The Size of the Light-Harvesting Antenna of Higher Plant Photosystem II Is Regulated by Illumination Intensity through Transcription of Antenna Protein Genes
M. M. Borisova-Mubarakshina*, D. V. Vetoshkina, N. N. Rudenko, G. N. Shirshikova, T. P. Fedorchuk, I. A. Naydov, and B. N. Ivanov
Institute of Basic Biological Problems, Russian Academy of Sciences, 142290 Pushchino, Moscow Region, Russia; E-mail: mubarakshinamm@gmail.com; ivboni@rambler.ru* To whom correspondence should be addressed.
Received December 4, 2013; Revision received January 9, 2014
In arabidopsis plants, with an increase in illumination intensity during growth the extent of reduction of the plastoquinone pool in the photosynthetic electron transport chain increased, whereas the effective quantum yield of photosynthesis decreased. After 5 days of growth under high illumination intensity, these parameters in high light returned to values observed in “shade-adapted” plants in low light. During the same period, the size of the antenna decreased, correlating with a decrease in the amounts of proteins of peripheral pigment–protein complexes. It was found that the decrease in the amounts of these proteins occurred due to suppression of transcription of their genes.
KEY WORDS: photosynthesis, adaptation to illumination intensity, plastoquinone pool, light harvesting antenna, gene expressionDOI: 10.1134/S0006297914060042
Abbreviations: Chl, chlorophyll; LHCII, the main light-harvesting complex of photosystem II; P680, electron donor in the reaction center of photosystem II; PAR, photosynthetically active radiation; PETC, photosynthetic electron transport chain; Pheo, pheophytin, the electron acceptor in photosystem II; PSI and PSII, photosystems I and II; qRT-PCR, real-time reverse transcription polymerase chain reaction; Rubisco, ribulose-1,5-bisphosphate carboxylase/oxygenase.
The intensity of the incident light defines many morphological and
functional properties of plants, first of all, properties of their
photosynthetic apparatus [1-3].
It is established that the size of the light-harvesting antenna
decreases in high light [2]. It is believed that
this decrease helps to avoid photodestruction, one cause of which in
high light is the increase in charge recombination frequency of
ion-radical pair P680+ Pheo–, and
appearance of triplet chlorophyll molecules that may react with triplet
oxygen to produce highly reactive singlet oxygen.
Light-harvesting antenna of PSII includes proteins of the core-complex CP47 and CP43, immediately adjacent to core-complex the monomeric antenna proteins Lhcb4 (CP29), Lhcb5 (CP26) and Lhcb6 (CP24), and homo- and heterotrimeric complexes of Lhcb1-3 proteins linked to the last ones with a varying degree of strength [4]. These trimers are usually denoted as light harvesting complex II (LHCII). Different pigment–protein complexes of the photosynthetic apparatus have characteristic pigment compositions. Core-complex contains only chlorophyll a, whereas peripheral pigment–protein complexes of the antenna contain both chlorophylls a and b in different ratio; it is noteworthy that the chlorophyll a/b ratio in the LHCII complex is close to 1.
The decrease in antenna size in arabidopsis plants under increase in illumination during growth may be the result of a decrease in the amount of almost any of the proteins of peripheral pigment–protein complexes [3, 5]. However, it was unknown until now at which stage, namely, transcription or translation, the biosynthesis of proteins of these complexes is regulated in response to illumination intensity changes. Data obtained in a recent study with barley mutant viridis zb63 lacking PSI [6] implied that during adaptation to increased illumination the biosynthesis of proteins of LHCII is suppressed at the stage of translation. In the present study, we found that in the course of adaptation during 5 days after transition from low- to high-light conditions, the decrease in the amounts of proteins of the peripheral light-harvesting antenna is caused by suppression of transcription of their genes. We also show that the quantum yield of photosynthesis, being decreased initially after the transition, is restored.
MATERIALS AND METHODS
Arabidopsis thaliana plants (ecotype Columbia) were grown in a climate chamber for 28 days at 19°C, light intensity 70 μmol photons·m–2·s–1 PAR, and 8-h light day. Afterwards some plants were transferred at the same light intensity with 16-h day. After 10 days, some of these plants were transferred to the high light conditions, 360 μmol photons·m–2·s–1 PAR with 16-h day. Chlorophyll (Chl) a fluorescence in leaves was measured using PAM-fluorometers PAM-101 and MiniPAM (Walz, Germany). Quantum yield (Y) and the coefficient of non-photochemical quenching of Chl fluorescence (qN) were calculated using the conventional formulas: Y = (F′m – Fs)/F′m and qN = (Fm – F′m)/(Fm – F0), where Fm is the maximum fluorescence yield in the dark-adapted leaves in response to a saturating pulse, Fs the steady-state fluorescence yield in the actinic light, F′m is the maximum fluorescence yield in response to a saturating pulse during steady-state illumination with the actinic light, and F0 is the fluorescence yield in leaves adapted during 20 min in the dark prior to illumination. Relative extent of plastoquinone pool reduction in leaves under illumination was assessed as 1 – (F′m – Fs)/(F′m – F′0) [7], where F′0 is the minimum yield of Chl fluorescence promptly after cessation of illumination. Gene expression levels were measured using qRT-PCR on the 5th day of plant growth under different light conditions. RNA was extracted using the BioRad Aurum total RNA Mini Kit and a standard protocol, and then 400 ng of each RNA sample was used in the reverse transcription procedure using the BioRad iScript Reverse Transcription Supermix reagents. The cDNA and specific primers [8] for gene At2g05070, encoding Lhcb2.2 (one form of Lhcb2), and genes At5g54270 and At1g15820, encoding Lhcb3 and Lhcb6, were used in qRT-PCR with a iQ5 real-time thermal cycler (BioRad) and reagents by Evrogen. Primers for the actin 7 gene, At5g09810 (forward 5′-GAAGGCTGGTTTTGCTGGTGAT-3′ and reverse 5′-CCATGTCATCCCAGTTACTTACAATACC-3′), were used as a positive control. Thylakoids were isolated as described earlier [9]. Denaturing electrophoresis was performed in gradient (12-18%) polyacrylamide gel using Tris-Tricine buffers [10]. Proteins were visualized by staining of gels with Coomassie. Chlorophyll content was determined in 95% ethanol extracts [11]. PAR was measured in the 400-700 nm range using an LI-190SA Quantum Sensor.
RESULTS AND DISCUSSION
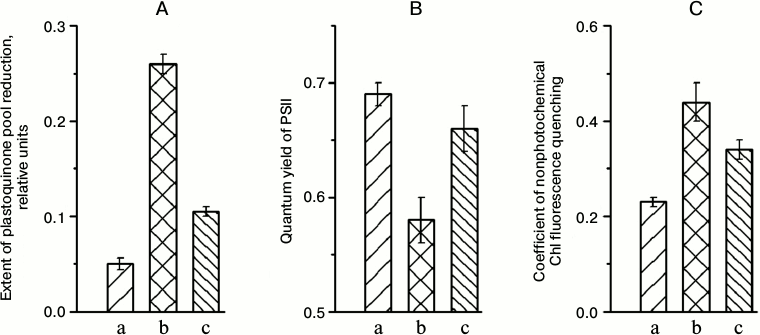
The extent of plastoquinone pool reduction in plants grown under low light (“shade-adapted” plants) was substantially higher under illumination with higher light than under illumination with light applied during growth (Fig. 1A, b vs. a). The increased extent of plastoquinone pool reduction can occur if the electron carriers following this pool in the “shade-adapted” PETC, as well as the “shade-adapted” carbon metabolism, have not enough capacities to provide in higher light the free transfer of all electrons entering the PETC from PSII equipped with “shade-adapted” light-harvesting antenna. After 5 days of adaptation to higher illumination, the relative extent of the plastoquinone pool reduction in this high light was much lower (Fig. 1A, c).
Fig. 1. Relative extent of plastoquinone pool reduction (A), quantum yield of PSII (B), and coefficient of non-photochemical quenching of Chl a fluorescence (C) in leaves of A. thaliana in the course of adaptation to a change in illumination intensity: a) plants grown under 70 μmol photons·m–2·s–1 PAR and parameters were measured at the same light intensity; b) plants grown under 70 μmol photons·m–2·s–1 PAR and parameters were measured at 360 μmol photons·m–2·s–1 PAR; c) plants transferred from low light to 360 μmol photons·m–2·s–1 PAR where they grew for 5 days, and parameters were measured at the same high-light intensity.
During the first day after the plants were transferred from the low-light to the high-light conditions, the quantum yield of PSII under steady-state illumination (an effective quantum yield of photosynthetic electron transport) was substantially lower in high light than in low light (Fig. 1B, b vs. a). For the same transfer, non-photochemical quenching of chlorophyll fluorescence became appreciably higher (Fig. 1C), indicating an increase in thermal dissipation of light energy; that provides protection from photoinhibition at increased light intensity. After 5 days under high light, the light energy came to be used more effectively, as it was evident from both the higher quantum yield and the lower non-photochemical quenching (Figs. 1B (c) and 1C (c)). A similar trend in the change in the functional properties of the PETC under the change of illumination level was observed by Walters et al. [12].
In plants kept in the dark for 20 min prior to measurements, the quantum yield of PSII was 0.80 ± 0.01 in the plants before their transfer to the high illumination intensity, 0.79 ± 0.01 promptly after this transfer, and 0.81 ± 0.01 after 5 days of growth at the higher light intensity. This meant that the sharp decrease in the PSII quantum yield in high light, which was observed early after transfer of “shade-adapted” plants to this light, and its subsequent increase in the same light after 5 days growth under illumination with this light were not caused by the change in the capability of PSII reaction centers for charge separation. Rather, a change occurring during the adaptation to high illumination conditions of the characteristics of electron transfer in other segments of the PETC were responsible for the changes in both the quantum yield of PSII and the extent of plastoquinone pool reduction (Figs. 1A and 1B).
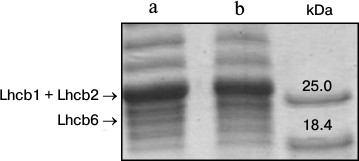
Lindahl et al. [13] showed that the size of light harvesting antenna of PSII in spinach leaves decreased after 3 days under increased illumination. In the present study, in 3-4 days after a transfer of arabidopsis plants to the high light, the Chl a/Chl b ratio increased on average from 2.0 to 2.4, indicating a decrease in the number of peripheral light harvesting complexes that contain Chl b, in other words, a reduction of the antenna size. To estimate the amount of peripheral antenna proteins, we conducted denaturing electrophoresis of thylakoid extracts. Densitometric analysis of the gels (Fig. 2) revealed that the density of bands corresponding to the sum of Lhcb1 and Lhcb2 proteins, and to Lhcb6 protein, in the plants grown for 5 days under 360 μmol photons·m–2·s–1 was 15% lower than in the plants that continued to grow under 70 μmol photons·m–2·s–1. This evidenced a decrease in the amount of the antenna protein during the adaptation to the high-light conditions, and it was in accordance with recently published data [5].
Fig. 2. Denaturing electrophoresis of proteins of thylakoids (8 μg Chl/ml) from leaves of 4-week-old A. thaliana plants grown for 5 days under different illumination intensity and at 16-h day. a, b) 70 and 360 μmol photons·m–2·s–1 PAR, respectively. Right-hand track, marker proteins.
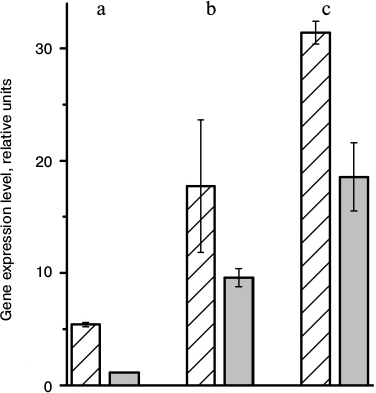
In 5 days after a transfer of arabidopsis plants to the higher illumination, a decrease in expression of genes At2g05070, At5g54270, and At1g15820 that encode antenna proteins Lhcb2.2, Lhcb3, and Lhcb6, respectively, was observed (Fig. 3). Accordingly, the decrease in the amount of these proteins under increase in illumination intensity is caused by lower inflow of newly synthesized molecules when spent molecules are disintegrated via a natural way. Thus, the regulation of the light-harvesting antenna size occurs at the stage of gene transcription of antenna proteins rather than at the stage of translation, as was earlier supposed [6].
Fig. 3. Expression level of genes At2g05070 (a), At5g54270 (b), and At1g115820 (c) encoding Lhcb2.2, Lhcb3, and Lhcb6, respectively, in leaves of A. thaliana plants grown under different illumination intensities. Hatched bars, 8-h day and 70 μmol photons·m–2·s–1; gray bars, 5 days after transfer to 16-h day and 360 μmol photons·m–2·s–1.
It has been assumed that the signal to change the size of antenna is the redox state of the plastoquinone pool [14, 15]. In the presented experiments, the extent of plastoquinone pool reduction increased considerably after transfer of plants to high illumination intensity (Fig. 1A), matching this assumption. To explain how the redox state of the plastoquinone pool situated within the thylakoid membranes can affect the protein biosynthesis system in cytoplasm (translation stage) or in nucleus, where the antenna proteins are encoded (transcription stage), we assumed earlier that the signal molecules are those H2O2 molecules that form in the thylakoid membrane in the reaction of plastohydroquinone with superoxide radicals generated in the PETC [16].
Processes initiated by the signal from the plastoquinone pool under high-light conditions led to antenna size decrease owing to decreased biosynthesis of antenna proteins, and accordingly to the lower incident light energy share feeding the reaction center of PSII. The resulting rate of charge separation together with increased amounts of both the electron carriers and Rubisco being synthesized under increased illumination intensity [17, 18] enabled relatively higher number of electrons to be transferred from PSII to other PETC segments; this was reflected in higher effective quantum yield of electron transfer (Fig. 1B, c).
Results of this work indicate that regulation of the size of the antenna, being a vital part of the mechanism of adaptation to high illumination intensity, which provides coordination of successive steps of electron transfer in the PETC and, thus more efficient use of light energy in photosynthesis, takes place at the stage of transcription of genes of peripheral antenna proteins.
This work was supported by the Russian Foundation for Basic Research (grant Nos. 12-04-31362 and 12-04-00320) and by the Ministry of Education and Science of the Russian Federation (grant No. 2012-1.2.2-12-000-1013-068).
REFERENCES
1.Krendeleva, T. E., Zamazova, L. P., and Tulbu, G.
V. (1975) Biol. Nauki, 9, 76-82.
2.Anderson, J. M. (1986) Annu. Rev. Plant
Physiol., 37, 93-136.
3.Bailey, S., Walters, R. G., Jansson, S., and
Horton, P. (2001) Planta, 213, 794-801.
4.Kochubey, S. M. (2001) The Organization of
Photosynthetic Apparatus of Higher Plants [in Russian], Alterpress,
Kiev.
5.Kouril, R., Wientjes, E., Bultema, J. B., Croce,
R., and Boekema, E. J. (2013) Biochim. Biophys. Acta,
1827, 411-419.
6.Frigerio, S., Campoli, C., Zorzan, S., Fantoni, L.
I., Crosatti, C., Drepper, F., Haehnel, W., Cattivelli, L.,
Morosinotto, T., and Bassi, R. (2007) J. Biol. Chem.,
282, 29457-29469.
7.Tullberg, A., Alexciev, K., Pfannschmidt, T., and
Allen, J. F. (2000) Plant Cell Physiol., 41,
1045-1054.
8.Xu, Y. H., Liu, R., Yan, L., Liu, Z. Q., Jiang, S.
C., Shen, Y. Y., Wang, X. F., and Zhang, D. P. (2012) J. Exp.
Botany, 63, 1095-1106.
9.Ignatova, L. K., Rudenko, N. N., Mudrik, V. A.,
Fedorchuk, T. P., and Ivanov, B. N. (2011) Photosynth. Res.,
110, 89-98.
10.Schagger, H., and von Jagow, G. (1987) Anal.
Biochem., 166, 368-379.
11.Lichtenthaler, H. K. (1987) Meth.
Enzymol., 148, 350-382.
12.Walters, R. G., Shephard, F., Rogers, J. J. M.,
Rolfe, S. A., and Horton, P. (2003) Plant Physiol., 131,
472-481.
13.Lindahl, M., Yang, D. H., and Andersson, B.
(1995) Eur. J. Biochem., 231, 503-509.
14.Escoubas, J. M., Lomas, M., LaRoche, J., and
Falkowski, P. G. (1995) Proc. Natl. Acad. Sci. USA, 92,
10237-10241.
15.Yang, D. H., Andersson, B., Aro, E.-M., and Ohad,
I. (2001) Photosynth. Res., 68, 163-174.
16.Mubarakshina, M. M., and Ivanov, B. N. (2010)
Physiol. Plant., 140, 103-110.
17.Shmeleva, V. L., and Ivanov, B. N. (1985)
Photosynthetica, 19, 402-411.
18.Chow, W. S., and Anderson, J. M. (1987) Austr.
J. Plant Physiol., 14, 1-8.