat Triticum aestivum L.), as one can see in Fig. 12 (based on data presented in work [33]).
Fig. 11. Correlation of the value of parameter ΦPSIImax and steady-state (under conditions of light adaptation) CFI parameters (ΦPSIIst.st. (a), qNPQ (b), and RFd (c)) in leaves of small-leaved linden (black circles) and rowan tree (white circles) by data of measurements before the beginning of mass yellowing of leaves (August 2009-2013).
Fig. 12. Correlation of the values of ΦPSIImax and ΦPSIIst.st. in leaves of wheat (from data of work [33]).
Measurements of the CFI parameters allowed us to non-invasively assess the physiological state of a leaf: its photosynthetic activity and activity of the protection of the PSA against light-induced damage [3, 4, 6, 7, 26, 47]. The parameter ΦPSIImax that characterizes the maximal quantum efficiency of PSII functioning is one of the most frequently used characteristics of a leaf. The parameter ΦPSIImax is sometimes considered as the best approach for evaluation of the stress-activated decrease in PSA activity [22]. Nevertheless, in some cases (for leaves of arboreous plants not undergoing stress in summer) we can see that the steady-state characteristics of CFI (such as the PSII “operating efficiency” ΦPSIIst.st., non-photochemical quenching coefficient qNPQ, and the vitality index RFd) are more adequate indicators of the decrease in PSA activity as a result of destructive processes in the leaf, whereas the ΦPSIImax value remains virtually insensitive to them. Our data (compare Figs. 9 and 11) show that for deciduous arboreous plants of the temperate climatic zone the parameter ΦPSIImax is a characteristic sensitive to the PSA state of the leaf during the late autumn (i.e. during active leaf fall). However, before the beginning of the mass yellowing of leaves (late August – early September) such steady-state characteristics as ΦPSIIst.st., qNPQ, and RFd seem to be preferential for express-assessment of PSA state.
We think that one of the most interesting results is the following. We have established a correlation between the Fv/Fm value characterizing the maximal photochemical activity of PSII and the “chromaticity coordinates” of a leaf characterizing its color. The chromaticity coordinates determined by the light reflection spectra quantitatively characterize the leaves hue, and this creates preconditions for a visual “expert assessment” of the physiological state of the leaves. A similar qualitative assessment every researcher has to perform for elimination of a “bad” and choosing a “good” material for his studies, but, as far as we know, the limits of possibilities of such expert assessment have not been analyzed quantitatively up to now. Our data show that the human eye can be used as a sufficiently effective qualitative “analyzer” of ΦPSIImax because the ΦPSIImax value correlates with the Chl content (more accurately, with the Chl/Car ratio) in the leaves of the studied plants. The limiting resolving capacity of the human eye (assessed by us on suggestion of a high correlation between ΦPSIImax and the relative content of Chl) concerning the ΦPSIImax value for the autumnal leaves of T. cordata is about 0.017. But the real resolution ability seems to be not higher than 0.1. However, to what extent these preliminary results can be extrapolated to other species of arboreous plants and other seasons of the active photosynthetic period of plants remains a problem for further studies.
This work is dedicated to the 100 anniversary of the birth of Academician Alexander Abramovich Krasnovsky whose pioneer works in chlorophyll photochemistry [48, 49], as well as the Russian scientific school created by him [3, 4, 19, 20, 25, 47, 50], have significantly determined achievements of Russian science in the field of biochemistry and biophysics of photosynthesis.
We are grateful to Prof. A. M. Chernorizov for his valuable recommendations.
This work was partially supported by the Russian Foundation for Basic Research (projects 12-04-31162 and 12-04-01267a).
REFERENCES
1.Venediktov, P. S., Volgin, S. L., Kazimirko, Y. V.,
Krendeleva, T. E., Kukarskikh, G. P., Makarova, V. V., Lavrukhina, O.
G., Pogosyan, S. I., Yakovleva, O. V., and Rubin, A. B. (1999)
Biofizika, 44, 1037-1047.
2.Rubin, A. B. (2005) Tekhnol. Zhivykh Sistem,
2, 47-68.
3.Karapetyan, N. V., and Bukhov, N. G. (1986)
Soviet Plant Physiol., 33, 1013-1026.
4.Kalaji, H. M., Goltsev, V., Bosa, K.,
Allakhverdiev, S. I., and Strasser, R. J. (2012) Photosynth.
Res., 114, 69-96.
5.Rabinowitch, E. I. (1951) Photosynthesis and
Related Processes, Vol. II, Interscience Publishers, Inc., New
York-London.
6.Govindjee (1995) Austral. J. Plant Physiol.,
22, 131-160.
7.Adams, W. W., III, and Demmig-Adams, B. (2004) in
Chlorophyll and Fluorescence, Springer, pp. 583-604.
8.Maxwell, K., and Johnson, G. N. (2000) J. Exp.
Bot., 51, 659-668.
9.Baker, N. R. (2008) Annu. Rev. Plant Biol.,
59, 89-113.
10.Ptushenko, V. V., Ptushenko, E. A.,
Samoilova, O. P., and Tikhonov, A. N. (2013) Biosystems,
114, 85-97.
11.Samoilova, O. P., Ptushenko, V. V., Kuvykin, I.
V., Kiselev, S. A., Ptushenko, O. S., and Tikhonov, A. N. (2011)
Biosystems, 105, 41-48.
12.Liu, Y. F., Qi, M. F., and Li, T. L. (2012)
Plant Sci., 196, 8-17.
13.Buonasera, K., Lambreva, M., Rea, G.,
Touloupakis, E., and Giardi, M. T. (2011) Anal. Bioanal. Chem.,
401, 1139-1151.
14.Kautsky, H., and Hirsch, A. (1931)
Naturwissenschaften, 19, 964.
15.Schreiber, U., Schliwa, U., and Bilger, W. (1986)
Photosynth. Res., 10, 51-62.
16.Kirzhanov, D. V., and Kukushkin, A. K. (2009)
Vestnik Mosk. Univer. Ser. 3. Fiz. Astron.,
6, 65-69.
17.Tikhonov, A. N. (2013) Photosynth. Res.,
116, 511-534.
18.Bilger, W., and Bjorkman, O. (1994)
Planta, 193, 238-246.
19.Krasnovsky, A. A., Jr. (2007) Biochemistry
(Moscow), 72, 1065-1080.
20.Krasnovsky, A. A., Jr. (1998) Membr. Cell
Biol., 12, 665-690.
21.Krieger-Liszkay, A. (2005) J. Exp. Bot.,
56, 337-346.
22.Demmig-Adams, B., Cohu, C. M., Muller, O., and
Adams, W. W., III (2012) Photosynth. Res., 113,
75-88.
23.Lambrev, P. H., Miloslavina, Y., Jahns, P., and
Holzwarth, A. R. (2012) Biochim. Biophys. Acta –
Bioenerg., 1817, 760-769.
24.Muller, P., Li, X.-P., and Niyogi, K. K. (2001)
Plant Physiol., 125, 1558-1566.
25.Heber, U., Lange, O. L., and Shuvalov, V. A.
(2006) J. Exp. Bot., 57, 1211-1223.
26.Lichtenthaler, H. K., Buschmann, C., Rinderle,
U., and Schmuck, G. (1986) Radiat. Environ. Biophys., 25,
297-308.
27.Lichtenthaler, H. K., Buschmann, C., and Knapp,
M. (2005) Photosynthetica, 43, 379-393.
28.Karavaev, V. A., Polyakova, I. B., Solntsev, M.
K., and Yurina, T. P. (1998) J. Luminescence, 76,
335-338.
29.Ptushenko, V. V., Karavaev, V. A., Solntsev, M.
K., and Tikhonov, A. N. (2013) Biophysics (Moscow), 58,
228-233.
30.Strasser, R. J., Tsimilli-Michael, M., and
Srivastava, A. (2004) in Chlorophyll and Fluorescence, Springer,
pp. 321-362.
31.Rohacek, K. (2002) Photosynthetica,
40, 13-29.
32.Lichtenthaler, H. K. (1987) J. Plant
Physiol., 131, 101-110.
33.Lu, C., and Zhang, J. (1998) Physiol.
Plant., 104, 239-247.
34.Adams, W. W., Winter, K., Schreiber, U., and
Schramel, P. (1990) Plant Physiol., 92, 1184-1190.
35.Merzlyak, M. N., Solovchenko, A. E., and
Gitelson, A. A. (2003) Postharvest Biol. Technol., 27,
197-211.
36.Merzlyak, M. N., Solovchenko, A. E., Smagin, A.
I., and Gitelson, A. A. (2005) J. Plant Physiol., 162,
151-160.
37.Solovchenko, A. (2010) Photoprotection in
Plants: Optical Screening-Based Mechanisms, Vol. 14, Springer.
38.Djudd, D., and Vishecky, G. (1975) Color in
Business, Science and Industry, John Wiley & Sons, New
York-London-Sydney-Toronto.
39.Lichtenthaler, H. K. (1987) Methods
Enzymol., 148, 350-382.
40.Bjorkman, O., and Demmig, B. (1987)
Planta, 170, 489-504.
41.Johnson, G. N., Young, A. J., Scholes, J. D., and
Horton, P. (1993) Plant Cell Environ., 16, 673-679.
42.MacAdam, D. L. (1942) J. Opt. Soc. Am.,
32, 247-273.
43.MacAdam, D. L. (1943) J. Opt. Soc. Am.,
33, 18-26.
44.Genty, B., Briantais, J.-M., and Baker, N. R.
(1989) Biochim. Biophys. Acta, 990, 87-92.
45.Fryer, M. J., Andrews, J. R., Oxborough, K.,
Blowers, D. A., and Baker, N. R. (1998) Plant Physiol.,
116, 571-580.
46.Lazar, D. (2003) J. Theor. Biol.,
220, 469-503.
47.Zivcak, M., Brestic, M., Balatova, Z.,
Drevenakova, P., Olsovska, K., Kalaji, H. M., Yang, X., and
Allakhverdiev, S. I. (2013) Photosynth. Res., 117,
529-546.
48.Krasnovsky, A. A., and Brin, G. P. (1947)
Dokl. Akad. Nauk SSSR, 58, 1087-1090.
49.Krasnovsky, A. A. (1948) Dokl. Akad. Nauk
SSSR, 61, 91-94.
50.Shuvalov, V. A., and Heber, U. (2003) Chem.
Phys., 294, 227-237.