Influence of Antioxidant SkQ1 on Accumulation of Mitochondrial DNA Deletions in the Hippocampus of Senescence-Accelerated OXYS Rats
P. S. Loshchenova1, O. I. Sinitsyna1,2, L. A. Fedoseeva1, N. A. Stefanova1, and N. G. Kolosova1,2,3*
1Institute of Cytology and Genetics, Siberian Division of the Russian Academy of Sciences, pr. Lavrent’eva 10, 630090 Novosibirsk, Russia; E-mail: kolosova@bionet.nsc.ru2Novosibirsk State University, ul. Pirogova 2, 630090 Novosibirsk, Russia
3Lomonosov Moscow State University, Institute of Mitoengineering, 119991 Moscow, Russia
* To whom correspondence should be addressed.
Received December 16, 2014; Revision received January 9, 2015
Reduction of efficiency of oxidative phosphorylation associated with aging and the development of neurodegenerative diseases including Alzheimer’s disease is thought to be linked to the accumulation of deletions in mitochondrial DNA (ΔmtDNA), which are seen as a marker of oxidative damage. Recently, we have shown that mitochondria-targeted antioxidant SkQ1 (10-(6′-plastoquinonyl)decyltriphenylphosphonium) can slow the development of signs of Alzheimer’s disease in senescence-accelerated OXYS rats. The purpose of this study was to explore the relationship between the development of neurodegenerative changes in the brain of OXYS rats and changes in the amount of mtDNA and the 4834-bp mitochondrial DNA deletion (ΔmtDNA4834) as well as the effect of SkQ1. We studied the relative amount of mtDNA and ΔmtDNA4834 in the hippocampus of OXYS and Wistar (control) rats at ages of 1, 2, 6, 10, and 20 days and 3, 6, and 24 months. During the period crucial for manifestation of the signs of accelerated aging of OXYS rats (from 1.5 to 3 months of age), we evaluated the effects of administration of SkQ1 (250 nmol/kg) and vitamin E (670 mmol/kg, reference treatment) on the amount of mtDNA and ΔmtDNA4834 and on the formation of the behavioral feature of accelerated senescence in OXYS rats – passive type of behavior in the open field test. In OXYS rats, the level of ΔmtDNA4834 in the hippocampus is increased compared to the Wistar rats, especially at the stage of completion of brain development in the postnatal period. This level remains elevated not only at the stages preceding the manifestation of the signs of accelerated brain aging and the development of pathological changes linked to Alzheimer’s disease, but also during their progression. However, at age of 24 months, there were no detectable differences between the two strains. SkQ1 treatment reduced the level of ΔmtDNA4834 in the hippocampus of Wistar and OXYS rats and slowed the formation of passive behavior in OXYS rats. These results support the possible use of SkQ1 for prophylaxis of brain aging.
KEY WORDS: aging, hippocampus, mitochondria, 4834-bp mitochondrial DNA deletion, mitochondria-targeted antioxidant SkQ1DOI: 10.1134/S0006297915050120
Abbreviations: bp, base pairs; mtDNA, mitochondrial DNA; ΔmtDNA, deletion in mitochondrial DNA; ΔmtDNA4834, 4834-bp mitochondrial DNA deletion; ROS, reactive oxygen species; SkQ1, antioxidant 10-(6′-plastoquinonyl)decyltriphenylphosphonium.
Human and animal aging is associated with gradual decline of cognitive
functions (especially learning ability and memory) and increased risk
of development of neurodegenerative diseases [1].
Mutations of mitochondrial DNA (mtDNA) and related mitochondrial
dysfunction play an important role in their development [2]. The proportion of mtDNA with deletions increases
with age. Their accumulation is most significant in tissues with a high
level of energy metabolism – muscles and brain – where the
level of deletions significantly depends on the region [3]. Accumulation of mtDNA deletions is considered as
one of the causes of the age-related decrease in the efficiency of
oxidative phosphorylation. It also affects age-related development of
neurodegenerative diseases, including Parkinson’s [4] and Alzheimer’s [5]
diseases. In turn, decrease in the efficiency of the work of the
respiratory chain leads to increased accumulation of mtDNA oxidative
damage and deletions due to oxidative stress – imbalance in
the systems of generation and detoxication of reactive oxygen species
(ROS). Hence, it is logical that antioxidants are widely used to
prevent age-related changes. However, so far there has been no
compelling evidence that antioxidants provide protection against
neurodegenerative changes accompanying aging-related cognitive
dysfunctions, and no studies have indicated that they can prevent
neurodegenerative diseases. Recently, it has been shown that the
mitochondria-targeted antioxidant SkQ1 has unique neuroprotective
potential [6-8]. Nanomolar
concentrations of this compound not only prevented, but also reduced
the severity of a number of signs of accelerated aging in OXYS
rats – a model of premature aging and aging-related diseases
created in the Institute of Cytology and Genetics, Siberian Branch of
the Russian Academy of Sciences [6, 9-16]. Development of
neurodegenerative changes in OXYS rats is associated with changes in
the metabolic pathway of Alzheimer’s disease: enhanced
accumulation in the cortex and hippocampus of the protein precursor of
beta amyloid and soluble amyloid β (1-42), formation of amyloid
plaques, and hyperphosphorylation of the tau protein – key
markers of the disease [7, 17]. In the case of OXYS rats, hyperphosphorylation
of the tau protein is registered already at the age of 3 months. By
this age, OXYS rats develop passive type behavior, increased anxiety,
impaired learning ability and memory, and prolonged post-tetanic
potentiation; MRI methods reveal neurodegenerative changes in the brain
[7, 17]. It is noteworthy that
manifestation of all these signs predates the increased accumulation of
oxidative stress markers, oxidized proteins, and lipids in brain
homogenates [18]. Recently, we showed that
prophylactic SkQ1 administration significantly slows accelerated brain
aging in OXYS rats and reduces the content of the key markers of
Alzheimer’s disease in it [7]; however, the
mechanism of its neuroprotective effect remains unclear.
According to Meissner et al., accumulation of deletions of mtDNA 4977-bp can serve as an ideal criterion of the rate of human aging and, hence, the efficiency of any agents affecting this rate [19]; primarily this criterion should be used in the cells of postmitotic tissues characterized by high energy consumption, muscle and nerve tissues [20, 21]. When modeling Alzheimer’s disease by administration of β-amyloid, increase in the amount of ΔmtDNA4834 was registered in rat hippocampus [22]. This is the deletion that corresponds to the deletion of 4977 bp of mtDNA in humans and results from recombination between two 16-nucleotide repeats (positions 8103-8118 and 12,937-12,952), leading to elimination of a number of genes: mt-nd5, mt-Tl, mt-Ts, mt-Th, mt-nd4, mt-nd4l, mt-Tr, mt-nd3, mt-Tg, mt-co3, mt-Atp6 [20, 23]. The purpose of this work was to study the relationship between the development of neurodegenerative changes in the brain of OXYS rats and the changes in the amount of mtDNA and ΔmtDNA4834, as well as the effect of SkQ1 on these parameters. To do this, we determined the relative amount of ΔmtDNA4834 and mtDNA in the hippocampus of OXYS and Wistar (control) rats of different ages and also studied the effect of SkQ1 administration on these parameters (SkQ1 was administered during the period critical for the manifestation of the signs of accelerated aging of OXYS rats – from the age of 1.5 to 3 months). We used vitamin E as a reference preparation. The effect of antioxidants on the accumulation of ΔmtDNA4834 was compared to their ability to prevent formation of behavioral sign of accelerated aging of OXYS rat brain – passive behavior in the “open field” test.
MATERIALS ANS METHODS
Animals and work with them. The work was carried out on male OXYS and Wistar rats based on the Center for Genetic Resources of Laboratory Animals at the Institute of Cytology and Genetics, Siberian Branch of the Russian Academy of Sciences (RFMEFI61914X0005 and RFMEFI61914X0010), in accordance with the “Rules of Work with Laboratory Animals”. The rats were kept in groups of five in 57 × 36 × 20-cm cages at temperature 22 ± 2°C with fixed mode of illumination (12 h light/12 h darkness) and free access to water and food, the standard pelleted chow for laboratory animals (Chara, Assortiment-Agro, Russia).
To study age-related changes in ΔmtDNA4834 and mtDNA content in hippocampus of OXYS and Wistar rats, we used animals aged 1, 2, 6, 10, 20 days and 3, 6, 24 months (5 animals per group). To evaluate the effects of antioxidants on these parameters, animals from experimental groups received with food SkQ1 (250 nmol per kg of body weight; synthesized in Moscow State University Research Institute of Mitoengineering) or vitamin E (tocopherol acetate; 670 mmol per kg of body weight; produced by Uralbiopharm, Russia) at the age from 1.5 to 3 months. Rats from the control group received only food. Each group contained 15 animals.
The effects of preparations on animal behavior in the “open field” test were studied on 3-month-old animals using a square chamber (100 × 100 cm) with plastic walls 40 cm high. A shadowless 100 W lamp located 100 cm above the field center provided illumination. The animal was placed in the corner of the chamber, and its locomotor activity was recorded for 5 min. The number of crossed squares and number of rearings were counted.
Extraction of total DNA from the hippocampus. Total (mitochondrial and nuclear) hippocampal DNA was extracted using the set WizardR Plus SV Genomic DNA Purification System (Promega, USA) according to the manufacturer’s protocol.
Real-time PCR. The relative amount of mtDNA and ΔmtDNA4834 were determined by real-time PCR using TaqMan probes. The method described by Nicklas et al. [24] was used to determine mtDNA directly in total DNA. The amount of mtDNA (using amplification of the D-loop region) was measured against the amount of nuclear DNA determined by the content of DNA of gene 18S rRNA. The amount of ∆mtDNA4834 (amplification of the region formed as a result of closing of the sequences flanking the deleted mtDNA site) was measured against the amount of mtDNA. Sequences of primers and probes selected using an Integrated DNA Technologies online oligo-analyzer (http://eu.idtdna.com/PrimerQuest/) are shown in the table. Oligonucleotides were synthesized in Biosan (Russia).
Primers and probes used in PCR
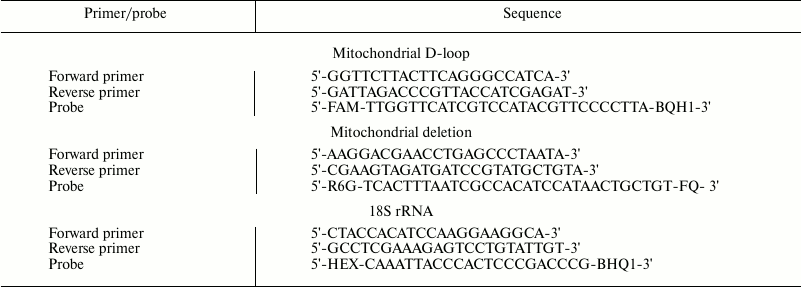
The reaction mixture (20 µl) contained 1× buffer for Taq-polymerase, 5 mM MgCl2, 200 nM dNTP (25 mM each), 400 nM of probe for D-loop and corresponding primers (forward and reverse), 0.2 U Taq Pol, and ~1 ng of the total DNA. Similar reaction mixture for 18S rRNA gene and mixture for ΔmtDNA4834 contained, respectively, 400 and 500 nM of corresponding primers and probe. The reaction was conducted under the following conditions: preheating 95°C – 30 s followed by 40 basic cycles: denaturation 94°C – 10 s, annealing 64°C – 15 s, elongation 72°C – 20 s. In each experiment, test samples of DNA with primers and probes to the D-loop (four repetitions per each DNA sample) and 18S rRNA gene (also four repetitions per sample) were placed on the same plate; the same pattern was followed in case of ΔmtDNA4834 and D-loop; to make a calibration curve, a standard DNA template (dilutions 1 : 1, 1 : 4, 1 : 16, and 1 : 64) with the same primers and probes (two repetitions per dilution) were placed on each plate. For each DNA sample, PCR was performed at least twice.
The same DNA sample for each reaction series was used as a standard DNA template for building calibration curves. Relative DNA amount was determined using calibration curves obtained based on standard DNA dilutions. Thus received standard calibrating curves were used to determine the initial level of target DNA (against the “standard” DNA), and this value for mtDNA was compared to nuclear DNA, and level of deleted mtDNA – to mtDNA.
Statistical analysis of the results was performed using the STATISTICA (version 6.0) software package. We used factorial analysis of variance (ANOVA) with post-hoc comparison of group averages (Newman–Keuls test). We considered the following parameters as independent factors: animal age and genotype on the analysis of age-related changes in the amount of mtDNA and ΔmtDNA4834; genotype and preparation at evaluation of antioxidant effects. Differences were considered significant at p < 0.05. Data are presented as M ± S.E.M.
RESULTS
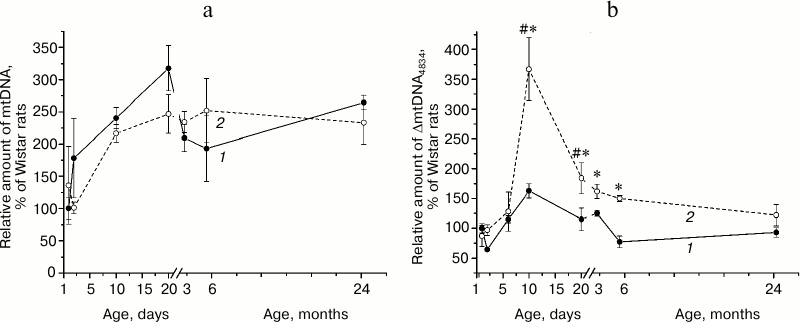
Amount of mtDNA and ΔmtDNA4834 in the hippocampus of rats of different ages. Hippocampal mtDNA content (Fig. 1a) did not depend on animal genotype (F1,36 = 0.23, p = 0.63) and changed with age (F7,36 = 9.41, p < 0.00001), these changes being most pronounced in the first days of life. This parameter was higher on the 20th day after birth than on the first day: in Wistar rats – 3 times (p < 0.004), in OXYS rats – 2 times (p < 0.025). By the age of 3 months the parameter somewhat decreased and remained at the same level at the age of 6 and 24 months. No significant interline difference in mtDNA content was detected in any of the age groups.
Amount of ΔmtDNA4834 (Fig. 1b) depended on animal age (F7,36 = 6.84, p < 0.00003) and genotype (F1,36 = 12.75, p < 0.001) and was significantly higher in OXYS rats. The maximum amount of deleted mtDNA was detected in rats of both lines at the age of 10 days; in OXYS rats this parameter was 3.5 times higher than in the first day after birth (p < 0.0001) and 2 times higher than in Wistar rats of the same age (p < 0.0002). By the age of 20 days, the amount of hippocampal ΔmtDNA4834 in OXYS rats decreased (p < 0.0001) and after that basically did not change, remaining higher than in Wistar rats (p < 0.05 for all ages) until the age of 24 months, when interline differences leveled (Fig. 1b).
Fig. 1. Age-related changes in the relative amount of mtDNA (a) and ΔmtDNA4834 (b) in the hippocampus of Wistar (1) and OXYS (2) rats. 100% refer to the corresponding figure in 1-day-old Wistar rats. * Reliable differences between rat lines; # reliable differences from the rats of the same line of the previous age group.
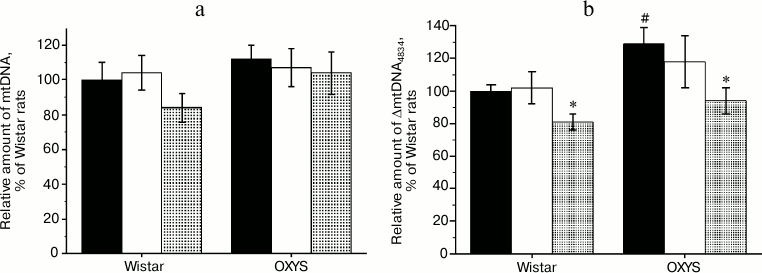
Effects of SkQ1 and vitamin E on amount of ΔmtDNA4834 and mtDNA in the hippocampus of Wistar and OXYS rats. As shown by the analysis of variance, the content of mtDNA (Fig. 2a) did not depend on animal genotype (F1,30 = 1.58, p = 0.219), and administration of antioxidants had no effect on it (F2,30 = 1.15, p = 0.341). Relative amount of ΔmtDNA4834 (Fig. 2b) was higher in OXYS rats (F1,30 = 5.77, p < 0.023), and it was affected by the factor “preparation” (F2,30 = 4.43, p < 0.021). However, comparison of group averages showed that only the SkQ1 effect was significant: only in case of SkQ1 administration the level of ΔmtDNA4834 in Wistar and OXYS rats was lower than in control animals of the respective line (p < 0.05).
Fig. 2. Relative amount of mtDNA (a) and ΔmtDNA4834 (b) in the hippocampus of control (black columns) and treated with vitamin E (white columns) and SkQ1 (gray columns) Wistar and OXYS rats. The animals received vitamin E and SkQ1 daily between 1.5 and 3 months of age (670 mmol/kg and 250 nmol/kg, respectively). * Reliable effect of the preparation; # reliable interline differences.
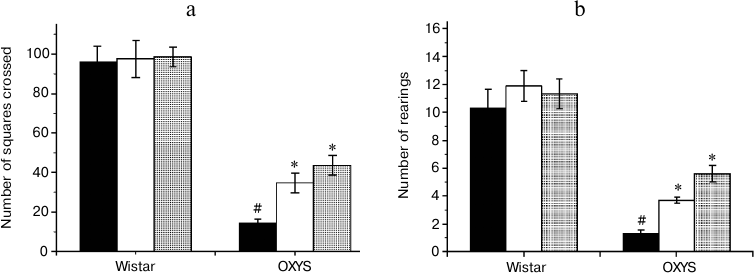
Effects of SkQ1 and vitamin E on behavior of OXYS and Wistar rats in the “open field” test. The number of squares crossed (Fig. 3a), an indicator of the animal motor activity, was significantly lower in 3-month-old OXYS rats than in Wistar rats of the same age (F1,63 = 187.6, p = 0.0001). The preparations affected the animals’ motor activity (F2,63 = 6.6, p = 0.003), but comparison of group averages showed that this parameter was significantly changed only in the OXYS rats: the number of squares crossed in rats receiving SkQ1 and vitamin E was 3 and 2.4 times higher than in rats from the control group (p < 0.001 for all animals).
The number of rearings (Fig. 3b) reflects both motor and exploratory activities of the animals. This parameter was lower in OXYS than in Wistar rats (F1,62 = 71.2, p = 0.0001), and the “preparation” factor affected it (F2,63 = 4.9, p = 0.01). However, post-hoc comparisons of group averages have shown that antioxidants significantly affected only the behavior of OXYS rats: in case of OXYS rats receiving SkQ1 and vitamin E, the number of rearings was, respectively, 4.2 and 2.8 times higher than in control group (p < 0.001 for all animals).
Fig. 3. Locomotor (а) and exploratory (b) activities of control (black columns) and treated with vitamin E (white columns) and SkQ1 (gray columns) Wistar and OXYS rats. The animals received vitamin E and SkQ1 daily between 1.5 and 3 months of age (670 mmol/kg and 250 nmol/kg, respectively). * Reliable effect of the preparation; # reliable interline differences.
Thus, analysis of the behavior of OXYS and Wistar rats in the open field test revealed the ability of SkQ1 and vitamin E to prevent age-dependent decrease in motor and exploratory activities in OXYS rats; these compounds had no effect on the same parameters of young Wistar rats.
DISCUSSION
Deletions of mtDNA are considered as a marker of oxidative damages, the number of which increases with age [2]. However, according to recent studies [25, 26] accumulation of mutations in mtDNA can affect the lifespan of only long-lived species (including humans), but not mice and rats. There is no available information on the dynamics of their accumulation in brain in the postnatal period. We only know that ΔmtDNA were identified in all studied postmortem brain samples of newborns and, according to the authors, could be caused by perinatal hypoxia and intensive therapy [27]. We evaluated the level of hippocampal ΔmtDNA4834 in postnatal period and periods of active manifestation (3 months) and progression (6 months) of signs of accelerated brain aging in OXYS rats, as well as at the age of 24 months, when all the signs of Alzheimer’s disease are most pronounced [7, 17]. It is noteworthy that the level of ΔmtDNA4834 was found to be maximal at the age of 10 days in both senescence-accelerated OXYS rats and Wistar rats. At this age, it was significantly higher than on the first day of life, and by the 20th day, this parameter decreased, while the number of mtDNA copies increased. Significant increase in ΔmtDNA4834 was observed during the period of adaptation to extra-uterine life. In humans, it lasts 28 days after birth, in rats – 14 days. During this period, a number of processes (proliferation, differentiation, migration, etc.) take place in mammalian brain. These processes determine the final maturation of the nervous system. After birth, many brain regions, including hippocampus, undergo development and histogenesis associated with the formation of interneuronal contacts and elimination of “transitional” cell populations by means of apoptosis [28]. High apoptotic activity and, according to the latest data, neurogenesis, suggest enhanced ROS generation [29]. It is at this age that the level of hippocampal ΔmtDNA4834 in OXYS rats was the highest compared to Wistar rats (3.5-fold difference). By the age of 3 months, OXYS rats demonstrate characteristic passive type of behavior, distorted learning ability and memory, and the first histomorphological signs of neurodegeneration that progress with age. The present study has shown that these events take place against a background of increased level of ΔmtDNA4834 in the hippocampus. However, at the age of 24 months, when all the signs of Alzheimer’s disease are fully pronounced in OXYS rats [17], no differences from Wistar rats were found in this parameter. Such a result, as well as the fact that the level of hippocampal ΔmtDNA4834 in both Wistar and OXYS rats of this age was no different from that of 3- and 6-month-old animals, first seem to be unexpected. However, it is consistent with data on increased ΔmtDNA4834 accumulation in the course of Alzheimer’s disease, but is does not correlate with the progression of the disease [30].
The results of the present study indirectly indicate that the development of OXYS rat brain in the early postnatal period proceeds under increased oxidative stress. This stress might be associated with hypoxia (we have earlier found the symptoms of adaptation to hypoxia in the brain of 2-3-week-old OXYS rats, when studying energy metabolism) [31]. As noted above, we found no differences from the control Wistar rats when evaluating markers of oxidative stress – level of oxidative damages of proteins and lipids in brain homogenates of young OXYS rats [17, 18]. At the same time, differential evaluation of the activity of free radical processes in various brain structures based on the level of products of lipid peroxidation has shown that it is in the hippocampus of 2- and 18-month-old OXYS rats that it is higher than in Wistar rats [32], which is consistent with the results of the present study.
Age-related increase in mitochondrial dysfunctions was first identified in the liver of OXYS rats, and it is considered to be a possible reason for their accelerated aging [33, 34]. Later structural and functional mitochondrial disorders were found in muscles [13] and hippocampus [17]: destruction of the cristae, matrix lysis, and reduction of the volume and surface density of mitochondria are detected already in 3-month-old OXYS rats, and these phenomena increase with age. Administration of SkQ1 not only slowed the development of destructive changes of mitochondria in muscles [13] and hippocampus (unpublished data), but also significantly improved their condition in Wistar and OXYS rats with already pronounced signs of accelerated aging. Long-term administration of SkQ1 prevented age-related accumulation of amyloid-β, increase in the level of protein precursor of the amyloid, τ-protein, and its phosphorylated form in OXYS rats to their level in Wistar rats of the same age [7].
In the present study, we have shown that prophylactic administration of SkQ1 during the period of active manifestation of the signs of accelerated aging in OXYS rats slowed the development of its behavioral manifestations: it increased locomotor and exploratory activities and reduced the level of ΔmtDNA4834 in hippocampus. SkQ1 also reduced the level of ∆mtDNA4834 in hippocampus of Wistar rats, but it had no effect on their behavior. This decrease in ΔmtDNA4834 accumulation could be caused by the direct antioxidant effect of SkQ1 [35] as well as by its ability to suppress ROS generation in mitochondria due to potentiation of mild uncoupling of oxidation and phosphorylation caused by fatty acids [36]. It should be noted that the reference preparation, vitamin E, also increased (although to a lesser extent) locomotive and exploratory activities in OXYS rats, but it had no significant effect on ΔmtDNA4834 level. We could find no reliable effects of antioxidants on the level of mtDNA in the hippocampus.
Thus, the most significant increase in the level of hippocampal ΔmtDNA4834 in OXYS rats, compared to Wistar rats, is observed during the period of completion of the postnatal brain formation; it remains increased at the stages preceding manifestation of phenotypic signs of accelerated brain aging and the development of Alzheimer’s disease as well as during their progression. We have shown that mitochondria-targeted antioxidant SkQ1 slows the accelerated aging of brain of OXYS rats, reducing the level of hippocampal ΔmtDNA4834. However, it does not seem possible that the reduction of ΔmtDNA4834 level is solely responsible for the effect of SkQ1, because vitamin E, while having no effect on ΔmtDNA4834 level, also somewhat reduced the severity of behavioral manifestations of accelerated brain aging in OXYS rats. Overall, the results are consistent with the idea that oxidative stress and mitochondrial dysfunction contribute significantly to the pathogenesis of Alzheimer’s disease and confirm the promising perspective of using SkQ1 for prophylaxis of brain aging.
The authors express their gratitude to V. P. Skulachev for critical discussion and long-term support of their research.
This work was financially supported by the Russian Foundation for Basic Research (grants 12-04-01495 and 15-04-06066), State Budget Project No VI.60.1.1, and the Interdisciplinary integration project of the Russian Academy of Sciences.
REFERENCES
1.Morley, J. E., Armbrecht, H. J., Farr, S. A., and
Kumar, V. B. (2012) The senescence accelerated mouse (SAMP8) as a model
for oxidative stress and Alzheimer’s disease, Biochim.
Biophys. Acta, 1822, 650-656.
2.Li, H., Liu, D., Lu, J., and Bai, Y. (2012)
Physiology and pathophysiology of mitochondrial DNA, Adv. Exp. Med.
Biol., 942, 39-51.
3.Kazachkova, N., Ramos, A., Santos, C., and Lima, M.
(2013) Mitochondrial DNA damage patterns and aging: revising the
evidences for humans and mice, Aging Dis., 4,
337-350.
4.Zhang, J., Montine, T. J., Smith, M. A., Siedlak,
S. L., Gu, G., Robertson, D., and Perry, G. (2002) The mitochondrial
common deletion in Parkinson’s disease and related movement
disorders, Parkinson. Rel. Disord., 8, 165-170.
5.Krishnan, K., Ratnaike, T., De Gruyter, H., Jaros,
E., and Turnbull, D. (2012) Mitochondrial DNA deletions cause the
biochemical defect observed in Alzheimer’s disease, Neurobiol.
Aging, 33, 2210-2214.
6.Stefanova, N. A., Fursova, A. Zh., and Kolosova, N.
G. (2010) Behavioral effects induced by mitochondria-targeted
antioxidant SkQ1 in Wistar and senescence-accelerated OXYS rats, J.
Alzheimers Dis., 21, 479-491.
7.Stefanova, N. A., Muraleva, N. A., Skulachev, V.
P., and Kolosova, N. G. (2014) Alzheimer’s disease-like pathology
in senescence-accelerated OXYS rats can be partially retarded with
mitochondria-targeted antioxidant SkQ1, J. Alzheimers Dis.,
38, 681-694.
8.Kapay, N. A., Popova, O. V., Isaev, N. K.,
Stelmashook, E. V., Kondratenko, R. V., Zorov, D. B., Skrebitsky, V.
G., and Skulachev, V. P. (2013) Mitochondria-targeted plastoquinone
antioxidant SkQ1 prevents amyloid-β-induced impairment of
long-term potentiation in rat hippocampal slices, J. Alzheimers
Dis., 36, 377-383.
9.Kolosova, N. G., Stefanova, N. A., Korbolina, E.
E., and Fursova, A. Zh. (2014) Senescence-accelerated OXYS rats: a
genetic model of premature aging and age-related diseases, Adv.
Gerontol., 4, 294-298.
10.Amstislavskaya, T. G., Maslova, L. N., Gladkikh,
D. V., Belousova, I. I., Stefanova, N. A., and Kolosova, N. G. (2010)
Effects of the mitochondria-targeted antioxidant SkQ1 on sexually
motivated behavior in male rats, Pharmacol. Biochem. Behav.,
96, 211-216.
11.Neroev, V. V., Archipova, M. M., Bakeeva, L. E.,
Fursova, A. Zh., Grigorian, E. N., Grishanova, A. Yu., Iomdina, E. N.,
Ivashchenko, Zh. N., Katargina, L. A., Khoroshilova-Maslova, I. P.,
Kilina, O. V., Kolosova, N. G., Kopenkin, E. P., Korshunov, S. S.,
Kovaleva, N. A., Novikova, Yu. P., Philippov, P. P., Pilipenko, D. I.,
Robustova, O. V., Saprunova, V. B., Senin, I. I., Skulachev, M. V.,
Sotnikova, L. F., Stefanova, N. A., Tikhomirova, N. K., Tsapenko, I.
V., Shchipanova, A. I., Zinovkin, R. A., and Skulachev, V. P. (2008)
Mitochondria-targeted plastoquinone derivatives as tools to interrupt
execution of the aging program. 4. Age-related eye disease. SkQ1
returns vision to blind animals, Biochemistry (Moscow),
73, 1317-1328.
12.Skulachev, V. P., Anisimov, V. N., Antonenko, Y.
N., Bakeeva, L. E., Chernyak, B. V., Erichev, V. P., Filenko, O. F.,
Kalinina, N. I., Kapelko, V. I., Kolosova, N. G., Kopnin, B. P.,
Korshunova, G. A., Lichinitser, M. R., Obukhova, L. A., Pasyukova, E.
G., Pisarenko, O. I., Roginsky, V. A., Ruuge, E. K., Senin, I. I.,
Severina, I. I., Skulachev, M. V., Spivak, I. M., Tashlitsky, V. N.,
Tkachuk, V. A., Vyssokikh, M. Y., Yaguzhinsky, L. S., and Zorov, D. B.
(2009) An attempt to prevent senescence: a mitochondrial approach,
Biochim. Biophys. Acta, 1787, 437-461.
13.Vays, V. B., Eldarov, C. M., Vangely, I. M.,
Kolosova, N. G., Bakeeva, L. E., and Skulachev, V. P. (2014)
Antioxidant SkQ1 delays sarcopenia-associated damage of mitochondrial
ultrastructure, Aging (Albany, NY), 6,
140-148.
14.Saprunova, V. B., Lelekova, M. A., Kolosova, N.
G., and Bakeeva, L. E. (2012) SkQ1 slows development of age-dependent
destructive processes in retina and vascular layer of eyes of Wistar
and OXYS rats, Biochemistry (Moscow), 77,
648-658.
15.Markovets, A. M., Fursova, A. Z., and Kolosova,
N. G. (2011) Therapeutic action of the mitochondria-targeted
antioxidant SkQ1 on retinopathy in OXYS rats linked with improvement of
VEGF and PEDF gene expression, PLoS One, 6, e21682.
16.Kolosova, N. G., Stefanova, N. A., Muraleva, N.
A., and Skulachev, V. P. (2012) The mitochondria-targeted antioxidant
SkQ1 but not N-acetylcysteine reverses aging-related biomarkers in
rats, Aging (Albany, NY), 4, 686-694.
17.Stefanova, N. A., Kozhevnikova, O. S., Vitovtov,
A. O., Maksimova, K. Y., Logvinov, S. V., Rudnitskaya, E. A.,
Korbolina, E. E., Muraleva, N. A., and Kolosova, N. G. (2014)
Senescence-accelerated OXYS rats: a model of age-related cognitive
decline with relevance to abnormalities in Alzheimer disease, Cell
Cycle, 13, 898-909.
18.Kolosova, N. G., Shcheglova, T. V., Sergeeva, S.
V., and Loskutova, L. V. (2006) Long-term antioxidant supplementation
attenuates oxidative stress markers and cognitive deficits in
senescent-accelerated OXYS rats, Neurobiol. Aging, 27,
1289-1297.
19.Meissner, C., Bruse, P., Mohamed, S. A., Schulz,
A., Warnk, H., Storm, T., and Oehmichen, M. (2008) The 4977 bp deletion
of mitochondrial DNA in human skeletal muscle, heart and different
areas of the brain: a useful biomarker or more, Exp. Gerontol.,
43, 645-652.
20.Gadaleta, M. N., Rainaldi, G., Lezza, A. M.,
Milella, F., Fracasso, F., and Cantatore, P. (1992) Mitochondrial DNA
copy number and mitochondrial DNA deletion in adult and senescent
rats, Mutat. Res., 275, 181-193.
21.DiMauro, S., Tanji, K., Bonilla, E., Pallotti,
F., and Schon, E. (2002) Mitochondrial abnormalities in muscle and
other aging cells: classification, causes, and effects, Muscle
Nerve, 26, 597-607.
22.Chen, T. F., Chiu, M. J., Huang, C. T., Tang, M.
C., Wang, S. J., Wang, C. C., and Huang, R. F. (2011) Changes in
dietary folate intake differentially affect oxidized lipid and
mitochondrial DNA damage in various brain regions of rats in the
absence/presence of intracerebroventricularly injected amyloid
β-peptide challenge, Br. J. Nutr., 105,
1294-1302.
23.Edris, W., Burgett, B., Colin, O., and Filburn,
C. (1994) Detection and quantitation by competitive PCR of an
age-associated increase in a 4.8-kb deletion in rat mitochondrial
DNA, Mutat. Res., 316, 69-78.
24.Nicklas, J., Brooks, E., Hunter, T., Single, R.,
and Branda, R. (2004) Development of a quantitative PCR (TaqMan) assay
for relative mitochondrial DNA copy number and the common mitochondrial
DNA deletion in the rat, Environ. Mol. Mutagen., 44,
313-320.
25.Kowald, A., and Kirkwood, T. B. (2013)
Mitochondrial mutations and aging: random drift is insufficient to
explain the accumulation of mitochondrial deletion mutants in
short-lived animals, Aging Cell, 12, 728-731.
26.Kowald, A., Dawson, M., and Kirkwood, T. B.
(2014) Mitochondrial mutations and ageing: can mitochondrial deletion
mutants accumulate via a size based replication advantage, J. Theor.
Biol., 340, 111-118.
27.Nadasi, E., Melegh, B., Seress, L., and
Kosztolanyi, G. (2004) Mitochondrial DNA deletions in newborn brain
samples, Orv. Hetil., 145, 1321-1325.
28.Kim, W. R., and Sun, W. (2011) Programmed cell
death during postnatal development of the rodent nervous system,
Dev. Growth Differ., 53, 225-235.
29.Walton, N. M., Shin, R., Tajinda, K., Heusner, C.
L., Kogan, J. H., Miyake, S., Chen, Q., Tamura, K., and Matsumoto, M.
(2012) Adult neurogenesis transiently generates oxidative stress,
PLoS One, 7, e35264.
30.Gerschutz, A., Heinsen, H., Grunblatt, E.,
Wagner, A. K., Bartl, J., Meissner, C., Fallgatter, A. J., Al-Sarraj,
S., Troakes, C., Ferrer, I., Arzberger, T., Deckert, J., Riederer, P.,
Fischer, M., Tatschner, T., and Monoranu, C. M. (2013) Neuron-specific
mitochondrial DANN deletion levels in sporadic Alzheimer’s
disease, Curr. Alzheimer Res., 10, 1041-1046.
31.Sergeeva, S., Bagryanskaya, E., Korbolina, E.,
and Kolosova, N. (2006) Development of behavioral dysfunctions in
accelerated-senescence OXYS rats is associated with early postnatal
alterations in brain phosphate metabolism, Exp. Gerontol.,
41, 141-150.
32.Kolosova, N. G., Shcheglova, T. V.,
Amstislavskaya, T. G., and Loskutova, L. V. (2003) Comparative analysis
of LPO products in brain structures of Wistar and OXYS rats of
different age, Bull. Exp. Biol. Med., 135, 593-596.
33.Shabalina, I. G., Kolosova, N. G., Grishanova, A.
Yu., Solovyov, V. N., Salganik, R. I., and Solovyova, N. A. (1995)
Oxidative phosphorylation activity, F0F1-ATPase,
and cytochrome content in liver mitochondria of rats with inherited
hyperproduction of free radicals, Biochemistry (Moscow),
60, 1563-1568.
34.Kolosova, N. G., Aydagulova, S. V.,
Nepomnyashchikh, G. I., Shabalina, I. G., and Shalbueva, N. I. (2001)
Dynamics of structural-functional changes in hepatocyte mitochondria of
prematurely aging OXYS rats, Bull. Eksp. Biol. Med.,
132, 235-240.
35.Skulachev, V. P., Antonenko, Y. N., Cherepanov,
D. A., Chernyak, B. V., Izyumov, D. S., Khailova, L. S., Klishin, S.
S., Korshunova, G. A., Lyamzaev, K. G., Pletjushkina, O. Y., Roginsky,
V. A., Rokitskaya, T. I., Severin, F. F., Severina, I. I., Simonyan, R.
A., Skulachev, M. V., Sumbatyan, N. V., Sukhanova, E. I., Tashlitsky,
V. N., Trendeleva, T. A., Vyssokikh, M. Y., and Zvyagilskaya, R. A.
(2010) Prevention of cardiolipin oxidation and fatty acid cycling as
two antioxidant mechanisms of cationic derivatives of plastoquinone
(SkQs), Biochim. Biophys. Acta, 1797, 878-889.
36.Plotnikov, E. Y., Silachev, D. N., Jankauskas, S.
S., Rokitskaya, T. I., Chupyrkina, A. A., Pevzner, I. B., Zorova, L.
D., Isaev, N. K., Antonenko, Y. N., Skulachev, V. P., and Zorov, D. B.
(2012) Mild uncoupling of respiration and phosphorylation as a
mechanism providing nephro- and neuroprotective effects of penetrating
cations of the SkQ family, Biochemistry (Moscow),
77, 1029-1037.