Analysis of Results of Acute Graft-Versus-Host Disease Prophylaxis with Donor Multipotent Mesenchymal Stromal Cells in Patients with Hemoblastoses after Allogeneic Bone Marrow Transplantation
I. N. Shipounova1, N. A. Petinati1, A. E. Bigildeev1, E. A. Zezina1,2, N. I. Drize1*, L. A. Kuzmina1, E. N. Parovichnikova1, and V. G. Savchenko1
1Hematological Scientific Center, Ministry of Health of Russian Federation, Novozykovsky Proezd 4, 125167 Moscow, Russia; E-mail: ndrize@yandex.ru; iranifontova@yandex.ru; loel@mail.ru; bigildeev.ae@mail.com; kuzlara@rambler.ru; elenap@blood.ru; svg@blood.ru2Lomonosov Moscow State University, Faculty of Biology, 119991 Moscow, Russia; E-mail: katerina.zezina@gmail.com
* To whom correspondence should be addressed.
Received July 3, 2014
Allogeneic bone marrow transplantation (allo-BMT) is currently the only way to cure many hematoproliferative disorders. However, allo-BMT use is limited by severe complications, the foremost being graft-versus-host disease (GVHD). Due to the lack of efficiency of the existing methods of GVHD prophylaxis, new methods are being actively explored, including the use of donors’ multipotent mesenchymal stromal cells (MMSC). In this work, we analyzed the results of acute GVHD (aGVHD) prophylaxis by means of MMSC injections after allo-BMT in patients with hematological malignancies. The study included 77 patients. They were randomized into two groups – those receiving standard prophylaxis of aGVHD and those who were additionally infused with MMSC derived from the bone marrow of hematopoietic stem cell donors. We found that the infusion of MMSC halves the incidence of aGVHD and increases the overall survival of patients. Four of 39 MMSC samples were ineffective for preventing aGVHD. Analysis of individual donor characteristics (gender, age, body mass index) and the MMSC properties of these donors (growth parameters, level of expression of 30 genes involved in proliferation, differentiation, and immunomodulation) revealed no significant difference between the MMSC that were effective or ineffective for preventing aGVHD. We used multiple logistic regression to establish a combination of features that characterize the most suitable MMSC samples for the prevention of aGVHD. A model predicting MMSC sample success for aGVHD prophylaxis was constructed. Significant model parameters were increased relative expression of the FGFR1 gene in combination with reduced expression levels of the PPARG and IGF1 genes. Depending on the chosen margin for probability of successful application of MMSC, this model correctly predicts the outcome of the use of MMSC in 82-94% of cases. The proposed model of prospective evaluation of the effectiveness of MMSC samples will enable prevention of the development of aGVHD in the maximal number of patients.
KEY WORDS: multipotent mesenchymal stromal cells (MMSC), allogeneic bone marrow transplantation (allo-BMT), acute graft-versus-host disease (aGVHD), relative gene expression level, multiple logistic regressionDOI: 10.1134/S0006297914120104
Allogeneic bone marrow transplantation (allo-BMT) is the only effective therapeutic strategy for curing many types of hematological disorders including leukemia [1]. Application of this method is restricted by severe complications driven by this procedure [2]. Acute graft-versus-host disease (aGVHD) is one of the main complications after allo-BMT [3]. It is the response of donor’s immune system to the recipient’s antigens [4]. With current methods of prophylaxis, 30-40% of patients develop aGVHD after allo-BMT from HLA-matched donors [5, 6]. The search for the donor is based on the match for at least six human leukocyte antigens (HLA). Stricter criteria would lead to decreased probability of finding an appropriate donor, so the search for more effective methods of aGVHD prophylaxis is an important task for clinicians performing allo-BMT.
Processes leading to the development of aGVHD begin before allo-BMT. The conditioning regimen applied prior to allo-BMT for the reduction of tumor burden, suppression of recipient’s immune system, and prevention of graft rejection also damages the recipient’s tissues [7]. This leads to activation of monocytes and tissue macrophages that secrete proinflammatory cytokines such as interleukin 1β (IL-1β) and interleukin 6 (IL-6) [8-10]. These cytokines activate the donor’s T cells, which in turn produce interleukin 2 (IL-2) and interferon γ (IFNγ) [9]. The release of the cytokines stimulates the expression of adhesion molecules and increases the number of MHC class 2 molecules presented on the surface of cells composing different tissues of the recipient [6, 11, 12], and thus favors the development of aGVHD.
Nevertheless, not all physiologic processes that follow allo-BMT lead to aGVHD, and some of them lead to its suppression [13, 14].
Recently, an increasing number of investigations addressing the possibility of using multipotent mesenchymal stromal cell (MMSC) infusions for the treatment and the prophylaxis of aGVHD is emerging. These bone marrow stromal cells can be cultivated in vitro. Their distinct features are the abilities to adhere to the plastic surface and differentiate upon corresponding induction [15]. MMSC secrete various cytokines, growth factors, and extracellular matrix molecules [16, 17]. These cells also have unique immunomodulating capacities [18]. Moreover, MMSC do not induce immune response, meaning that for clinical applications any donor’s cells could be used without checking histocompatibility [19, 20]. In vivo studies have demonstrated the possibility of MMSC application for the prevention of development and for the treatment of aGVHD. These findings allowed the application of MMSC in clinical practice for aGVHD treatment [21-23]. However, the exact mechanism of their immunomodulating effect is still obscure, and animal models have not yet provided unambiguous results. A few studies had shown high efficiency of MMSC for preventing aGVHD [24, 25].
Thus, features of MMSC might solve the main bone marrow transplantation problem, which is the search for an effective method for aGVHD prophylaxis.
MATERIALS AND METHODS
Patients. A prospective randomized study was initiated in October 2008. The study was approved by the Ethics Committee of the Hematology Research Center, Russian Ministry of Health. All patients and bone marrow donors signed informed consent to participate in the study.
To date, the study has included 77 patients, 37 women and 40 men, aged 17 to 63 years (median 34 years), who underwent allo-BMT from HLA-identical siblings in the Department of High-Dose Chemotherapy, Depression of Hematopoiesis, and Bone Marrow Transplantation, Hematology Research Center, in the period from October 2008 to April 2014. Allo-BMT was performed for each patient as a stage of leukemia treatment. Myeloablative conditioning that included cyclophosphamide combined with busulfan was administered to 43 patients. A low-intensity conditioning regimen was administered to 34 patients in remission and included fludarabine, busulfan, and anti-thymocytic globulin.
The patients were randomized into two groups of equivalent gender and age. Block randomization was performed on the day of allo-BMT. Patients in the first group received standard prophylaxis of aGVHD, and for a second group in addition to the standard prevention of aGVHD, MMSC derived from the corresponding hematopoietic stem cell donor was administered. The MMSC were infused intravenously at the moment of recovery in peripheral blood leukocytes to 109/liter. On average, 1.2 (0.9-1.65)·106 MMSC were injected per kilogram of the patient’s body weight. As standard aGVHD prophylaxis, patients received cyclosporin combined with methotrexate, and some received mycophenolate mofetil or prednisolone in addition.
Acute GVHD was graded according to internationally accepted criteria [26].
Donors. MMSC were isolated from bone marrow of 77 donors (35 female and 42 male) ranging in age from 13 to 59 years (median 34).
MMSC. MMSC were derived from 25-30 ml of bone marrow from donors. For separation of mononuclear cells, the bone marrow was mixed with an equal volume of alpha-MEM (ICN, USA) containing 0.2% methylcellulose (1500 cP; Sigma-Aldrich, USA). After 40 min, erythrocytes and granulocytes had mostly precipitated, while the mononuclear cells remained in suspension. The upper fraction (suspension) was collected and centrifuged for 10 min at 450g. The sediment was suspended in cultivation medium composed of alpha-MEM supplemented with 4% donor platelet lysate [27], 2 mM L-glutamine (ICN), 100 U/ml penicillin (Ferein, Russia), 50 µg/ml streptomycin (Ferein), and 2 U/ml heparin (Sigma). The cells were cultured at 27·106 cells per T175 cm2 culture flask (Corning-Costar, USA). When a confluent monolayer of cells formed, the cells were washed with 0.02% EDTA (ICN) in physiologic solution (Sigma-Aldrich) and then trypsinized (ICN). The cells were then seeded at 4·103 cells per cm2 of flask area. The cultures were maintained in hypoxic conditions at 37°C with 5% CO2 and 5% O2 in the atmosphere. MMSC were harvested in 6% dextran (trade name “Poliglukin”, produced by public corporation Biokhimik, Russia) and cryopreserved with 10% dimethylsulfoxide (ROTH, Germany) or diluted to (3-7)·106 cells/ml of 6% dextran and in such suspension were injected intravenously to the patients. Cryoconservation did not affect the immunomodulating properties of MMSC as demonstrated in the PhD thesis of N. A. Petinati (synopsis of thesis of N. A. Petinati “Prophylaxis of graft-versus-host disease for patients with hematological malignancies after allogeneic bone marrow transplantation with donor multipotent mesenchymal stromal cells”, Moscow, 2013).
The criteria for eligibility of MMSC to be clinically applied included spindle-shaped morphology, absence of visible clots, standard immunophenotype [28] for surface molecules [29], and proven ability to differentiate in vitro along osteogenic and adipogenic lineages [30].
Estimation of gene relative expression levels in MMSC. Total RNA was extracted from MMSC by a standard method with guanidine thiocyanate [31]. cDNA was synthesized in the reverse transcription reaction using a mixture of oligo(dT) primers and random hexamers. The gene expression level was quantified by real-time quantitative PCR using hydrolysis fluorescent probes (TaqMan; Thermo Scientific, USA) and a Rotor-Gene 6000 apparatus (Corbett Research, Australia). Gene-specific primers were designed by the authors and synthesized by Syntol (Russia). All primers and probes can be provided upon request. The relative gene expression level was determined by normalizing the expression of each target gene to that of BACT and GAPDH and was calculated using the ΔΔCt method [32] for each MMSC sample.
Analysis of ability of MMSC to inhibit lymphocyte proliferation. Peripheral blood from healthy donors was separated on Lymphoprep (density 1.077 g/cm3) (MP Biomedicals, USA). The resulting mononuclear cell fraction (PBMC) was washed twice with RPMI-1640 medium without serum and adjusted to concentration 5·107/ml. Then 3 µM fluorescent dye CFSE (Molecular Probes, USA) was added to the cells; the cells were incubated 10 min at 37°C and then washed twice in RPMI-1640 with 10% fetal calf serum. Lymphocyte blast transformation was induced by phytohemagglutinin (PHA). PHA was added to the cell suspension to final concentration of 5 µg/ml. The PBMC were plated at 2·105 per well in 96-well plates onto MMSC that had been seeded at concentration 103 cells per well 3 days earlier. PBMC with MMSC were incubated for 4 days at 37°C under 5% CO2. PBMC cultured without MMSC were used as controls. The relative number of proliferating lymphocytes cultured on MMSC was compared with control samples on day 4 by flow cytometry. Cells were stained with 7-AAD (Sigma) prior to the analysis. Lymphocytes after blast transformation were gated using forward and side light scattering. Dead cells were excluded by 7-AAD staining. Peaks corresponding to proliferating and nonproliferating cells were determined on the histograms built on the CFSE fluorescence channel. The proportion of non-dividing cells cultivated without MMSC was considered to be S. The proportion of non-dividing cells cultivated with MMSC was considered to be Sm. The ratio of Sm to S reflected the MMSC-induced inhibition of lymphocyte proliferation.
Statistical analysis. The data in the tables are presented as mean ± standard error. The data were analyzed using Student’s t-test for independent samples with Microsoft Excel software. A logistic regression model for predicting the success of aGVHD prevention using MMSC was built in RStudio software (RStudio, Inc., USA). To assess the reliability of the model, the Pearson test for concordance was used. Patient survival was assessed using the Kaplan–Meier method.
RESULTS
Thirty-nine patients received MMSC for aGVHD prophylaxis. Cells were injected at the moment of blood cell reconstitution after allo-BMT (judged by leukocyte count of 109/liter). This time point was chosen due to the beginning of active graft growth, which often coincides with aGVHD manifestation. MMSC were injected after 19-54 days from allo-BMT (median, 28 days). Short-term fever and chills occurred in most patients during the first 24 h after MMSC injection. No other complications were revealed. Observation of the patients for 100 days (typical time of aGVHD manifestation) revealed two-fold decrease in aGVHD development in the group of patients who received MMSC when compared to the standard prophylaxis group. The results of prophylaxis are presented in Table 1.
Table 1. History of patients depending on
type of aGVHD prophylaxis
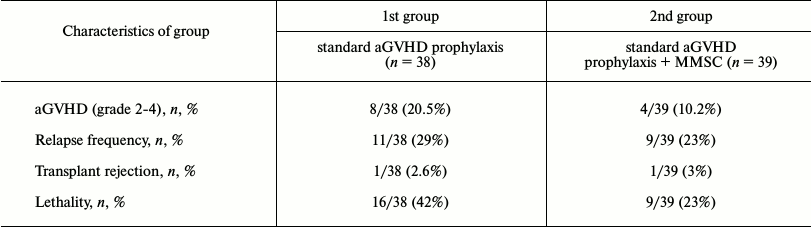
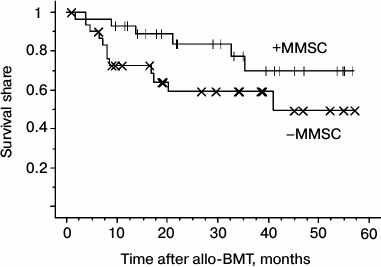
The data suggest that MMSC injection did not affect the engraftment as well as the relapse frequency. However, the relapse caused death in the group of standard prophylaxis more often than in the group receiving MMSC (8 from 11 patients (73%) versus 5 from 9 patients (55.5%)). Another cause of death apart from relapse was septic shock, which was also more often lethal in the standard prophylaxis group (data not shown). Therefore, total survival of the patients was higher in the MMSC receiving group (figure).
Total survival of patients (either subject or not to aGVHD prophylaxis with MMSC after allo-BMT)
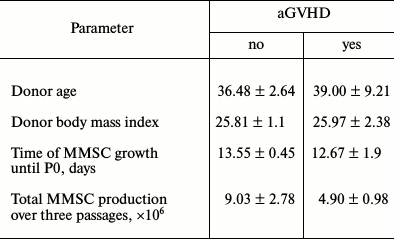
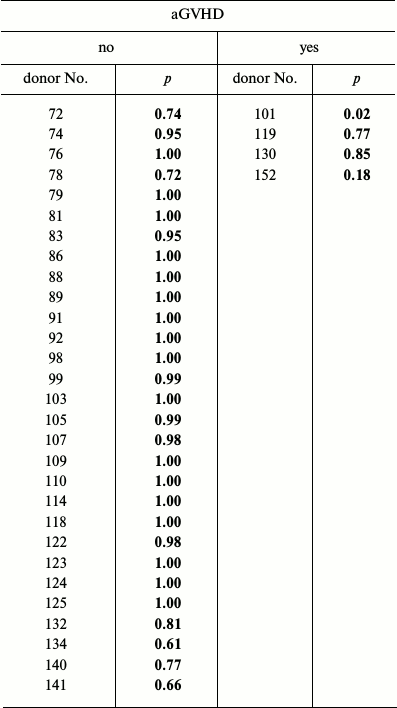
Among 39 patients receiving MMSC, four developed aGVHD grade 2-4. Some characteristics of MMSC donors are presented in Table 2. Analysis of these characteristics between donors whose MMSC did or did not prevented aGVHD development revealed no differences. Notably, cumulative MMSC production was decreased, though insignificantly, in cultures that proved ineffective for aGVHD prophylaxis.
Table 2. Comparison of some characteristics
of donors and MMSC obtained from bone marrow of those donors depending
on success of aGVHD prophylaxis
Donors’ MMSC that succeeded or failed to prevent aGVHD were compared for their ability to inhibit lymphocyte proliferation. No differences were revealed. The relative number of nonproliferating lymphocytes was doubled when compared to the control (lymphocytes cultivated without MMSC) in both groups. In the successful prophylaxis group the increase was 2.3 ± 0.23, and in the failed prophylaxis group it was 2.0 ± 0.32 times.
The relative expression levels of some genes regulating various properties of MMSC (differentiation level, proliferation, and immunomodulation ability) in the groups that succeeded or failed in aGVHD prophylaxis were analyzed. The results are presented in Table 3.
Table 3. Relative level of gene expression
in donor MMSC
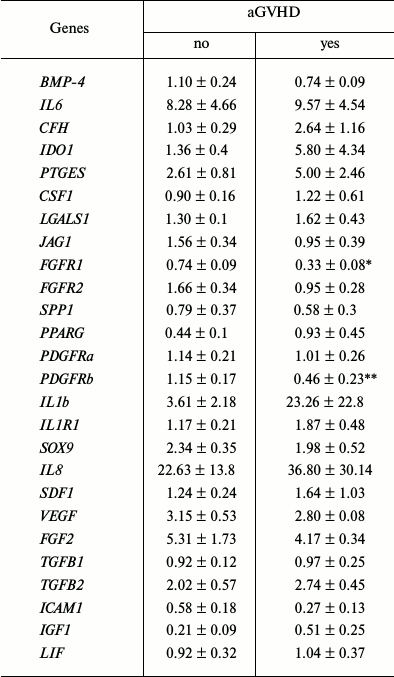
Note: Significant differences at * p = 0.005 and ** p =
0.048.
Expression level of the vast majority of analyzed genes did not differ in these two groups. The only exceptions were the FGFR1 and PDGFRb genes.
No universal indicator for prediction of MMSC eligibility for aGVHD prophylaxis was revealed. Therefore, all the data including donors’ characteristics, MMSC growth parameters, and relative gene expression level were analyzed by multiple logistic regression. This resulted in the informative combination of three parameters: relative expression levels of FGFR1, PPARG, and IGF1 genes. The logistic regression equation was as follows:
logit(P) = 0.75 + 10.897·FGFR1 – 4.272·PPARG – 2.014·IGF1, (1)
where logit(P) = ln[P/(1 – P)], P is probability of successful prophylaxis, and FGFR1, PPARG, and IGF1 are relative expression levels of the named genes in the tested sample. To assess the reliability of the model, the Pearson test for concordance was used, and p = 0.0053 was obtained. The calculated efficiency of this model was 94%. Addition of any other parameter to this model led to decrease in efficiency of the model. All MMSC samples previously used for aGVHD prophylaxis were analyzed by this model. Probability of successful prophylaxis was calculated as follows:
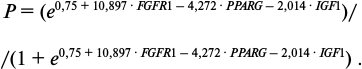 (2)
(2)Calculated probabilities (P) for each MMSC sample are listed in Table 4. The average calculated probability of success in the group of successful prophylaxis was 0.94, while in the group of failed prophylaxis it was 0.45. Thus, the combination of elevated relative expression level of FGFR1 with decreased levels of expression of PPARG and IGF1 could be considered favorable for using such MMSC sample for aGVHD prophylaxis.
Table 4. Calculated probability of
successful aGVHD prophylaxis with given MMSC samples
DISCUSSION
In this study, we attempted to evaluate the ability of MMSC from different healthy donors to prevent the development of aGVHD. This has both theoretical and practical value. MMSC have been used for the treatment of aGVHD for a few years now, and the efficiency of this therapy is ambiguous. There was no improvement in the patient’s condition in some cases [22, 23, 33]. No attempt to predict the quality of the MMSC sample resulted in an unambiguous result so far. Most protocols for aGVHD treatment with MMSC use cells derived from the bone marrow of a third donor (https://clinicaltrials.gov). However, because there is still no adequate criteria, preliminary evaluation of the quality of such samples is impossible.
The protocol investigating the efficiency of aGVHD prophylaxis with MMSC was initiated in the National Hematological Scientific Center, Ministry of Health, Moscow, Russia (https://clinicaltrials.gov, ID – NCT01941394). Currently it is a single randomized trial, and its preliminary results have demonstrated high efficiency of MMSC infusion for aGVHD prophylaxis [25]. According to the protocol, each patient was injected with MMSC samples derived from exactly the same donor’s bone marrow used for the allo-BMT to that patient. The MMSC infusion was performed at the moment of activation of the donor’s hematopoiesis, when peripheral blood leukocyte count reached 1000 cells/µl. Thus, at the moment of MMSC injection the hematopoietic system of each patient was in a similar state regardless of the diagnosis, conditioning regime, and immunosuppression therapy. Patients were randomly assigned to the two groups: in the first group, they received standard aGVHD prophylaxis; in the second group, they additionally received one injection of MMSC. Among 39 patients who received MMSC, four developed aGVHD grades 2-4 (10.2%); in the standard prophylaxis group, eight of 38 developed aGVHD of the same grades (21.1%). According to the published data, the frequency of aGVHD after allo-BMT is 30-40% [34, 35]. In our investigation, the frequency is lower than usual, probably due to the insufficiently large number of patients included in the protocol so far. Nevertheless, two-fold decrease in the aGVHD frequency after MMSC injection is an important and significant achievement in the treatment of patients after allo-BMT, which led to significant improvement in the patients’ survival.
Any transplantation apart from the one from an identical twin supposes differences in a large number of histocompatibility antigens. Donor selection is based on the six basic HLA genes. Bone marrow could be transplanted from related, unrelated, or even a haploidentical donor. Frequently bone marrow transplantation is the only way to cure the patient, thus any acceptable donor is used. The functionality of the graft as well as the probability of developing aGVHD depends on multiple factors; some of them are still unknown and could not be taken into consideration. Obviously, the cohort of patients is not homogeneous in potential for aGVHD development. It is impossible to take into account individual differences between the patients now, as mechanisms of development of aGVHD are still obscure.
To find the crucial differences in MMSC that failed to prevent aGVHD, the donors’ characteristics (gender, age, body mass index), the features of MMSC from those donors (growth parameters, levels of the expression of genes regulating the proliferation, differentiation, and immunomodulation) were analyzed. The analysis of most of these parameters did not reveal any significant differences. A tendency to decrease in cumulative MMSC cell production in the group with ineffective prophylaxis was noted. It was combined with a significant decrease in expression of receptors to growth factors FGF2 and PDGFb. The level of expression of the genes coding the immunomodulating factors did not differ in the two groups of MMSC. The main mechanism of MMSC inhibition of immune response is the secretion of IL6, CFH, IDO1, PTGES, CSF1, LGALS1, and other factors [18]. The MMSC samples did not differ at the level mRNA encoding these factors, but the protein production might be different and this was not analyzed in this study. As no straightforward comparison revealed the differences between effective and ineffective in aGVHD prophylaxis MMSC samples, all parameters were analyzed by the multiple logistic regression method. This statistic technique revealed the informative combination of FGFR1, PPARG, and IGF1 gene expression levels. Elevation of IGF1 expression level is known to occur in stromal cells of old animals [36]. Decrease in expression level of the receptor for the basic fibroblast growth factor (FGFR1) combined with increase in the expression levels of differentiation (PPARG) and aging (IGF1) markers indicates that the MMSC that failed to prevent aGVHD comprise a population with dominating mature cells. MMSC were shown to be a heterogeneous population consisting of cells with different levels of maturation [16, 37]. Obviously, a more mature MMSC population prevents aGVHD less effectively, and the action mechanism does not involve the immunomodulating factors studied in this work and the ability to inhibit lymphocyte proliferation. Based on the multiple logistic regression model, the outcome of the aGVHD prophylaxis was retrospectively confirmed in 94% of the cases. The outcome of the prophylaxis was considered positive if its calculated P was more than 0.5. With such conditions, all effective cases were indeed described as positive; however, in two out of four ineffective cases false positive result was observed. The occurrence of false positive results in practice could lead to the inclusion of improper MMSC samples. To avoid this, the margin allowing level of P was elevated to 0.95. With this condition, all MMSC samples that failed to prevent aGVHD were correctly defined as negative by the model. However, this allowance led to the occurrence of false negative results in the effective MMSC group. Six of 29 MMSC samples that successfully prevented aGVHD were defined as negative by the model (the calculated P was lower than 0.95). According to international statistics, aGVHD does not occur in 60% of patients independently of the prophylaxis method. That is why one cannot be sure that those MMSC samples defined as negative by the model indeed prevented aGVHD. Use of more stringent criteria seems to be appropriate. Such rigorous approach to the validation of potentially successful in aGVHD prophylaxis MMSC samples allows creating an MMSC bank with the most appropriate for prophylaxis samples. Those chosen samples could also be used for patients whose donor’s MMSC did not fit the applied criteria for efficiency of aGVHD prophylaxis.
Therefore, for the first time a model for prospective evaluation of MMSC sample efficiency in aGVHD prophylaxis after allo-BMT is proposed. This model stands at the beginning of a long study of the mechanisms of immune response inhibition by MMSC. Obviously, there are other factors affecting the efficiency of this inhibition that remain unknown. Identification of these factors and their inclusion into the new model will increase its specificity and help to prevent aGVHD in the maximum number of patients.
The authors thank the team of the Department of High-Dose Chemotherapy, Depressions of Hemopoiesis, and Bone Marrow Transplantation of the National Hematological Scientific Center.
This study was supported by Russian Foundation for Basic Research grant 12-04-00457a.
REFERENCES
1.Appelbaum, F. R. (2001) Haematopoietic cell
transplantation as immunotherapy, Nature, 411,
385-389.
2.Tabbara, I. A., Zimmerman, K., Morgan, C., and
Nahleh, Z. (2002) Allogeneic hematopoietic stem cell transplantation:
complications and results, Arch. Intern. Med., 162,
1558-1566.
3.Billingham, R. E. (1966-1967) The biology of
graft-versus-host reactions, Harvey Lect., 62, 21-78.
4.Welniak, L. A., Blazar, B. R., and Murphy, W. J.
(2007) Immunobiology of allogeneic hematopoietic stem cell
transplantation, Annu. Rev. Immunol., 25, 139-170.
5.Appelbaum, F. R. (2003) The current status of
hematopoietic cell transplantation, Annu. Rev. Med., 54,
491-512.
6.Ferrara, J. L. M., Levine, J. E., Reddy, P., and
Holler, E. (2009) Graft-versus-host disease, Lancet, 373,
1550-1561.
7.Filipovich, A. H., Weisdorf, D., Pavletic, S.,
Socie, G., Wingard, J. R., Lee, S. J., Martin, P., Chien, J.,
Przepiorka, D., Couriel, D., Cowen, E. W., Dinndorf, P., Farrell, A.,
Hartzman, R., Henslee-Downey, J., Jacobsohn, D., McDonald, G.,
Mittleman, B., Rizzo, J. D., Robinson, M., Schubert, M., Schultz, K.,
Shulman, H., Turner, M., Vogelsang, G., and Flowers, M. E. (2005)
National Institutes of Health consensus development project on criteria
for clinical trials in chronic graft-versus-host disease: I. Diagnosis
and staging working group report, Biol. Blood Marrow
Transplant., 11, 945-956.
8.Ferrara, J. L., Levy, R., and Chao, N. J. (1999)
Pathophysiological mechanisms of acute graft-vs.-host disease, Biol.
Blood Marrow Transplant., 5, 347-356.
9.Visentainer, J. E. L., Lieber, S. R., Persoli, L.
B. L., Vigorito, A. C., Aranha, F. J. P., de Brito Eid, K. A.,
Oliveira, G. B., Miranda, E. C., and de Souza, C. A. (2003) Serum
cytokine levels and acute graft-versus-host disease after HLA-identical
hematopoietic stem cell transplantation, Exp. Hematol.,
31, 1044-1050.
10.Xun, C. Q., Thompson, J. S., Jennings, C. D.,
Brown, S. A., and Widmer, M. B. (1994) Effect of total body
irradiation, busulfan-cyclophosphamide, or cyclophosphamide
conditioning on inflammatory cytokine release and development of acute
and chronic graft-versus-host disease in H-2-incompatible transplanted
SCID mice, Blood, 83, 2360-2367.
11.Leeuwenberg, J. F., Van Damme, J., Meager, T.,
Jeunhomme, T. M., and Buurman, W. A. (1988) Effects of tumor necrosis
factor on the interferon-gamma-induced major histocompatibility complex
class II antigen expression by human endothelial cells, Eur. J.
Immunol., 18, 1469-1472.
12.Norton, J., and Sloane, J. P. (1991) ICAM-1
expression on epidermal keratinocytes in cutaneous graft-versus-host
disease, Transplantation, 51, 1203-1206.
13.Chao, N. J. (1997) Graft-versus-host disease: the
viewpoint from the donor T cell, Biol. Blood Marrow Transplant.,
3, 1-10.
14.Taylor, P. A., Noelle, R. J., and Blazar, B. R.
(2001) CD4(+)CD25(+) immune regulatory cells are required for induction
of tolerance to alloantigen via costimulatory blockade, J. Exp.
Med., 193, 1311-1318.
15.Deans, R. J., and Moseley, A. B. (2000)
Mesenchymal stem cells: biology and potential clinical uses, Exp.
Hematol., 28, 875-884.
16.Colter, D. C., Sekiya, I., and Prockop, D. J.
(2001) Identification of a subpopulation of rapidly self-renewing and
multipotential adult stem cells in colonies of human marrow stromal
cells, Proc. Natl. Acad. Sci. USA, 98, 7841-7845.
17.Majumdar, M. K., Thiede, M. A., Haynesworth, S.
E., Bruder, S. P., and Gerson, S. L. (2000) Human marrow-derived
mesenchymal stem cells (MSCs) express hematopoietic cytokines and
support long-term hematopoiesis when differentiated toward stromal and
osteogenic lineages, J. Hematother. Stem Cell Res., 9,
841-848.
18.Jones, B. J., and McTaggart, S. J. (2008)
Immunosuppression by mesenchymal stromal cells: from culture to clinic,
Exp. Hematol., 36, 733-741.
19.Aggarwal, S., and Pittenger, M. F. (2005) Human
mesenchymal stem cells modulate allogeneic immune cell responses,
Blood, 105, 1815-1822.
20.Le Blanc, K., Rasmusson, I., Sundberg, B.,
Gotherstrom, C., Hassan, M., Uzunel, M., and Ringden, O. (2004)
Treatment of severe acute graft-versus-host disease with third party
haploidentical mesenchymal stem cells, Lancet, 363,
1439-1441.
21.Samuelsson, H., Ringden, O., Lonnies, H., and Le
Blanc, K. (2009) Optimizing in vitro conditions for
immunomodulation and expansion of mesenchymal stromal cells,
Cytotherapy, 11, 129-136.
22.Prockop, D. J., Brenner, M., Fibbe, W. E.,
Horwitz, E., Le Blanc, K., Phinney, D. G., Simmons, P. J., Sensebe, L.,
and Keating, A. (2010) Defining the risks of mesenchymal stromal cell
therapy, Cytotherapy, 12, 576-578.
23.Ringden, O., Uzunel, M., Rasmusson, I.,
Remberger, M., Sundberg, B., Lonnies, H., Marschall, H. U., Dlugosz,
A., Szakos, A., Hassan, Z., Omazic, B., Aschan, J., Barkholt, L., and
Le Blanc, K. (2006) Mesenchymal stem cells for treatment of
therapy-resistant graft-versus-host disease, Transplantation,
81, 1390-1397.
24.Tisato, V., Naresh, K., Girdlestone, J.,
Navarrete, C., and Dazzi, F. (2007) Mesenchymal stem cells of cord
blood origin are effective at preventing but not treating
graft-versus-host disease, Leukemia, 21, 1992-1999.
25.Kuzmina, L. A., Petinati, N. A., Parovichnikova,
E. N., Lubimova, L. S., Gribanova, E. O., Gaponova, T. V.,
Shipounova, I. N., Zhironkina, O. A., Bigildeev, A. E., Svinareva, D.
A., Drize, N. J., and Savchenko, V. G. (2012) Multipotent mesenchymal
stromal cells for the prophylaxis of acute graft-versus-host disease
– a phase II study, Stem Cells Int., 2012; DOI:
10.1155/2012/968213.
26.Glucksberg, H., Storb, R., Fefer, A., Buckner, C.
D., Neiman, P. E., Clift, R. A., Lerner, K. G., and Thomas, E. D.
(1974) Clinical manifestations of graft-versus-host disease in human
recipients of marrow from HL-A-matched sibling donors,
Transplantation, 18, 295-304.
27.Lange, C., Cakiroglu, F., Spiess, A.,
Cappallo-Obermann, H., Dierlamm, J., and Zander, A. R. (2007)
Accelerated and safe expansion of human mesenchymal stromal cells in
animal serum-free medium for transplantation and regenerative medicine,
J. Cell Physiol., 213, 18-26.
28.Dominici, M., Le Blanc, K., Mueller, I.,
Slaper-Cortenbach, I., Marini, F., Krause, D., Deans, R., Keating, A.,
Prockop, D. J., and Horwitz, E. (2006) Minimal criteria for defining
multipotent mesenchymal stromal cells. The International Society for
Cellular Therapy position statement, Cytotherapy, 8,
315-317.
29.Fong, T. A., and Mosmann, T. R. (1990)
Alloreactive murine CD8+ T cell clones secrete the Th1 pattern of
cytokines, J. Immunol., 144, 1744-1752.
30.Svinareva, D. A., Shipunova, I. N.,
Ol’shanskaia, I. V., Momotiuk, K. S., Drize, N. I., and
Savchenko, V. G. (2010) The basic properties of mesenchymal stromal
cells from the donor bone marrow: superficial markers, Ter.
Arkhiv, 82, 52-56.
31.Chomczynski, P., and Sacchi, N. (1987)
Single-step method of RNA isolation by acid guanidinium
thiocyanate–phenol–chloroform extraction, Anal.
Biochem., 162, 156-159.
32.Schmittgen, T. D., and Livak, K. J. (2008)
Analyzing real-time PCR data by the comparative CT method, Nat.
Protoc., 3, 1101-1108.
33.Ringden, O., and Le Blanc, K. (2011) Mesenchymal
stem cells for treatment of acute and chronic graft-versus-host
disease, tissue toxicity and hemorrhages, Best Pract. Res. Clin.
Haematol., 24, 65-72.
34.Appelbaum, F. R. (2003) The current status of
hematopoietic cell transplantation, Annu. Rev. Med., 54,
491-512.
35.Ferrara, J. L. M., Levine, J. E., Reddy, P., and
Holler, E. (2009) Graft-versus-host disease, Lancet, 373,
1550-1561.
36.Mayack, S. R., Shadrach, J. L., Kim, F. S., and
Wagers, A. J. (2010) Systemic signals regulate ageing and rejuvenation
of blood stem cell niches, Nature, 463, 495-500.
37.Bigildeev, A. E., Zhironkina, O. A., Shipounova,
I. N., Sats, N. V., Kotyashova, S. Y., and Drize, N. I. (2012)
Clonal composition of human multipotent mesenchymal stromal cells,
Exp. Hematol., 40, 847-856.