Low Concentrations of Uncouplers of Oxidative Phosphorylation Prevent Inflammatory Activation of Endothelial Cells by Tumor Necrosis Factor
V. P. Romaschenko1,2, R. A. Zinovkin1,3,4, I. I. Galkin1, V. V. Zakharova1,2, A. A. Panteleeva1, A. V. Tokarchuk1,2, K. G. Lyamzaev1,4, O. Yu. Pletjushkina1,4, B. V. Chernyak1,4, and E. N. Popova1,4*
1Lomonosov Moscow State University, Belozersky Institute of Physico-Chemical Biology, 119991 Moscow, Russia; fax: +7 (495) 939-0338; E-mail: k_popova_ch@mail.ru2Lomonosov Moscow State University, Faculty of Bioengineering and Bioinformatics, 119991 Moscow, Russia
3Lomonosov Moscow State University, Faculty of Biology, 119991 Moscow, Russia
4Lomonosov Moscow State University, Institute of Mitoengineering, 119991 Moscow, Russia
* To whom correspondence should be addressed.
Received December 25, 2014; Revision received January 12, 2015
In endothelial cells, mitochondria play an important regulatory role in physiology as well as in pathophysiology related to excessive inflammation. We have studied the effect of low doses of mitochondrial uncouplers on inflammatory activation of endothelial cells using the classic uncouplers 2,4-dinitrophenol and 4,5,6,7-tetrachloro-2-trifluoromethylbenzimidazole, as well as the mitochondria-targeted cationic uncoupler dodecyltriphenylphosphonium (C12TPP). All of these uncouplers suppressed the expression of E-selectin, adhesion molecules ICAM1 and VCAM1, as well as the adhesion of neutrophils to endothelium induced by tumor necrosis factor (TNF). The antiinflammatory action of the uncouplers was at least partially mediated by the inhibition of NFκB activation due to a decrease in phosphorylation of the inhibitory subunit IκBα. The dynamic concentration range for the inhibition of ICAM1 expression by C12TPP was three orders of magnitude higher compared to the classic uncouplers. Probably, the decrease in membrane potential inhibited the accumulation of penetrating cations into mitochondria, thus lowering the uncoupling activity and preventing further loss of mitochondrial potential. Membrane potential recovery after the removal of the uncouplers did not abolish its antiinflammatory action. Thus, mild uncoupling could induce TNF resistance in endothelial cells. We found no significant stimulation of mitochondrial biogenesis or autophagy by the uncouplers. However, we observed a decrease in the relative amount of fragmented mitochondria. The latter may significantly change the signaling properties of mitochondria. Earlier we showed that both classic and mitochondria-targeted antioxidants inhibited the TNF-induced NFκB-dependent activation of endothelium. The present data suggest that the antiinflammatory effect of mild uncoupling is related to its antioxidant action.
KEY WORDS: inflammation, endothelium, adhesion molecules, mitochondria, uncoupling of oxidative phosphorylation, penetrating cations of SkQ familyDOI: 10.1134/S0006297915050144
Abbreviations: C12TPP, dodecyltriphenylphosphonium; DNP, 2,4-dinitrophenol; FCS, fetal calf serum; mtDNA, mitochondrial DNA; NAC, N-acetylcysteine; nDNA, nuclear DNA; ROS, reactive oxygen species; SkQ1, plastoquinolyl-10(6′-decyltriphenyl)phosphonium; TMRM, tetramethylrhodamine methyl ester; TNF, tumor necrosis factor; TTFB, 4,5,6,7-tetrachloro-2-trifluoromethylbenzimidazole.
Enhanced level of tumor necrosis factor (TNF) in blood causes abnormal
activation and damage of endothelium (endothelial dysfunctions) [1-4]. One of the basic mechanisms
of endothelial activation under the influence of inflammatory cytokines
consists in the induction of expression of adhesion molecules (such as
ICAM1 and VCAM1) and selectins (such as E-selectin and P-selectin) on
the endothelial cell surface. These molecules ensure the adhesion and
subsequent transmigration of leukocytes across the vascular barrier
into tissues [5]. The major regulator of the
expression of adhesion molecules under the influence of TNF is
transcription factor NFκB [6].
The studies of recent years have shown that mitochondria modulate the response of different cells to inflammatory stimuli [7-9]. In endothelial cells, mitochondria, not being the main source of ATP [10], play the most important signaling role in both normal and pathological states of endothelium [9, 11-13]. It is supposed that the mitochondria of endothelial cells are homeostatic regulators of the calcium signaling system [14], as well as NO and reactive oxygen species (ROS) generation [15]. It has been shown that TNF-induced endothelial activation decreases under the influence of the inhibitor of the first complex of the mitochondrial respiratory chain and the inhibitors of mitochondrial pore opening [16]. ROS play a key role in vascular physiology and pathophysiology [17]. Previously, we have shown that mitochondrial ROS in endothelial cells transmit the signal of apoptosis induced by high doses of TNF [18]. It is known that TNF induces the production of ROS, including mitochondrial ROS [19, 20]. Recently, we have shown [21] that the expression of adhesion molecules in aortas is reduced in old mice administered for a long time with the mitochondrial-targeted antioxidant SkQ1 (plastoquinolyl-10(6′-decyltriphenyl)phosphonium). In endothelial cells, SkQ1, as well as classic antioxidants in 1000-fold higher concentrations, inhibited the TNF-dependent activation of NFκB [21]. The activation of NF-κB can be regulated by different redox-sensitive components of the signaling pathways; however, contradictory data obtained in different laboratories prevent obtaining a complete picture of what is going on here [22].
It is known that a slight decrease in mitochondrial membrane potential without stopping ATP synthesis (so-called “mild” uncoupling) can substantially reduce ROS generation in mitochondria [23-25]. In endothelial cells, both the uncouplers of oxidative phosphorylation and the enhanced expression of mitochondrial uncoupling proteins (UCPs) decreased ROS generation by mitochondria [26-29]. In an ex vivo model on the coronary arteries of old rats, the uncoupler carbonyl cyanide p-trifluoromethoxyphenylhydrazone (FCCP) reduced the mitochondrial oxidative stress and NFκB-dependent expression of adhesion molecules [30]. It is supposed that uncouplers may act in the cells, first, by preventing hyperpolarization of mitochondria and thereby suppressing ROS generation [25] and, second, by inducing the expression of antioxidant enzymes [31].
Previously, it has been shown that penetrating cations can demonstrate the properties of “mild” mitochondrial-targeted uncouplers [24, 25]. These compounds contribute to the cyclic transmembrane transfer of fatty acids, so stimulating their protonophoric activity. The decrease in membrane potential reduces the accumulation of cations in mitochondria and thereby reduces their uncoupling activity. Thus, lipophilic cations acquire the properties of self-regulated uncouplers, causing limited depolarization of the mitochondrial membrane over a wide range of concentrations.
The goal of this work was to study the mechanism of action of mitochondrial-targeted uncouplers on inflammatory activation of endothelial cells. We show that the long-term treatment with lipophilic cation C12TPP (dodecyltriphenylphosphonium) as well as with classic uncouplers DNP (2,4-dinitrophenol) and TTFB (4,5,6,7-tetrachloro-2-trifluoromethylbenzimidazole) results in the suppression of NFκB-dependent expression of adhesion molecules and decreases neutrophil adhesion to the surface of TNF-stimulated endothelial cells.
MATERIALS AND METHODS
Cell cultures. Human endothelial EA.hy926 cells were grown on DMEM medium (Gibco, USA) containing 10% fetal calf serum (FCS) (HyClone, USA) and HAT (hypoxanthine/aminopterin/tyrosine) (Sigma, USA). Primary endothelial cells of human umbilical vein (HUVEC) kindly provided by M. A. Lagarkova (Institute of General Genetics, Russian Academy of Sciences) were grown on medium for primary endothelial cell cultures with all necessary additives (EGM-2 BulletKit; Lonza, USA) and used in the experiments in passages 2-4. The culture mats and plates used for HUVEC were pretreated with 2% aqueous solution of gelatin (Sigma). Human promyelocytic lymphoma HL-60 cells were grown in RPMI 1640 medium (Gibco) containing 10% FCS (HyClone). All cells were cultivated at 37°C in the presence of 5% CO2.
The penetrating cations (C12TPP and SkQ1; synthesized by G. A. Korshunova and N. V. Sumbatyan, Belozersky Institute of Physico-Chemical Biology, Lomonosov Moscow State University), uncouplers (DNP and TTFB), oligomycin, and deoxyglucose (Sigma) were added to the completely confluent monolayer of EA.hy926 cells 3 h before the measurement of mitochondrial membrane potential or 12 h before the measurement of ATP content. In all other experiments, endothelial cells were cultivated to a completely confluent monolayer (3 days) in the presence of penetrating cations, uncouplers, and antioxidants, followed by replacement of the culture medium by a new one containing 0.2% FCS, with the addition of TNF (kindly provided by L. N. Shingarova, Institute of Bioorganic Chemistry, Russian Academy of Sciences) after 12 h.
Assessment of mitochondrial membrane potential. Mitochondrial membrane potential was assessed with TMRM (tetramethylrhodamine methyl ester) fluorescent probe (Sigma), which electrophoretically accumulates in mitochondria. The cells were incubated with TMRM (100 nM, 15 min) and removed from the plates with trypsin-EDTA solution (Gibco); fluorescence was measured with a flow cytofluorimeter (Beckman Coulter Cytomics FC 500; Beckman Coulter, USA).
Measurement of ATP content in cells. The content of ATP in the cells was measured with a CLS II ATP Bioluminescence Assay Kit (Hoffmann-La Roche, Switzerland). The samples were prepared according to the manufacturer’s instructions. The measurements were made with a plate luminometer (Victor X5; PerkinElmer, USA).
DNA/RNA isolation and reverse transcription. DNA and RNA were isolated with DNeasy Blood & Tissue (Qiagen, USA) and RNeasy Mini Kit (Qiagen), respectively. The quality of isolated RNA was assessed by spectrophotometry, measuring the A260/A280 and A260/A230 absorption ratios. RNA was treated with DNase (Fermentas, Lithuania). cDNA was obtained as follows: 2 µg of denatured RNA, 0.1 µg of random hexamer primers, 0.2 µg of oligo(dT), and 0.5 mM of dNTP was incubated in 1× buffer (Invitrogen, USA) with 200 U of Superscript III revertase for 50 min at 43°C.
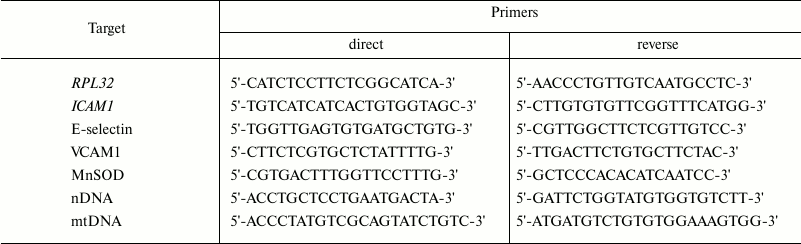
Real-time PCR was carried out in an iCycler iQ amplifier (Bio-Rad, USA). The expression of the target genes was determined using EVA Green mixture (Sintol, Russia) according to the manufacturer’s instructions. The primer sequences are given in the table.
Primers for determination of mRNA content by real-time PCR
Before the reaction, the mixture was heated at 95°C for 3 min. PCR was carried out according to the protocol (45 cycles): denaturing (95°C), 15 s; annealing (56°C), 30 s; elongation (72°C), 30 s. Fluorescence intensity was recorded at the stage of elongation. After the completion of PCR, reaction specificity was assessed by melting curve analysis in the range of 55-95°C. The experiments were repeated three times for each DNA or cDNA sample. The efficiency of amplification with the selected pairs of primers was 95-102%. The level of expression was calculated with allowance for amplification efficiency of the primers. The mRNA expression values of the target genes were normalized for the expression values of the RPL32 genes; the relative level of mtDNA/nDNA was determined analogously.
Assessment of neutrophil adhesion to endothelium. The EA.hy926 cells were incubated with 5 ng/ml of TNF for 8 h, twice washed with fresh RPMI 1640 medium, and incubated for 30 min at 37°C with the suspension of HL-60 cells labeled with BCECF-DA (Molecular Probes, USA). The unbound cells were washed off with RPMI 1640, the preparation was fixed with 2% paraformaldehyde, and images were obtained using an Axiovert 250 fluorescence microscope (Carl Zeiss, Germany).
Western blot. Immunoblotting was performed as described [18] using primary antibodies against p-IκBα, GAPDH, COXIV (Cell Signaling, USA), and tubulin (Sigma). Horseradish peroxidase-labeled antibodies against rabbit or mouse immunoglobulins (Sigma) were used as secondary antibodies. Visualization was performed with ECL (Amersham, USA) according to the manufacturer’s instructions.
Assessment of number of autophagosomes and mitochondrial reticulum structure. Autophagosomes were visualized in living cells with Cyto-ID® Autophagy Detection Kit (Enzo, USA) according to the manufacturer’s instructions. Simultaneously, mitochondria were detected with 200 nM MitoTracker Red (Molecular Probes) added to the cells. The images were obtained with the Axiovert 250 microscope. The number of autophagosomes and the length of mitochondria in the cells were determined automatically with MotionTracking 8.84.3 (http://motiontracking.mpi-cbg.de). Mitochondria were considered as fragmented if their length was less than 2 µm.
Statistical processing of results. The images were analyzed using ImageJ (http://imagej.nih.gov/). The results were statistically processed with Statistica 6.0 software. The data in all histograms and diagrams are presented as the mean values and standard error of the mean. Statistical significance was determined by Student’s t-criterion.
RESULTS AND DISCUSSION
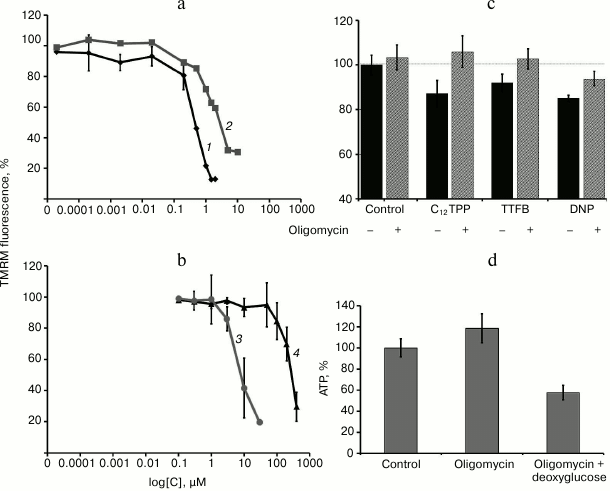
Effects of penetrating cations and classic uncouplers on mitochondrial membrane potential and ATP synthesis. Previously, it has been shown that penetrating cations of the SkQ family are mitochondria-targeted uncouplers of oxidative phosphorylation [24, 25]. Now we have confirmed that the lipophilic cations C12TPP and SkQ1 reduce mitochondrial membrane potential in EA.hy926 cells (Fig. 1a) just as classic uncouplers do (Fig. 1b). It is interesting that C12TPP in this cell model proved to be a more effective uncoupler than SkQ1, which contradicts the data obtained previously in isolated mitochondria [24]. Probably, the presence of reactive quinone residue in SkQ1 molecules results in its partial binding to cellular structures and reduces the efficiency of accumulation in mitochondria. We found that a slight (by 7-10%) decrease in membrane potential under the influence of low concentrations of cationic compounds and classic uncouplers was not accompanied by complete cessation of oxidative phosphorylation in EA.hy926 cells. This conclusion is based on the fact that the inhibitor of mitochondrial ATP-synthase, oligomycin (5 µg/ml), increased membrane potential in the presence of C12TPP (0.2 nM), DNP (5 µM), and TTFB (0.1 µM) (Fig. 1c).
Fig. 1. Effects of penetrating cations and classical uncouplers on mitochondrial membrane potential in EA.hy926 cells. Mitochondrial depolarization: a) under the influence of penetrating cations (C12TPP (1), SkQ1 (2)); b) under the influence of uncouplers (TTFB (3), DNP (4)). c) Oligomycin (5 µg/ml) prevents mitochondrial depolarization induced by low doses of C12TPP (0.2 nM), DNP (5 µM), and TTFB (0.1 µM); d) effects of oligomycin (5 µg/ml) and deoxyglucose (5 mM) on ATP content in the cells.
It should be noted that even the complete block of mitochondrial ATP-synthase by oligomycin had no substantial effect on the level of ATP in EA.hy926 cells, and only the suppression of glycolysis by the addition of 5 mM deoxyglucose resulted in its decrease (Fig. 1d). These results are in good agreement with the literature data showing that ATP in endothelium is produced mainly due to glycolysis [10]. Thus, both penetrating cations and classic uncouplers at the selected concentrations induce “mild” uncoupling by decreasing the mitochondrial membrane potential without complete cessation of oxidation phosphorylation and without a drop in the level of ATP.
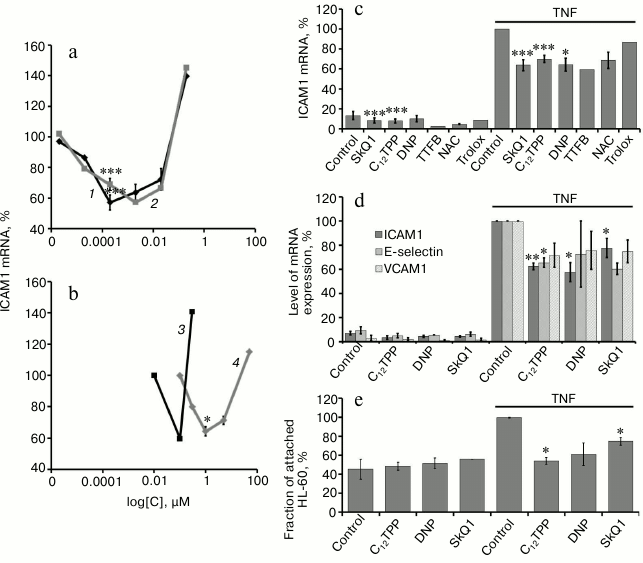
“Mild” uncoupling suppresses NFκB-dependent activation of TNF-stimulated endothelial cells. We have used the in vitro approaches with TNF activation of the cells to study the effect of “mild” uncoupling on the inflammatory response of endothelial cells (EA.hy926 and HUVEC) [21]. The assessment criteria of activation were enhanced expression of mRNA of adhesion molecules (ICAM1 and VCAM1) and E-selectin and increase in promyelocytic leukemia HL-60 cell adhesion to the endothelial cell monolayer. Previously, we have shown that TNF stimulates expression of mRNA and accumulation of adhesion molecules on the cell surface and the secretion of proinflammatory interleukins-6 and -8 in these cells [21]. The data in Fig. 2a show that the penetrating cations C12TPP and SkQ1 in EA.hy926 endothelial cells suppress the expression of ICAM1 over a broad range of concentrations (0.2-20 nM) with similar efficiency. DNP and TTFB at the concentrations causing mitochondrial depolarization by no more than 7-10% also suppressed the expression of ICAM1, but a slight increase in the concentration of the uncouplers eliminated or even reversed this effect (Fig. 2b). The range of effective concentrations for the classic uncouplers was considerably (by three orders of magnitude) narrower than for the penetrating cations C12TPP and SkQ1 (Fig. 2, a and b). The differences between the two types of uncouplers are probably explained by the fact that the decrease in membrane potential is accompanied by the reduction of accumulation of penetrating cations in mitochondria, which reduces their uncoupling activity and leads to stabilization of the potential. It is important to note that the penetrating cations and uncouplers suppress ICAM1 expression to the same extent as the classic antioxidants (Fig. 2c).
Fig. 2. Penetrating cations, classic uncouplers, and antioxidants suppress TNF-induced activation of endothelial cells. a-c) Expression of ICAM1 mRNA in EA.hy926 cells after TNF stimulation (0.25 ng/ml, 4 h): 1) SkQ1; 2) C12TPP; 3) TTFB; 4) DNP. d) Expression of ICAM1, VCAM1, and E-selectin mRNA in HUVEC cells. e) Adhesion of HL-60 cells to EA.hy926 cells activated by TNF (5 ng/ml, 12 h). Concentrations of added compounds (c-e): SkQ1 (0.2 nM), C12TPP (0.2 nM), DNP (5 µM), TTFB (1 µM), NAC (5 mM), and Trolox (200 µM). N ≥ 4, * p ≤ 0.05, ** p ≤ 0.01, *** p ≤ 0.001.
“Mild” uncoupling also inhibited other indicators of endothelial activation under the influence of TNF. In the HUVEC, the penetrating cations C12TPP (0.2 nM) and SkQ1 (0.2 nM) and classic uncouplers DNP (5 µM) and TTFB (0.1 µM), together with ICAM1, suppressed the expression of E-selectin and VCAM1 (Fig. 2d).
The adhesion molecules and selectins stop the rolling and promote lymphocyte attachment to vascular endothelium, which is necessary for lymphocyte transmigration into tissues [5]. We studied the adhesion of human promyelocytic leukemia HL-60 cells (these cells express the ligands for adhesion molecules and selectins on their surface) to the monolayer of EA.hy926 cells to assess the functional activity of adhesion molecules. Both penetrating cations and classic uncouplers suppressed the adhesion of HL-60 cells to the surface of endothelial cells (Fig. 2e).
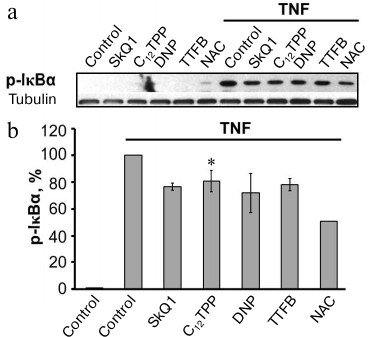
The major regulator of adhesion molecule expression during endothelial activation by TNF is the transcription factor NFκB [6]. Previously, we have shown that the inhibition of NFκB in EA.hy926 cells considerably suppressed the TNF-stimulated expression of ICAM1 [21]. The activation of NFκB under the influence of TNF includes phosphorylation followed by proteolysis of the inhibitory subunit IκBα, which retains NFκB in the cytoplasm [32]. All of the used uncouplers suppressed the phosphorylation of IκBα at the same concentrations that suppressed the expression of ICAM1 (Fig. 3).
Fig. 3. Penetrating cations, classic uncouplers, and antioxidant suppress TNF-stimulated phosphorylation of IκBα. EA.hy926 cells were stimulated by TNF (0.5 ng/ml, 15 min). Concentrations of the added compounds are as in Fig. 2, c-e. a) Results of typical Western blotting; b) results of densitometric analysis of Western blots; N = 5, * p ≤ 0.05.
Thus, it has been shown that long-term “mild” uncoupling of oxidative phosphorylation results in the suppression of NFκB-dependent activation of endothelial cells under the influence of TNF.
“Mild” uncoupling can induce mechanisms of endothelial cell resistance to the activating effect of TNF. The antiinflammatory effects of penetrating cations C12TPP and SkQ1 manifested themselves only after a long-term (3-day) treatment of the cells with these cations. We have observed such delayed effect of SkQ1 previously in many cell models [18, 33-35]. We revealed that the removal of both penetrating cations (C12TPP, SkQ1) and classic uncouplers (DNP and TTFB) from the medium 12 h before the addition of TNF had no effect on their ability to suppress the expression of ICAM1 and other inflammatory markers (see “Materials and Methods”). Using high concentrations of the uncouplers, we showed that the mitochondrial membrane potential was completely recovered upon removal of DNP (400 µM) from the medium, but the mitochondria remained depolarized for at least 12 h upon removal of C12TPP (1 µM). This indicates that classic uncouplers are able to induce mechanisms inhibiting the proinflammatory effect of TNF in the absence of uncoupling. At the same time, the action of penetrating cations can be additionally ensured by their direct uncoupling effect.
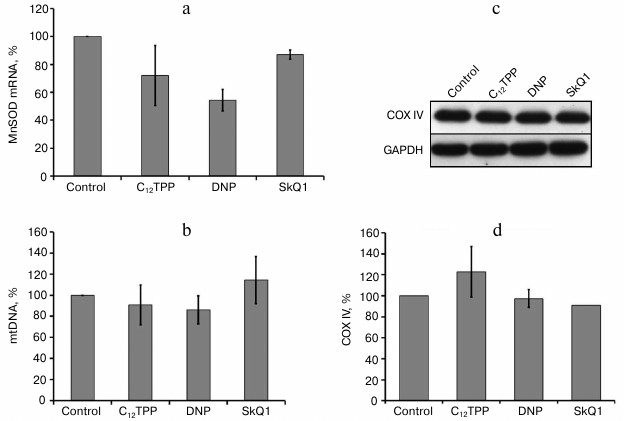
One of the major inducible mitochondrial antioxidant enzymes is manganese superoxide dismutase (MnSOD). We checked the possibility of its induction but found no stimulation of its expression by uncouplers (Fig. 4a).
Fig. 4. Penetrating cations and uncouplers have no effect on the content of mtDNA and mitochondrial protein COXIV, as well as on the expression of MnSOD in endothelial cells. a) Expression of mRNA of MnSOD in HUVEC; b) content of mtDNA in EA.hy926 cells. The content of COXIV in EA.hy926 cells; c) typical Western blot; d) densitometric analysis of Western blots, N = 3. Concentrations of added compounds are as in Fig. 2, c-e.
Potential mechanisms for induction of antiinflammatory effects of “mild” uncoupling. Weak mitochondrial stresses such as partial membrane depolarization, inhibition of ATP synthesis, intensification of ROS generation, and excessive accumulation of Ca2+ can induce the mechanisms of intensification of bioenergetic functions, quality control, and antioxidant protection of mitochondria, which leads to the intensification of cell adaptation to unfavorable conditions [31]. For example, the long-term administration of low DNP doses in mice extended their mean lifespan, suppression of oxidative damage to membranes, proteins, and DNA, and stimulation of mitochondrial biogenesis in muscles and adipose tissue [36, 37].
We estimated the effect of long-term “mild” uncoupling on the quantity and structure of mitochondria, as well as on general autophagy and mitochondrial-specific autophagy (mitophagy). In EA.hy926 cells, the uncouplers did not induce any substantial changes in the content of mtDNA (Fig. 4b) and the expression of an inner membrane marker protein – cytochrome c oxidase subunit IV (COXIV) (Fig. 4, c and d). It may be supposed that the partial uncoupling under these conditions does not trigger the mechanisms of “retrograde signaling”, which are activated, e.g. during starvation, and stimulate mitochondrial biogenesis, because the level of ATP in these cells was actually independent of oxidative phosphorylation (Fig. 1d).
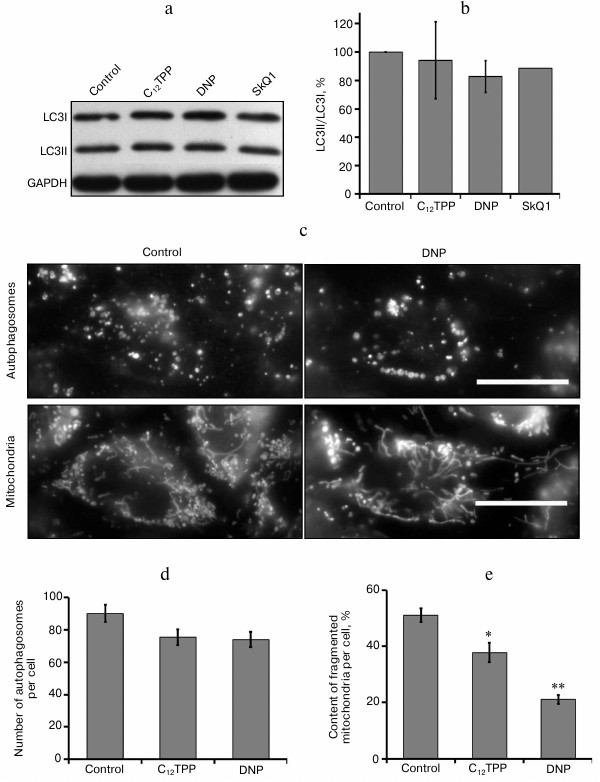
The “mild” uncoupling can contribute to improvement of mitochondrial population quality by stimulating the mechanisms for quality control and elimination of damaged organelles. At high concentrations, the uncouplers cause intensive accumulation of autophagosomes [38]. At the same time, the long-term incubation with low concentrations of the uncouplers inducing antiinflammatory effect did not intensify lipidization of the LC3 protein, which is necessary for autophagy induction (Fig. 5, a and b), and had no effect on the content of autophagosomes in these cells (Fig. 5, c and d). The findings demonstrate the absence of stimulation of bulk autophagy during “mild” uncoupling but do not allow the estimation of mitophagy. We analyzed this process by simultaneous staining of mitochondria and autophagosomes, but we did not observe substantial colocalization of autophagosomes and mitochondria either in the initial cells or after the incubation with the uncouplers (data not shown).
Fig. 5. Effects of C12TPP and DNP on number of autophagosomes and mitochondrial structure in EA.hy926 cells. The content of two LC3 protein forms, initial (I) and lipidized (II): a) typical Western blot; b) results of densitometric analysis of Western blots, N = 3; c) fluorescence microscopy, scale bar 15 µm; d) number of autophagosomes per cell, N = 11-15; e) relative content of completely fragmented mitochondria per cell, N = 7-10, * p ≤ 0.01, ** p ≤ 0.001.
The long-term “mild” uncoupling, not increasing the content of mitochondria in endothelial cells, caused structural changes of the mitochondrial reticulum (Fig. 5, c and e). Previously, we have shown that the mitochondria in EA.hy926 cells, which form a completely confluent monolayer, are extremely heterogeneous. Among them, there was a marked population of fragmented mitochondria with low potential [18]. Analogous mitochondrial heterogeneity was also observed in fibroblasts [34]. As a result of long-term (4-day) treatment of endothelial cells with low concentrations of C12TPP (0.2 nM) or DNF (5 µM), the quantity of completely fragmented mitochondria in the cells decreased (Fig. 5, c and e). These changes could be a consequence of simultaneous activation of mitochondrial biogenesis and selective mitophagy without changing the total number of mitochondria. On the other hand, the influence of long-term “mild” uncoupling on mitochondrial structure could be a result of antioxidant effect. Previously, we have shown that mitochondria are sensitive sensors of oxidative stress in cells. Prooxidants induced the fragmentation of elongated mitochondria, while antioxidants (both classic and mitochondria-targeted) prevented this effect [39]. It can be supposed that the decrease in the fraction of fragmented mitochondria with a low potential contributes to decrease in mitochondrial production of ROS. The main indication of potential association between the mechanism of proinflammatory effects of the uncouplers and the decrease in content of ROS is the fact that the classic antioxidants NAC (1-5 mM) and Trolox (100-200 µM) noticeably suppress the TNF-induced activation of NFκB (Fig. 3) and the expression of ICAM1 in EA.hy926 cells (Fig. 2a).
The findings indicate that the long-term “mild” uncoupling of oxidative phosphorylation induces resistance of endothelial cells to the proinflammatory action of TNF activating the NFκB-dependent signaling pathway. Probably, one of the causes of these antiinflammatory effects is reduced ROS generation in mitochondria. It is known that the enhanced level of TNF in blood is a concomitant of many pathological states, as well as aging [40]. The main target of the damaging effect of TNF is endothelium, the dysfunctions of which result in the development of various cardiovascular pathologies [41]. Previously, models of renal and cerebral ischemia demonstrated high therapeutic efficiency of cationic uncouplers [42]. We assume that the basic mechanism determining the therapeutic effect of uncouplers is their antiinflammatory effect on vascular endothelium.
We have discovered for the first time that even a minor decrease in mitochondrial membrane potential can alter the physiological state of endothelium and protect it from destructive effects of proinflammatory cytokines. These results open prospects for using “mild” mitochondria-targeted uncouplers as effective angioprotectors in therapy for different pathologies associated with excessive inflammation.
The authors are deeply grateful to V. P. Skulachev for his constant interest in our work and for his assistance. We heartily congratulate Vladimir Petrovich on his birthday and wish him many years of fruitful work for the sake of science.
The authors are grateful to L. N. Shingarova (Institute of Bioorganic Chemistry, Russian Academy of Sciences), M. A. Lagarkova (Institute of General Genetics, Russian Academy of Sciences), S. A. Evfratov (Faculty of Chemistry, Lomonosov Moscow State University), and Ya. L. Kalaidzidis (Faculty of Bioengineering and Bioinformatics, Lomonosov Moscow State University) for providing materials and software and for assistance in the work.
This work was supported by the Russian Science Foundation (project No. 14-14-00055, experiments with mitochondrial uncouplers) and the Russian Foundation for Basic Research (project No. 13-04-40309-N, experiments with antioxidants). The relative amount of ATP was measured with a VICTOR™ X5 Multilabel Plate Reader (PerkinElmer, USA) purchased in the framework of priority trends for development of Lomonosov Moscow State University (PNR X5-13).
REFERENCES
1.Bruunsgaard, H., Skinhoj, P., Pedersen, A. N.,
Schroll, M., and Pedersen, B. K. (2000) Ageing, tumor necrosis
factor-α (TNF-α) and atherosclerosis, Clin. Exp.
Immunol., 121, 255-260.
2.Chung, H. Y., Sung, B., Jung, K. J., Zou, Y., and
Yu, B. P. (2006) The molecular inflammatory process in aging,
Antioxid. Redox Signal., 8, 572-581.
3.Csiszar, A., Ungvari, Z., Koller, A., Edwards, J.
G., and Kaley, G. (2003) Aging-induced proinflammatory shift in
cytokine expression profile in coronary arteries, FASEB J.,
17, 1183-1185.
4.Dandona, P., Aljada, A., and Bandyopadhyay, A.
(2004) Inflammation: the link between insulin resistance, obesity and
diabetes, Trends Immunol., 25, 4-7.
5.Springer, T. A. (1994) Traffic signals for
lymphocyte recirculation and leukocyte emigration: the multistep
paradigm, Cell, 76, 301-314.
6.Roebuck, K. A., and Finnegan, A. (1999) Regulation
of intercellular adhesion molecule-1 (CD54) gene expression, J.
Leukoc. Biol., 66, 876-888.
7.Park, J., Choi, H., Min, J. S., Park, S. J., Kim,
J. H., Park, H. J., Kim, B., Chae, J. I., Yim, M., and Lee, D. S.
(2013) Mitochondrial dynamics modulate the expression of
pro-inflammatory mediators in microglial cells, J. Neurochem.,
127, 221-232.
8.West, A. P., Shadel, G. S., and Ghosh, S. (2011)
Mitochondria in innate immune responses, Nature Rev. Immunol.,
11, 389-402.
9.Davidson, S. M., and Duchen, M. R. (2007)
Endothelial mitochondria: contributing to vascular function and
disease, Circ. Res., 100, 1128-1141.
10.Culic, O., Gruwel, M. L., and Schrader, J. (1997)
Energy turnover of vascular endothelial cells, Am. J. Physiol.,
273, 205-213.
11.Addabbo, F., Ratliff, B., Park, H. C., Kuo, M.
C., Ungvari, Z., Csiszar, A., Krasnikov, B., Sodhi, K., Zhang, F.,
Nasjletti, A., and Goligorsky, M. S. (2009) The Krebs cycle and
mitochondrial mass are early victims of endothelial dysfunction:
proteomic approach, Am. J. Pathol., 174, 34-43.
12.Madamanchi, N. R., and Runge, M. S. (2007)
Mitochondrial dysfunction in atherosclerosis, Circ. Res.,
100, 460-473.
13.Schulz, E., Dopheide, J., Schuhmacher, S.,
Thomas, S. R., Chen, K., Daiber, A., Wenzel, P., Munzel, T., and
Keaney, J. F., Jr. (2008) Suppression of the JNK pathway by induction
of a metabolic stress response prevents vascular injury and
dysfunction, Circulation, 118, 1347-1357.
14.Wrzosek, A., Lukasiak, A., Gwozdz, P., Malinska,
D., Kozlovski, V. I., Szewczyk, A., Chlopicki, S., and Dolowy, K.
(2009) Large-conductance K+ channel opener CGS7184 as a
regulator of endothelial cell function, Eur. J. Pharmacol.,
602, 105-111.
15.Poburko, D., Lee, C. H., and van Breemen, C.
(2004) Vascular smooth muscle mitochondria at the cross roads of
Ca2+ regulation, Cell Calcium, 35,
509-521.
16.Joo, H. K., Lee, Y. R., Lim, S. Y., Lee, E. J.,
Choi, S., Cho, E. J., Park, M. S., Ryoo, S., Park, J. B., and Jeon, B.
H. (2012) Peripheral benzodiazepine receptor regulates vascular
endothelial activations via suppression of the voltage-dependent anion
channel-1, FEBS Lett., 586, 1349-1355.
17.Feletou, M., and Vanhoutte, P. M. (2006)
Endothelial dysfunction: a multifaceted disorder (The Wiggers Award
Lecture), Am. J. Physiol. Heart Circ. Physiol., 291,
985-1002.
18.Galkin, I. I., Pletjushkina, O. Yu., Zinovkin, R.
A., Zakharova, V. V., Biryukov, I. S., Chernyak, B. V., and Popova, E.
N. (2014) Mitochondria-targeted antioxidants prevent the tumor necrosis
factor induced apoptosis of endothelial cells, Biochemistry
(Moscow), 79, 124-130.
19.Rahman, A., Kefer, J., Bando, M., Niles, W. D.,
and Malik, A. B. (1998) E-selectin expression in human endothelial
cells by TNF-α-induced oxidant generation and NF-κB
activation, Am. J. Physiol., 275, L533-544.
20.Deshpande, S. S., Angkeow, P., Huang, J., Ozaki,
M., and Irani, K. (2000) Rac1 inhibits TNF-α-induced endothelial
cell apoptosis: dual regulation by reactive oxygen species, FASEB
J., 14, 1705-1714.
21.Zinovkin, R. A., Romaschenko, V. P., Galkin, I.
I., Zakharova, V. V., Pletjushkina, O. Y., Chernyak, B. V., and Popova,
E. N. (2014) Role of mitochondrial reactive oxygen species in
age-related inflammatory activation of endothelium, Aging
(Albany N. Y.), 6, 671-674.
22.Brigelius-Flohe, R., and Flohe, L. (2011) Basic
principles and emerging concepts in the redox control of transcription
factors, Antioxid. Redox Signal., 15, 2335-2381.
23.Korshunov, S. S., Skulachev, V. P., and Starkov,
A. A. (1997) High protonic potential actuates a mechanism of production
of reactive oxygen species in mitochondria, FEBS Lett.,
416, 15-18.
24.Severin, F. F., Severina, I. I., Antonenko, Y.
N., Rokitskaya, T. I., Cherepanov, D. A., Mokhova, E. N., Vyssokikh, M.
Y., Pustovidko, A. V., Markova, O. V., Yaguzhinsky, L. S., Korshunova,
G. A., Sumbatyan, N. V., Skulachev, M. V., and Skulachev, V. P. (2010)
Penetrating cation/fatty acid anion pair as a mitochondria-targeted
protonophore, Proc. Natl. Acad. Sci. USA, 107,
663-668.
25.Skulachev, V. P., Antonenko, Y. N., Cherepanov,
D. A., Chernyak, B. V., Izyumov, D. S., Khailova, L. S., Klishin, S.
S., Korshunova, G. A., Lyamzaev, K. G., Pletjushkina, O. Y., Roginsky,
V. A., Rokitskaya, T. I., Severin, F. F., Severina, I. I., Simonyan, R.
A., Skulachev, M. V., Sumbatyan, N. V., Sukhanova, E. I., Tashlitsky,
V. N., Trendeleva, T. A., Vyssokikh, M. Y., and Zvyagilskaya, R. A.
(2010) Prevention of cardiolipin oxidation and fatty acid cycling as
two antioxidant mechanisms of cationic derivatives of plastoquinone
(SkQs), Biochim. Biophys. Acta, 1797, 878-889.
26.Duval, C., Negre-Salvayre, A., Dogilo, A.,
Salvayre, R., Penicaud, L., and Casteilla, L. (2002) Increased reactive
oxygen species production with antisense oligonucleotides directed
against uncoupling protein 2 in murine endothelial cells, Biochem.
Cell Biol., 80, 757-764.
27.Fink, B. D., Reszka, K. J., Herlein, J. A.,
Mathahs, M. M., and Sivitz, W. I. (2005) Respiratory uncoupling by UCP1
and UCP2 and superoxide generation in endothelial cell mitochondria,
Am. J. Physiol. Endocrinol. Metab., 288, 71-79.
28.Lee, K. U., Lee, I. K., Han, J., Song, D. K.,
Kim, Y. M., Song, H. S., Kim, H. S., Lee, W. J., Koh, E. H., Song, K.
H., Han, S. M., Kim, M. S., Park, I. S., and Park, J. Y. (2005) Effects
of recombinant adenovirus-mediated uncoupling protein 2 overexpression
on endothelial function and apoptosis, Circ. Res., 96,
1200-1207.
29.Park, J. Y., Park, K. G., Kim, H. J., Kang, H.
G., Ahn, J. D., Kim, H. S., Kim, Y. M., Son, S. M., Kim, I. J., Kim, Y.
K., Kim, C. D., Lee, K. U., and Lee, I. K. (2005) The effects of the
overexpression of recombinant uncoupling protein 2 on proliferation,
migration and plasminogen activator inhibitor 1 expression in human
vascular smooth muscle cells, Diabetologia, 48,
1022-1028.
30.Ungvari, Z., Orosz, Z., Labinskyy, N., Rivera,
A., Xiangmin, Z., Smith, K., and Csiszar, A. (2007) Increased
mitochondrial H2O2 production promotes
endothelial NF-κB activation in aged rat arteries, Am. J.
Physiol. Heart Circ. Physiol., 293, 37-47.
31.Barbour, J. A., and Turner, N. (2014)
Mitochondrial stress signaling promotes cellular adaptations, Int.
J. Cell Biol., 2014, 156020.
32.Tilstra, J. S., Clauson, C. L., Niedernhofer, L.
J., and Robbins, P. D. (2011) NF-κB in aging and disease,
Aging Dis., 2, 449-465.
33.Popova, E. N., Pletjushkina, O. Y., Dugina, V.
B., Domnina, L. V., Ivanova, O. Y., Izyumov, D. S., Skulachev, V. P.,
and Chernyak, B. V. (2010) Scavenging of reactive oxygen species in
mitochondria induces myofibroblast differentiation, Antioxid. Redox
Signal., 13, 1297-1307.
34.Izyumov, D. S., Domnina, L. V., Nepryakhina, O.
K., Avetisyan, A. V., Golyshev, S. A., Ivanova, O. Yu., Korotetskaya,
M. V., Lyamzayev, K. G., Pletjushkina, O. Yu., Popova, E. N., and
Chernyak, B. V. (2010) Mitochondria as sources of reactive oxygen
species during oxidative stress. The study with novel
mitochondria-targeted antioxidants on the basis of “Skulachev
ions”, Biochemistry (Moscow), 75, 123-129.
35.Agapova, L. S., Chernyak, B. V., Domnina, L. V.,
Dugina, V. B., Efimenko, A. Yu., Fetisova, E. K., Ivanova, O. Yu.,
Kalinina, N. I., Lichinitser, M. R., Lukashev, A. N., Khromova, N. V.,
Kopnin, B. P., Korotetskaya, M. V., Pletjushkina, O. Yu., Popova, E.
N., Shagieva, G. S., Skulachev, M. V., Stepanova, E. V., Titova, E. V.,
Tkachuk, V. A., Vasilyev, Yu. M., and Skulachev, V. P. (2008)
Mitochondria-targeted plastoquinone derivative as a tool interrupting
the aging program. 3. SkQ1 suppresses tumor development from
p53-deficient cells, Biochemistry (Moscow), 73,
1300-1316.
36.Caldeira da Silva, C. C., Cerqueira, F. M.,
Barbosa, L. F., Medeiros, M. H., and Kowaltowski, A. J. (2008) Mild
mitochondrial uncoupling in mice affects energy metabolism, redox
balance and longevity, Aging Cell, 7, 552-560.
37.Cerqueira, F. M., Laurindo, F. R., and
Kowaltowski, A. J. (2011) Mild mitochondrial uncoupling and calorie
restriction increase fasting eNOS, akt and mitochondrial biogenesis,
PLoS One, 6, e18433.
38.Lyamzaev, K. G., Izyumov, D. S., Avetisyan, A.
V., Yang, F., Pletjushkina, O. Y., and Chernyak, B. V. (2004)
Inhibition of mitochondrial bioenergetics: the effects on structure of
mitochondria in the cell and on apoptosis, Acta Biochim. Pol.,
51, 553-562.
39.Pletjushkina, O. Y., Lyamzaev, K. G., Popova, E.
N., Nepryakhina, O. K., Ivanova, O. Y., Domnina, L. V., Chernyak, B.
V., and Skulachev, V. P. (2006) Effect of oxidative stress on dynamics
of mitochondrial reticulum, Biochim. Biophys. Acta, 1757,
518-524.
40.Bradley, J. R. (2008) TNF-mediated inflammatory
disease, J. Pathol., 214, 149-160.
41.Hirata, Y., Nagata, D., Suzuki, E., Nishimatsu,
H., Suzuki, J.-I., and Nagai, R. (2010) Diagnosis and treatment of
endothelial dysfunction in cardiovascular disease, Int. Heart
J., 51, 1-6.
42.Plotnikov, E. Yu., Silachev, D. N., Jankauskas,
S. S., Rokitskaya, T. I., Chuprykina, A. A., Pevzner, I. B., Zorova, L.
D., Isaev, N. K., Antonenko, Yu. N., Skulachev, V. P., and Zorov, D. B.
(2012) Partial uncoupling of respiration and phosphorylation as one of
the pathways of implementation of the nephro- and neuroprotective
effects of penetrating cations of the SkQ family, Biochemistry
(Moscow), 77, 1029-1037.