The A2B Adenosine Receptor Colocalizes with Adenosine Deaminase in Resting Parietal Cells from Gastric Mucosa
R. M. Arin*, A. I. Vallejo, Y. Rueda, O. Fresnedo, and B. Ochoa
Department of Physiology, Faculty of Medicine and Dentistry, University of the Basque Country UPV/EHU, Sarriena s/n, 48940 Leioa, Spain; fax: +34 (946) 015-662; E-mail: rosamaria.arin@ehu.es; begona.ochoa@ehu.es; olatz.fresnedo@ehu.es; yuri.rueda@ehu.es; anaivallejo@gmail.com* To whom correspondence should be addressed.
Received July 24, 2014; Revision received September 19, 2014
The A2B adenosine receptor (A2BR) mediates biological responses to extracellular adenosine in a wide variety of cell types. Adenosine deaminase (ADA) can degrade adenosine and bind extracellularly to adenosine receptors. Adenosine modulates chloride secretion in gastric glands and gastric mucosa parietal cells. A close functional link between surface A2BR and ADA has been found on cells of the immune system, but whether this occurs in the gastrointestinal tract is unknown. The goal of this study was to determine whether A2BR and ADA are coexpressed at the plasma membrane of the acid-secreting gastric mucosa parietal cells. We used isolated gastric parietal cells after purification by centrifugal elutriation. The membrane fraction was obtained by sucrose gradient centrifugation. A2BR mRNA expression was analyzed by RT-PCR. The surface expression of A2BR and ADA proteins was evaluated by Western blotting, flow cytometry and confocal microscopy. Our findings demonstrate that A2BR and ADA are expressed in cell membranes isolated from gastric parietal cells. They show a high degree of colocalization that is particularly evident in the surface of contact between parietal cells. The confocal microscopy data together with flow cytometry analysis suggest a tight association between A2BR and ADA that might be specifically linked to glandular secretory function.
KEY WORDS: adenosine receptor, adenosine deaminase, purine nucleoside signaling, cell membrane, gastric mucosa parietal cellDOI: 10.1134/S0006297915010149
Abbreviations: A2BR, A2B adenosine receptor; ADA, adenosine deaminase; RT-PCR, reverse transcription-polymerase chain reaction.
Adenosine is a regulatory metabolite, a purine nucleoside that is
continuously produced in intracellular and extracellular locations, and
it modulates a broad spectrum of physiological processes. Some injured
tissues produce high levels of adenosine, which is further released by
cells in response to metabolic stress or during tissue hypoxia or
ischemia [1].
The effects of extracellular adenosine are mediated by G protein-coupled proteins known as adenosine receptors [2]. There are four subtypes of adenosine receptors: A1, A2A, A2B, and A3. The A2B adenosine receptor (A2BR) is the least studied and still remains the most enigmatic adenosine receptor subtype because of the relatively low potency of adenosine at this receptor (EC50 value of 24 µM) [2] and the very few specific agonists that have been described so far. However, there is a growing interest in A2BR in recent years as it has been shown to play a role in inflammation and cancer and is even considered a promising new pharmacotherapeutic target [3]. Of particular note is that during inflammatory ischemia extracellular adenosine is elevated to levels sufficient for A2BR activation, and A2BR is transcriptionally regulated by factors implicated in inflammatory hypoxia [4], suggesting that A2BR may contribute to control inflammation during tissue hypoxia [5].
Adenosine deaminase (ADA) is an enzyme present in intracellular and extracellular compartments that catalyzes the irreversible deamination of adenosine in inosine. Extracellular ADA is capable of interacting with the cell surface, whereby ADA may be found as an ectoenzyme [6, 7]. Surface ADA or ectoADA is a multifunctional protein that, apart from degrading extracellular adenosine, can act as a co-stimulating molecule in certain activation processes and as a communication bridge between cells [8]. Four anchor proteins are known for ADA: A1, A2A, and A2B adenosine receptors and the broadly expressed protein CD26/dipeptidyl peptidase IV [9, 10]. On the other hand, ectoADA participates in signal transduction modulation by interacting with A1 and A2B adenosine receptors [9]. Some authors have described a close functional relationship between A2BR and ADA, especially when they are coexpressed on the cell surface [11, 12].
Previous works have demonstrated that adenosine and adenosine analogs stimulate the secretion of acid in isolated glands and parietal cells from rabbit gastric mucosa [13-15]. Gastric acid secretion by parietal cells depends on the apical extrusion of Cl– ions, and it has been demonstrated that the function of chloride carriers is regulated by A2B receptor in other cell types. The binding of adenosine to A2BR in human intestinal epithelium results in activation of electrogenic Cl– secretion during inflammation [16]. In colonic epithelium cells, A2BR are expressed in the luminal and basolateral membranes, where they stimulate chloride secretion [17]. Adenosine can act through apical A2BR to stimulate chloride secretion directly through the cystic fibrosis conductance regulator protein in cell line mIMCD-K2, a murine model of the renal inner medullary collecting duct [18].
In this context, we aimed to elucidate if the acid-secreting parietal cells express A2BR in their membrane and, if so, whether it colocalizes with ADA.
MATERIALS AND METHODS
Isolation of parietal cells from rabbit gastric mucosa. Male and female New Zealand rabbits weighing 2.5-4 kg were used. Housing and experimental procedures were in accordance with the Spanish (RD 1201/2005) and European (2003/65/CE Directive and 2007/526/CE Recommendation) guidelines for the use of laboratory animals. The rabbits were euthanized by the administration of an anesthetic preparation of pentobarbital. The gastric mucosa was isolated as described by Berglindh and Obrink [19]. Briefly, the stomach was perfused with PBS, and the mucosa was separated by blunt dissection. The gastric mucosa cells were isolated following the method of Fryklund et al. [20] as modified by Ainz et al. [13]. Parietal cells were purified by centrifugal elutriation as reported previously [21]. Cell viability was measured by the trypan blue exclusion test and averaged 90-95%. Cell functionality was monitored in every preparation by measuring hydrochloric acid secretion in response to 10–3 to 10–7 M histamine stimulation as a surrogate index (data not shown).
Isolation of adenosine-free plasma membranes. We followed the method by Muallem et al. [22] with slight modifications. Parietal cells (60·106 cells/ml in 20 mM Tris-HCl, pH 7.4, 250 mM sucrose, 0.5 mM EDTA, 0.54 mM dithiothreitol, 5 μg/ml leupeptin, 15.7 μg/ml benzamidine) were homogenized in a Potter-Elvehjem homogenizer. The 700g supernatant was layered over a 47% sucrose solution and ultracentrifuged in a swinging-bucket rotor (100,000g, 45 min, 4°C). Plasma membranes were collected from the interphase, washed once (120,000g, 20 min, 4°C) in 50 mM Tris-HCl, pH 7.4, and incubated with ADA (2 U/ml, 1 h, room temperature) to degrade endogenous adenosine. The washed membranes were stored in the same buffer at –80°C until used.
Immunostaining experiments: flow cytometry and confocal microscopy. The staining procedure was conducted exactly as described by Mirabet et al. [11]. Nonpermeabilized isolated parietal cells (4·106 in PBS) were fixed in 2% (w/v) paraformaldehyde, 60 mM sucrose in PBS, pH 7.4, for 15 min at room temperature. After washing with 20 mM glycine in PBS, the cells were blocked with 1% BSA and immunostained using rabbit anti-human A2BR [11] or anti-human ADA [23] and a FITC-conjugated secondary anti-rabbit IgG antibody (Boehringer Mannheim, Germany). Primary antibodies were developed, characterized, and kindly provided by Prof. Rafael Franco from the University of Barcelona (Barcelona, Spain).
Flow cytometry analysis was done using an EPICS Profile flow cytometer (Coulter Corporation, USA). The parameters used to select cell subpopulations for analysis were forward and side light scattering. Cell sorting was performed using EPICS Profile equipment.
For confocal microscopy analysis, cells were immunostained as for flow cytometry except that a TRITC-conjugated secondary antibody (Boehringer Mannheim) was used to visualize ADA. Cells were mounted with Immuno-FluoreTM mounting medium and were scanned using a Leica TCS 4D confocal scanning laser microscope adapted to an inverted Leitz DMIRBE microscope (Leica Lasertechnik GmbH, Germany). Colocalization analysis was performed using Multi Color software (version 2.0; Leica Lasertechnik GmbH).
mRNA analysis by reverse transcription polymerase chain reaction (RT-PCR). Total RNA was extracted from 10·106 parietal cells using Trizol Reagent (Invitrogen, Spain) and cDNAs were obtained by retrotranscription (SuperScript II RT; Invitrogen) following the manufacturers’ instructions. ADORA2B mRNA was detected by end point analysis of PCR products using two pairs of primers: 5′-CGTTCCATGCCATCAACTG/TTGACATCCGTCTGGCAGAGA-3′ and 5′-TCGTCAACCCCATCGTGTATG/TCCTAGGTGGGCATGTGGAA-3′ (AY630339) at 0.6 µM. The PCR was run in 40 amplification cycles (30 s at 94°C, 1 min at 60°C, and 1 min at 72°C, followed by a 10 min incubation at 72°C) using activated (1 min at 95°C) recombinant HotStar Taq DNA polymerase (Qiagen, Germany). The amplicons (202 and 251 bp) were visualized after electrophoretic migration in a 1.5% agarose gel by ethidium bromide staining.
Western blotting and protein quantification. Protein concentration was determined by the Bradford method [24] using BSA as a standard. Protein denaturation and fractionation by 7.5% SDS-PAGE and immunodetection of A2BR and ADA were performed on isolated plasma membranes from gastric parietal cells as described by Herrera et al. [12] using 20-25 µg protein per lane. The same antibodies as for confocal microscopy and flow cytometry and peroxidase-conjugated goat anti-rabbit IgG (Boehringer Mannheim) were used as primary and secondary antibodies, respectively. DDT1 MF-2 (DDT1) hamster smooth muscle cells (ATCC, USA) and commercially available pure ADA (ICN Biomedical Inc., USA) were used as positive control for A2BR and ADA detection, respectively.
RESULTS AND DISCUSSION
Adenosine has been shown to play a significant role in gastric function. It suppresses gastric acid secretion in guinea pigs [25], dogs [26], and rats [27]. In mouse stomach, adenosine has dual actions on regulating gastric somatostatin release: stimulatory at high concentrations through A2A receptor and inhibitory at low concentrations through A1 receptor. Therefore, adenosine seems to modulate gastric function by several mechanisms [28].
In addition, previous pharmacological studies have shown that adenosine and some of its analogs stimulate gastric secretion of acid in glands and parietal cells from rabbit gastric mucosa [13-15]. As a first approach to the participation of A2BR in the regulation of acid secretion by gastric parietal cells, here we analyze whether A2BR is expressed in the parietal cell plasma membrane in rabbit gastric mucosa.
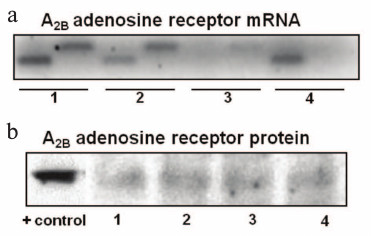
To know if parietal cells expressed the A2BR-encoding ADORA2B gene, RT-PCR was performed in four different parietal cell preparations with two different primer sets. Figure 1a shows that all the preparations were positive for at least one of the amplicons, although preparation 3 showed lower amplification degree. Differences might be related to the functional states of mucosal glands.
Fig. 1. Expression of A2BR mRNA (a) and protein (b) in gastric mucosa parietal cells. Four different preparations of purified parietal cells were used without any further stimulation. a) Total RNA was extracted from cells and RT-PCR analysis of A2BR mRNA was performed as described in “Materials and Methods” using two distinct sets of primers. The PCR products were separated by electrophoresis in a 1.5% agarose gel. Consecutive lanes contain the same RNA preparation probed with different pairs of primers. Enrichment with parietal cells was higher in preparations 1 and 2. b) Plasma membranes were isolated from parietal cells and subjected to immunoblotting of A2BR as described in “Materials and Methods” using 20-25 µg of protein per lane. DDT1 hamster smooth muscle cells were used as a positive (+) control.
With the aim of analyzing A2BR protein localization in plasma membrane and of testing its potential functionality, we isolated plasma membranes from gastric parietal cells. Western blotting analysis revealed an immunoreactive band of about 45 kDa in all the four parietal cell preparations (Fig. 1b). DDT1 smooth muscle cells were used as positive control. Some controversy exists about the molecular weight of A2BR, and other authors have reported it to be 34.8, 36, or 50 kDa [29-31]. On the other hand, the faintness of the signal obtained in the Western blot may be because the antibody used is not rabbit but human A2BR specific and the recognition might be only partial, or that during the resting state not all A2BR of parietal cells are localized in the membrane [17].
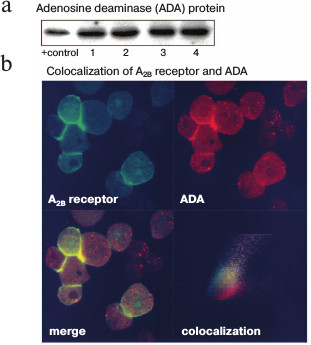
As mentioned before, A2BR is one of the anchor proteins of ADA [9, 10]. This enzyme deactivates adenosine by deaminating it into inosine. Some authors have described a tight functional relationship between the two proteins; ectoADA can interact with A2BR when both are expressed on cell surface [9, 11, 12]. Western blotting showed that ADA was present in the isolated plasma membrane of rabbit parietal cells (Fig. 2a). The molecular weight observed (40 kDa) agrees with that reported for other species [32, 33]. No secondary immunoreactive bands were observed.
Fig. 2. Immunodetection of cell surface ADA and A2BR in gastric mucosa parietal cells. a) Plasma membranes, isolated from four different preparations of parietal cells, were subjected to immunoblotting of ADA as described in “Materials and Methods” using 20-25 µg of protein per lane. Commercially available pure ADA was used as a positive (+) control. b) Parietal cells isolated from rabbit gastric mucosa were used without any further stimulation. Cells were processed for confocal microscopy analysis as described in “Materials and Methods”. They were fixed and labeled with 50 µg/ml FITC-anti-A2BR (upper left image, green) and 50 µg/ml TRITC-anti-ADA (upper right image, red) antibodies. The merge of the two images shows colocalization of cell surface A2BR and ADA (lower left image, yellow). The lower right image shows the confocal cytofluorogram in which yellow represents global colocalization of the two proteins.
The presence of A2BR and ADA in the plasma membrane of parietal cells was also analyzed by confocal microscopy of nonpermeabilized samples (Fig. 2b). Two different cell types can be distinguished in the mucosal cell preparations: parietal cells, which present surface expression of both A2BR (in green) and ADA (in red), and chief cells, which do not show significant surface ADA or A2BR signal. The intracellular fluorescent signal was probably due to spontaneous permeabilization. Notably, A2BR and ADA expression levels were higher in cell–cell contact areas, either parietal–parietal or parietal–chief cells. The merge of both signals (in yellow) shows that there is a high degree of A2BR and ADA colocalization in parietal cells, while chief cells only express ADA. The cytofluorogram in the fourth panel in Fig. 2b shows that the colocalizing areas correspond to the pixels with the highest fluorescence intensity, indicating highly specific neighboring of A2BR and ADA.
It has been previously suggested that ADA might modulate A2BR-ligand binding in other cell types [12]. As in gastric parietal cells, ADA expression level and ADA–A2BR colocalization degree are higher in cell contact areas in many cell types [10, 12]. A role has been suggested for ADA in the modulation of cell adhesion in immune synapse, possibly working as a bridge between A2BR and other plasma membrane proteins [10]. According to Pacheco et al. [9], ADA bound to A2BR on the surface of dendritic cells can interact with CD26 on T cell surface, enhancing T cell activation and proliferation.
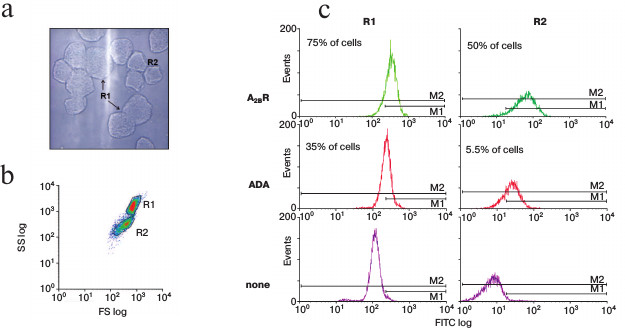
Flow cytometry analysis (Fig. 3b) reflects the differences in morphology and size observed by transmission microscopy (Fig. 3a) between different cell types present in the mucosal cell suspensions. Parietal cells are bigger [34, 35] and have a more granulated appearance [36] than other stomach epithelial cells, probably due to the great number of microvilli and tubulovesicles they have, which is one of their main characteristics [37-39]. Chief cells are smaller and non-granulated.
Fig. 3. Analysis of the cell subpopulations in isolated parietal cell preparations. a) Image of the transmitted light in the visual field shown in Fig. 2b. b) Spot diagram of the flow cytometry analysis of a representative parietal cell preparation. The X-axis represents frontal dispersion of light (FS), which indicates cell size, and the Y-axis represents lateral dispersion (SS), which indicates cell complexity. R1 and R2 cell subpopulations were identified. Only fluorescence of cells in areas R1 and R2 was measured. c) Monoparametric representation of fluorescence in R1 and R2 cell subpopulations. The minimum number of cells used was 20,500. The X-axis represents FITC fluorescence intensity (FITC log, arbitrary units) and the Y-axis represents the number of cells that show fluorescence (events). About 75% of R1 cells and 50% of R2 cells expressed A2BR, and 35% of R1 cells and 5.5% of R2 cells expressed ADA. Top down: anti-A2BR-FITC fluorescence, anti-ADA-FITC fluorescence, and autofluorescence. Histograms were analyzed with WinMDI 2.8.
In our preparations, two cell subpopulations are observed. According to cell size and cytoplasmic complexity distribution, the R1 subpopulation comprises parietal cells whereas the R2 subpopulation comprises small parietal cells and contaminant chief cells. The distribution of ADA and A2BR in immunostained R1 and R2 cell subpopulation is shown in Fig. 3c. In the lower panel, cell autofluorescence distribution is shown. The high level of autofluorescence of R1 cells is a typical feature of parietal cells owing to the great number of mitochondria these cells contain [36, 38]. However, chief cells do not have elevated autofluorescence levels [40] and therefore these data confirm that cell separation was done according to cell type.
Cytometry was also performed using only the secondary antibody (none) to assess the fluorescence due to unspecific binding; it accounts for 4.5% of the fluorescence emitted by immunostained R1 cells and for 39.4% of that of R2 cells (data not shown). Those data were used to calculate specific primary antibody binding to cell surface. About 75% of R1 cells and 50% of R2 cells expressed A2BR, while 35% of R1 cells and only 5.5% of R2 cells expressed ADA.
In conclusion, resting gastric mucosa parietal cells did express A2BR and ADA. The presence of potentially functional, plasma membrane-associated A2BR and ADA in isolated parietal cells is maximal in cell contact areas where they showed the highest degree of colocalization. It would be interesting to continue the investigation on the functional relevance of this neighboring, especially to assess the relevance of A2BR in the physiology of gastric acid secretion.
We are indebted to all the members of Dr. Rafael Franco and Dr. Carmen Lluis research group in the University of Barcelona Department of Biochemistry and Molecular Biology for experimental help and advice received. We thank the technical help from Dr. Yolanda Chico, Dr. Patricia Aspichueta, and Dr. Rosaura Navarro from the Department of Physiology of the University of the Basque Country.
This study was funded by grants from the Department of Education, Universities and Research of the Basque Government (IT336/10) and the University of the Basque Country (NUPV 06/21 and UFI11/20).
REFERENCES
1.Van, L. A., and Eltzschig, H. K. (2007) Role of
pulmonary adenosine during hypoxia: extracellular generation, signaling
and metabolism by surface adenosine deaminase/CD26, Expert Opin.
Biol. Ther., 7, 1437-1447.
2.Fredholm, B. B., Izerman, A. P., Jacobson, K. A.,
Klotz, K. N., and Linden, J. (2001) International Union of
Pharmacology. XXV. Nomenclature and classification of adenosine
receptors, Pharmacol. Rev., 53, 527-552.
3.Thimm, D., Schiedel, A. C., Sherbiny, F. F., Hinz,
S., Hochheiser, K., Bertarelli, D. C., Maass, A., and Muller, C. E.
(2013) Ligand-specific binding and activation of the human adenosine
A(2B) receptor, Biochemistry, 52, 726-740.
4.Kong, T., Westerman, K. A., Faigle, M., Eltzschig,
H. K., and Colgan, S. P. (2006) HIF-dependent induction of adenosine
A2B receptor in hypoxia, FASEB J., 20,
2242-2250.
5.Aherne, C. M., Kewley, E. M., and Eltzschig, H. K.
(2011) The resurgence of A2B adenosine receptor signaling, Biochim.
Biophys. Acta, 1808, 1329-1339.
6.Franco, R., Valenzuela, A., Lluis, C., and Blanco,
J. (1998) Enzymatic and extraenzymatic role of ecto-adenosine deaminase
in lymphocytes, Immunol. Rev., 161, 27-42.
7.Franco, R., Casado, V., Ciruela, F., Saura, C.,
Mallol, J., Canela, E. I., and Lluis, C. (1997) Cell surface adenosine
deaminase: much more than an ectoenzyme, Prog. Neurobiol.,
52, 283-294.
8.Gines, S., Marino, M., Mallol, J., Canela, E. I.,
Morimoto, C., Callebaut, C., Hovanessian, A., Casado, V., Lluis, C.,
and Franco, R. (2002) Regulation of epithelial and lymphocyte cell
adhesion by adenosine deaminase–CD26 interaction, Biochem.
J., 361, 203-209.
9.Pacheco, R., Martinez-Navio, J. M., Lejeune, M.,
Climent, N., Oliva, H., Gatell, J. M., Gallart, T., Mallol, J., Lluis,
C., and Franco, R. (2005) CD26, adenosine deaminase, and adenosine
receptors mediate costimulatory signals in the immunological synapse,
Proc. Natl. Acad. Sci. USA, 102, 9583-9588.
10.Gracia, E., Farre, D., Cortes, A., Ferrer-Costa,
C., Orozco, M., Mallol, J., Lluis, C., Canela, E. I., McCormick, P. J.,
Franco, R., Fanelli, F., and Casado, V. (2013) The catalytic site
structural gate of adenosine deaminase allosterically modulates ligand
binding to adenosine receptors, FASEB J., 27,
1048-1061.
11.Mirabet, M., Herrera, C., Cordero, O. J., Mallol,
J., Lluis, C., and Franco, R. (1999) Expression of A2B adenosine
receptors in human lymphocytes: their role in T cell activation, J.
Cell. Sci., 112, 491-502.
12.Herrera, C., Morimoto, C., Blanco, J., Mallol,
J., Arenzana, F., Lluis, C., and Franco, R. (2001) Comodulation of
CXCR4 and CD26 in human lymphocytes, J. Biol. Chem.,
276, 19532-19539.
13.Ainz, L. F., Salgado, C., Gandarias, J. M.,
Gomez, R., Vallejo, A., and Gil-Rodrigo, C. E. (1993)
P1(A2/Ra)-purinoceptors may mediate the stimulatory effect of adenosine
and adenosine analogs on acid formation in isolated rabbit parietal
cells, Pharmacol. Res., 27, 319-334.
14.Ainz, L. F., Gil-Rodrigo, C. E., Gomez, R.,
Malillos, M., Requejo, D., and Gandarias, J. M. (1989) Effects of
various physiologic adenine derivatives on the secretion of acid in
isolated gastric glands in rabbits, Rev. Esp. Fisiol.,
45, 281-286.
15.Gil-Rodrigo, C. E., Galdiz, B., Gandarias, J. M.,
Gomez, R., and Ainz, L. F. (1990) Characterization of the effects of
adenosine, adenosine 5′-triphosphate and related purines on acid
secretion in isolated rabbit gastric glands, Pharmacol. Res.,
22, 103-113.
16.Colgan, S. P., Fennimore, B., and Ehrentraut, S.
F. (2013) Adenosine and gastrointestinal inflammation, J. Mol. Med.
(Berl.), 91, 157-164.
17.Wang, L., Kolachala, V., Walia, B.,
Balasubramanian, S., Hall, R. A., Merlin, D., and Sitaraman, S. V.
(2004) Agonist-induced polarized trafficking and surface expression of
the adenosine 2b receptor in intestinal epithelial cells: role of SNARE
proteins, Am. J. Physiol. Gastrointest. Liver Physiol.,
287, G1100-G1107.
18.Rajagopal, M., and Pao, A. C. (2010) Adenosine
activates a2b receptors and enhances chloride secretion in kidney inner
medullary collecting duct cells, Hypertension, 55,
1123-1128.
19.Berglindh, T., and Obrink, K. J. (1976) A method
for preparing isolated glands from the rabbit gastric mucosa, Acta
Physiol. Scand., 96, 150-159.
20.Fryklund, J., Wallmark, B., Larsson, H., and
Helander, H. F. (1984) Effect of omeprazole on gastric secretion in
H+,K+-ATPase and in pepsinogen-rich cell
fractions from rabbit gastric mucosa, Biochem. Pharmacol.,
33, 273-280.
21.Arin, R. M., Rueda, Y., Casis, O., Gallego, M.,
Vallejo, A. I., and Ochoa, B. (2014) Basolateral expression of GRP94 in
parietal cells of gastric mucosa, Biochemistry (Moscow),
79, 8-15.
22.Muallem, S., Burnham, C., Blissard, D.,
Berglindh, T., and Sachs, G. (1985) Electrolyte transport across the
basolateral membrane of the parietal cells, J. Biol. Chem.,
260, 6641-6653.
23.Aran, J. M., Colomer, D., Matutes, E.,
Vives-Corrons, J. L., and Franco, R. (1991) Presence of adenosine
deaminase on the surface of mononuclear blood cells: immunochemical
localization using light and electron microscopy, J. Histochem.
Cytochem., 39, 1001-1008.
24.Bradford, M. M. (1976) A rapid and sensitive
method for the quantitation of microgram quantities of protein
utilizing the principle of protein-dye binding, Anal.
Biochem., 72, 248-254.
25.Heldsinger, A. A., Vinik, A. I., and Fox, I. H.
(1986) Inhibition of guinea-pig oxyntic cell function by adenosine and
prostaglandins, J. Pharmacol. Exp. Ther., 237,
351-356.
26.Gerber, J. G., Nies, A. S., and Payne, N. A.
(1985) Adenosine receptors on canine parietal cells modulate gastric
acid secretion to histamine, J. Pharmacol. Exp. Ther.,
233, 623-627.
27.Yip, L., and Kwok, Y. N. (2004) Role of adenosine
A2A receptor in the regulation of gastric somatostatin release, J.
Pharmacol. Exp. Ther., 309, 804-815.
28.Yang, G. K., Chen, J. F., Kieffer, T. J., and
Kwok, Y. N. (2009) Regulation of somatostatin release by adenosine in
the mouse stomach, J. Pharmacol. Exp. Ther., 329,
729-737.
29.Tan-Allen, K. Y., Sun, X. C., and Bonanno, J. A.
(2005) Characterization of adenosine receptors in bovine corneal
endothelium, Exp. Eye Res., 80, 687-696.
30.Olanrewaju, H. A., Qin, W., Feoktistov, I.,
Scemama, J. L., and Mustafa, S. J. (2000) Adenosine A(2A) and A(2B)
receptors in cultured human and porcine coronary artery endothelial
cells, Am. J. Physiol. Heart Circ. Physiol., 279,
H650-656.
31.Linden, J., Thai, T., Figler, H., Jin, X., and
Robeva, A. S. (1999) Characterization of human A(2B) adenosine
receptors: radioligand binding, Western blotting, and coupling to G(q)
in human embryonic kidney 293 cells and HMC-1 mast cells, Mol.
Pharmacol., 56, 705-713.
32.Richard, E., Alam, S. M., Arredondo-Vega, F. X.,
Patel, D. D., and Hershfield, M. S. (2002) Clustered charged amino
acids of human adenosine deaminase comprise a functional epitope for
binding the adenosine deaminase complexing protein CD26/dipeptidyl
peptidase IV, J. Biol. Chem., 277,
19720-19726.
33.Beraudi, A., Traversa, U., Villani, L., Sekino,
Y., Nagy, J. I., and Poli, A. (2003) Distribution and expression of A1
adenosine receptors, adenosine deaminase and adenosine
deaminase-binding protein (CD26) in goldfish brain, Neurochem.
Int., 42, 455-464.
34.Sakai, H., Okada, Y., Morii, M., and Takeguchi,
N. (1989) Anion and cation channels in the basolateral membrane of
rabbit parietal cells, Pflugers Arch., 414,
185-192.
35.Karam, S. M. (2010) A focus on parietal cells as
a renewing cell population, World J. Gastroenterol.,
16, 538-546.
36.Chew, C. S. (1994) Parietal cell culture: new
models and directions, Annu. Rev. Physiol., 56,
445-461.
37.Miller, M. L., Andringa, A., Zavros, Y.,
Bradford, E. M., and Shull, G. E. (2010) Volume density, distribution,
and ultrastructure of secretory and basolateral membranes and
mitochondria predict parietal cell secretory (dys)function, J.
Biomed. Biotechnol., 2010, 394198.
38.Chew, C. S., Chen, X., Parente, J. A., Jr.,
Tarrer, S., Okamoto, C., and Qin, H. Y. (2002) Lasp-1 binds to
non-muscle F-actin in vitro and is localized within multiple
sites of dynamic actin assembly in vivo, J. Cell.
Sci., 115, 4787-4799.
39.Lapierre, L. A., Avant, K. M., Caldwell, C. M.,
Ham, A. J., Hill, S., Williams, J. A., Smolka, A. J., and Goldenring,
J. R. (2007) Characterization of immunoisolated human gastric parietal
cells tubulovesicles: identification of regulators of apical recycling,
Am. J. Physiol. Gastrointest. Liver Physiol., 292,
G1249-1262.
40.Geibel, J., Abraham, R., Modlin, I., and Sachs,
G. (1995) Gastrin-stimulated changes in Ca2+ concentration
in parietal cells depends on adenosine 3′,5′-cyclic
monophosphate levels, Gastroenterology, 109,
1060-1067.