REVIEW: Spectral Dependence of Chlorophyll Biosynthesis Pathways in Plant Leaves
O. B. Belyaeva* and F. F. Litvin
Lomonosov Moscow State University, Faculty of Biology, 119991 Moscow, Russia; fax: +7 (495) 939-4309; E-mail: belyaeva0104@gmail.com* To whom correspondence should be addressed.
Received August 23, 2015
This review covers studies on the dependence of chlorophyll photobiosynthesis reactions from protochlorophyllide on the spectral composition of actinic light. A general scheme of the reaction sequence for the photochemical stage in chlorophyll biosynthesis for etiolated plant leaves is presented. Comparative analysis of the data shows that the use of light with varied wavelengths for etiolated plant illumination reveals parallel transformation pathways of different protochlorophyllide forms into chlorophyllide, including a pathway for early photosystem II reaction center P-680 pigment formation.
KEY WORDS: chlorophyll, protochlorophyllide, photoreduction, fluorescence spectra, absorption spectraDOI: 10.1134/S0006297915130076
Abbreviations: Chl, chlorophyll; Chld, chlorophyllide; Pchl, protochlorophyll; Pchld, protochlorophyllide; POR, protochlorophyllide oxidoreductase; PS I(II), photosystem I(II); RC, reaction center.
Intensive chlorophyll synthesis occurs in green plant leaves. The final
light-dependent stage of this process is a combination of reactions of
various nature: photophysical, photochemical, and biochemical. As a
result of this multistep and branching process, not only a chlorophyll
molecule is synthesized, but also several types of specific
pigment–protein structures are formed that contribute to the
functioning of two photosystems (PS I and PS II) and light-harvesting
complexes. High chlorophyll concentration in green leaves creates
technical difficulties for studying the mechanism of its photochemical
synthesis from its immediate precursor protochlorophyllide, since the
content of protochlorophyllide and its intermediates is two to three
orders of magnitude lower than that of chlorophyll. This justifies
using plant materials with low pigment content for studying chlorophyll
biosynthesis, such as etiolated (grown in the dark) plants or those in
the early stages of greening. Methods derived from these experiments
and modern methodological approaches allow us to analyze chlorophyll
biosynthesis directly in green plant leaves.
It should be emphasized that the results of studying etiolated plant greening are not only fundamental, but also applied, since plant germination and de-etiolation largely define the efficiency and success of agriculture.
Spectral methods are essential for studying the final stage of chlorophyll biosynthesis. Photosynthetic pigments have characteristic spectra in the visible light region in solutions and in pigment–protein complexes. Their analysis provides multifaceted information not only on structural changes in molecules, but also on intermolecular pigment–pigment and pigment–protein interactions. Several spectrally different forms of protochlorophyllide are present in etiolated and green plant leaves. The main forms are Pchld 633/628, Pchld 643/637, and the usually dominant Pchld 655/650 (the numbers indicate the positions of fluorescence and absorption maxima) [1-5]. Several minor long-wavelength forms are also known [5-10]. The bulk of chlorophyll is synthesized by conversion of the dominant precursor form Chld 655/650.
GENERAL SCHEME OF BIOSYNTHESIS OF DIFFERENT CHLOROPHYLL FORMS
DURING GREENING OF ETIOLATED LEAVES
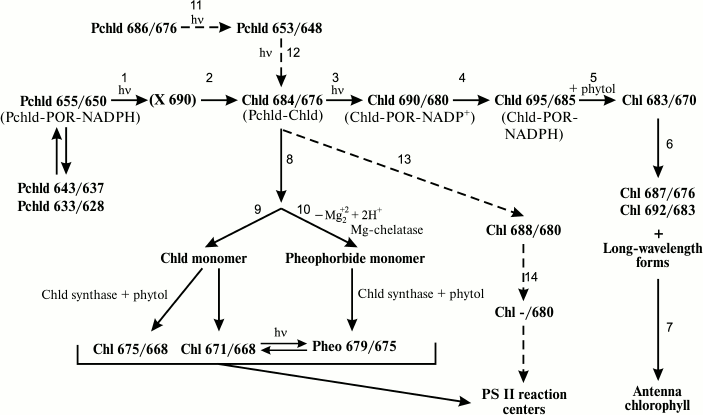
The results of long-term studies of chlorophyll biosynthesis in etiolated leaves have shown that the light-dependent stage of biosynthesis of different chlorophyll forms in a leaf is a complicated, branching chain of photochemical and dark reactions involving several chlorophyll precursor forms. The history of the gradual development and complication of the general reaction scheme for the light-dependent chlorophyll biosynthesis stage is described in [11, 12]. We suggested a general scheme of biosynthetic reactions for the main chlorophyll forms present in photosystem reaction centers (RC) and light-harvesting complex [12, 13] (figure) that includes primary photophysical reactions of protochlorophyllide photoreduction (reactions (1) and (2) on the scheme, see review [14]) as well as branching and parallel chlorophyll–protein complex formation pathways for light-harvesting antennae (reactions (3)-(7) on the scheme), and PS II reaction center chlorophylls and pheophytins (reactions (8)-(14) on the scheme, see review [13]). The biogenetic pathways of the PS I reaction center are less explored. However, in the study of chlorophyll formation in Chlorella vulgaris B-15 mutant cells with a complete genetic block of chlorophyll synthesis in the dark [15], along with photoreactions of Pchld 655/650 and Pchld 640/635 protochlorophyllide forms known for green algae, photoconversion of a long-wavelength esterified form of chlorophyll precursor Pchl 682/672 into a stable Chl 715/696 form was found. Chl 715/696 was proposed to be a PS I RC pigment.
General scheme of the reaction sequence at the photochemical stage of chlorophyll biosynthesis in greening plant leaves. Pchld, protochlorophyllide; Chld, chlorophyllide; Chl, chlorophyll. The numbers indicate the positions of fluorescence (first index) and absorption (second index) maxima for pigment forms. Broken arrows show the pathway of P-680 pigment generation from the long-wavelength protochlorophyllide form
Schoefs and Frank [16] proposed a cyclical scheme of chlorophyll biosynthesis and photoactive protochlorophyllide complex regeneration, two cycles of which essentially correspond to the direct and branched reaction sequences in our scheme.
PRIMARY REACTIONS OF PROTOCHLOROPHYLLIDE PHOTOREDUCTION:
NONFLUORESCENT INTERMEDIATES AND PRIMARY FLUORESCENT CHLOROPHYLL
FORMS
The key reaction for the final light-dependent chlorophyll biosynthesis stage in plant leaves is the photoreduction of its direct precursor protochlorophyllide (reactions (1) and (2) on the general scheme). This reaction is a joining of two hydrogen atoms to positions C17 and C18 of a double bond in a protochlorophyllide molecule. High photoreaction efficiency in etiolated leaves is due to the fact that it is carried out inside a photoactive complex composed of protochlorophyllide, the hydrogen donor NADPH, and the protochlorophyllide oxidoreductase (POR) photoenzyme (see review [17]). Facilitated by the active triple complex structure, spatial relationships of protochlorophyllide and hydrogen donors are created that are favorable for the photoreduction reaction.
Nonfluorescent intermediates. With low-temperature fluorescence spectroscopy and time-resolved absorption spectroscopy, protochlorophyllide photoreduction was found to be a complicated process involving several fast light and dark reactions, forming two or three short-lived intermediates characterized by intense protochlorophyllide fluorescence quenching [12, 18-44].
We proposed the following scheme for the primary reactions [37]:
Here, R and X690 are nonfluorescent intermediates, one of which (X690) might be composed of two components formed simultaneously and at different rates: R697 and R688 (the numbers correspond to absorption maxima).
Short-lived R and X690 intermediates are characterized by a singlet ESR signal with the g-factor of a free electron [35, 36, 38]. The results of studies indicate formation of complexes with charge transfer occurring during the primary photophysical chlorophyll biosynthesis stage [12, 43, 45]. Spectral characteristics of two nonfluorescent intermediates (R and X690) in vivo suggest that their light-induced formation corresponds to formation of complexes with partial and full charge transfer [12, 45, 46]:
The results of study of primary protochlorophyllide photoreduction reactions for reconstructed triple complexes: Pchld–POR–NADPH at 180 K, obtained by absorption spectroscopy, ESR spectroscopy, ENDOR spectroscopy, and Stark spectroscopy [43], suggested that the nonfluorescent intermediate formation involves hydride ion transfer to form a charge-transfer complex. Photon absorption by a protochlorophyllide molecule was proposed to result in temporal charge separation along the double C17=C18 bond, facilitating ultrafast hydride ion transfer from NADPH to position C17 of the protochlorophyllide molecule [43, 47]. The resulting complex with charge transfer facilitates proton transfer from the tyrosine phenol group of POR to the C18 atom of the protochlorophyllide molecule in the subsequent dark reaction.
Primary fluorescent chlorophyll forms. The nonfluorescent X690 intermediate formed under illumination at low temperatures is converted to chlorophyll by a dark pathway after elevating the temperature of a studied sample. When using integral white light for illumination of etiolated leaves at 77 K, a virtually simultaneous formation of two primary chlorophyllide forms is observed after raising the temperature. Their fluorescence maxima are at 695 and 684 nm, with corresponding absorption bands at 684 and 676 nm [27, 28, 48]. The band ratio depends on temperature and the spectral composition of the actinic light. Afterwards, the Chld 695/684 form is converted into a shorter wavelength Chld 684/676 form in a dark reaction. The formation of only one primary chlorophyllide form with a shorter wavelength was observed in etioplast samples.
The results of studies of primary reactions of protochlorophyllide photoreduction in plant leaves using time-resolved fluorescence spectroscopy at room temperature [49] showed dark transformation of nonfluorescent intermediates resulting in formation of four primary chlorophyllide forms characterized by fluorescence maxima at 684, 690, 695-697, and 706 nm. Similar results were obtained by analyzing primary fluorescent chlorophyllide forms with low-temperature fluorescence spectroscopy and Gaussian component analysis of differential spectra [48]. Long-wavelength primary chlorophyllide forms with fluorescence maxima at 696 and 706 nm were found to transform into shorter wavelength forms with fluorescence maxima at 675 and 684 nm, respectively, at room temperature. The form with fluorescence maximum at 684 nm is likely to be the Chld 684/676 form, which is a precursor in the biosynthesis pathway for PS II reaction center and light-harvesting complex pigments (reaction (2) in the figure). The formation of several primary labile chlorophyllide forms apparently indicates an early branching of pathways for generating functionally different native pigment forms.
DEPENDENCE OF CHLOROPHYLL GENERATION PATHWAYS FOR DIFFERENT
CHLOROPHYLL FORMS IN ETIOLATED LEAVES ON SPECTRAL COMPOSITION OF
EXCITING LIGHT
Generation sequence of primary chlorophyllide forms depending on the spectral composition of the actinic light. As mentioned earlier, the ratio of bands at 695 and 684 nm, which appear under illumination at low temperature, depends on the spectral composition of the actinic light. Specific experiments [11, 45] showed that blue light (with a maximum at 470 nm) leads to formation of only the 695 nm band in the fluorescence spectrum after raising the temperature, which gradually shifts to 684 nm. After red light (>600 nm) illumination of leaves, only one shorter wavelength maximum at 684 nm appeared in the spectrum after raising the temperature of the sample. These data suggest the presence of two types of active protochlorophyllide–protein complexes with virtually identical spectral characteristics in the red region in etiolated leaves. Both forms are transformed into chlorophyllide through a stage of nonfluorescent intermediate formation. One of the precursors can transform directly into a shorter wavelength Chld 684/676 chlorophyllide form. A hypothetical reaction scheme can be represented as follows:
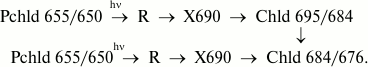
The existence in vivo of an additional protochlorophyllide form, Pchld 653/648, which is similar to the main active form in spectral characteristics was revealed later [9, 50]. Moreover, the second pathway may involve transformation of a shorter wavelength Pchld 643/637 precursor form whose fluorescence is quenched due to highly effective energy migration towards the main active form. Using derivative spectroscopy and Gaussian component analysis of light-minus-dark differential fluorescence spectra [9], the fluorescence band of the photoactive Pchld 655/650 form was found to include two components with maxima at 653 and 657 nm. The Pchld 653/648 form is accumulated the most in very young (2-3-day-old) etiolated leaves that have not yet formed prolamellar bodies [16, 51]. It is found in green plant leaves grown under normal photoperiodic conditions after a dark period [9], and in greening leaves after several hours of illumination, when prolamellar bodies are destroyed and thylakoids develop [52]. Ignatov and Litvin [50] found the Pchld 653/648 form to be generated under the action of red light from the minor long-wavelength form Pchld 686/676.
PS II RC chlorophyll formation in plant leaves at early etiolation stage. Role of long-wavelength protochlorophyllide form. Elucidation of the PS II RC P-680 chlorophyll biosynthesis pathway was made possible using light of various wavelengths for illumination of etiolated leaves in studying the biosynthesis of the native chlorophyll form in young leaves of etiolated plants (3-4-days after germination) [50]. A brief illumination of young etiolated plants by white light resulted (as well as in the case of 7-10-day-old etiolated plants) in Chld 684/676 formation, which is a product of the first photoreaction. However, at room temperature this intermediate participated in not only one (as in 7-10 day leaves), but in two dark reactions: Chld 684/676 → Chl 675/670 and Chld 684/676 → Chl 688/680 → Chl -/680 (reactions (8) and (13) on the scheme). Analysis of this process for young plants revealed that during the first 3-5 s of white light illumination, Chld 684/676 is accumulated without any noticeable changes in absorption and fluorescence bands of Pchld 655/650, even though the photoconversion of protochlorophyllide did occur (as shown by extracts). Experiments using monochromatic long-wavelength (680 nm) light at 4°C showed this paradoxical lag phase to be insubstantial. This is explained by photoconversion of the lowly fluorescent long-wavelength Pchld 686/676 form into Pchld 653/648, which is transformed into Chld 684/676 in the next light reaction (reactions (11) and (12) on the scheme).
Therefore, for the early stages of plant development, another pathway of protochlorophyllide transformations was detected in addition to the main branched reaction chain leading to the synthesis of nonfluorescent chlorophyll Chl -/680 (perhaps a PS II RC pigment) from the long-wavelength protochlorophyllide Pchld 686/676 through the formation of Pchld 653/648, whose spectral properties are very close (but not identical) to those of the basic form of protochlorophyllide Pchld 655/650.
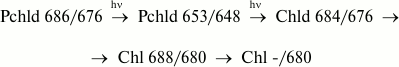
Prioritized formation of the precursor complex for RC during seed germination is apparently not accidental, and its early biogenesis causes top priority reaction center formation, as contrasted with other PS II components.
In juvenile leaves, photoconversion of the long-wavelength Pchld 686/676(440) form into the shorter wavelength form occurs, which may be connected to photo-disaggregation of large protochlorophyllide oligomers into smaller ones for creating the necessary steric environment for photoreduction of protochlorophyllide into chlorophyllide. The POR dimer disaggregation is supposedly unlikely in this case, since the Pchld 686/676(440) → Pchld 653/648(440) photoreaction is observed at 77 K as well. The disaggregation hypothesis is also supported by the fact that this photoreaction is blocked in the presence of D2O, which is an inhibitor of pigment disaggregation in Chld/Pchld-containing greening leaf complexes [50]. Aging of etiolated plants causes the accumulation of the main active protochlorophyllide form Pchld 655/650(448), whose photoconversion results mainly in antenna chlorophyll formation.
As noted earlier, the pathway of the PS II reaction center P-680 pigment biosynthesis from the long-wavelength protochlorophyllide form was observed by spectral methods under illumination of etiolated leaves by red light at 4°C. At room temperature, these reactions (reactions (11) and (12) on the scheme) are harder to detect since under long-wavelength illumination, the main chlorophyll precursor form Pchld 655/650 is transformed as well, and both parallel reaction pathways occur under these conditions.
As shown earlier in our studies using monochromatic 690-nm illumination (using a monochromator) [53] or 694 nm (using a ruby laser) [54], at room temperature the long-wavelength Pchld 710/697 form and the newly synthesized chlorophyllide sensitize the photoconversion of the main Pchld 655/650 precursor form. If the temperature was decreased, Pchld 655/650 sensitization dropped rapidly.
These results indicate that a part of the minor long-wavelength protochlorophyllide form is included in the photoactive complex of the chlorophyll precursor and is not only connected energetically to the main protochlorophyllide Pchld 655/650 form, but also directly participates in chlorophyll formation, in particular, the formation of PS II RC chlorophyll.
FINAL STAGES OF CHLOROPHYLL BIOSYNTHESIS IN GREEN LEAVES OF
PLANTS
A question arises of whether the pathways and mechanisms of chlorophyll photobiosynthesis in greening leaves are similar to those during the accumulation and renewal of its storage in adult green plant leaves, when the bulk of the pigment is already synthesized. This process provides the necessary photosynthesis capacity, a high level of energy absorption (in the antennae) and its conversion into chemical bond energy. For this purpose, a high rate of chlorophyll biosynthesis must ensure pigment accumulation in growing leaves as well as compensate for its loss in destructive processes, photodestruction in particular, which is especially prevalent under intense illumination.
Using low temperature (77 K) fluorescence spectrophotometry, which decreases overlapping of chlorophyll and protochlorophyllide spectral bands, temporary darkening of green leaves was shown to cause accumulation of a chlorophyll precursor that is spectroscopically identical to the main active protochlorophyllide form in etiolated leaves (the main fluorescence maximum is at 655 nm). This compound rapidly vanishes under the following illumination of darkened leaves [9, 55-58]. In [58], it was shown that after 16 h of darkening, three protochlorophyllide forms accumulate in leaves, which are characterized by the same spectral parameters as the forms in etiolated leaves. Fluorescence maxima were observed at 633 nm (the main maximum in the blue region of the excitation spectrum is at 440 nm), 642 nm (444 nm), and 655 nm (448 nm). The main photoactive form was Pchld 655/650 (448 nm). The band at 642 nm disappeared quite slowly and with low quantum yield. The decrease of this band at 642 nm may be connected not to protochlorophyllide photoreduction, but to photodestruction of the pigment. Taking into account the role of Pchld 653/648(440) (the short-wavelength component of the main active protochlorophyllide form) as a PS II RC Chl a precursor in juvenile etiolated leaves (see section “Dependence of chlorophyll generation pathways for different chlorophyll forms on spectral composition of exciting light in etiolated leaves”), we conducted an experiment with monochromatic illumination of dark-adapted green barley leaves with the same setup as in the experiments with etiolated leaves. The measurements of fluorescence spectra for dark-adapted leaves under successive illuminations were conducted as such: 650 nm → 680 nm → 650 nm. It was found that for green leaves, as well as for young etiolated leaves, Chl a biosynthesis includes the Pchld 653/648(440) precursor form, which is in turn a product of a photochemical reaction of a longer wavelength protochlorophyllide form with an absorption maximum near 680 nm. We propose two chlorophyll biosynthesis pathways to occur in green and etiolated leaves.
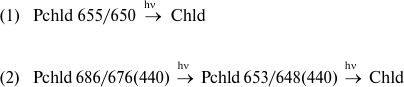
Therefore, we concluded that in the process of chlorophyll photobiosynthesis from its precursor protochlorophyllide, parallel pathways of conversion of different protochlorophyllide forms are found depending on the actinic light wavelength. Using red light, a pathway of early PS II RC P-680 pigment formation was revealed, whose precursor is the long-wavelength protochlorophyllide form. The cause of variations in spectral characteristics of primary fluorescent chlorophyllide forms Chld 695/684 and Chld 684/676, depending on the spectral composition of the actinic light (see last section), can therefore be proposed to be preferential red light absorption by minor long-wavelength protochlorophyllide forms with their further disaggregation into Pchld 653/648, which is transformed on illumination into Chld 684/676, meaning that a parallel Pchld 684/676 biosynthesis pathway is observed (reactions (11) and (12) on the general scheme). When using blue or white light, this reaction is not detected against the background of the more effective reaction of Pchld 655/650 conversion through the Chld 695/684 form (not shown on the scheme) into Chld 684/676 (reactions (1) and (2) on the scheme) with the following formation of light-harvesting complex chlorophyll.
The differences in primary reaction sequences for chlorophyll biosynthesis in initial stages of greening, which depend on spectral composition of the actinic light, can be explained by participation of various protochlorophyllide spectral forms in the reactions of reaction center and antennae chlorophyll biosynthesis.
Moreover, use of red light demonstrated that in dark-adapted adult green plants chlorophyll photobiosynthesis is carried out through the same protochlorophyllide active forms as in etiolated leaves, and their conversion pathways are similar to those observed for greening etiolated leaves.
REFERENCES
1.Kahn, A., Boardman, N. K., and Thorn, S. W. (1970)
Energy transfer between protochlorophyllide molecules: evidence for
multiple chromophores in the photoactive
protochlorophyllide–protein complex in vivo and in
vitro, J. Mol. Biol., 48, 85-101.
2.Dujardin, E., and Sironval, C. (1970) The reduction
of protochlorophyllide into chlorophyllide. III. The
phototransformability of the protochlorophyllide lipoprotein complex
found in darkness, Photosynthetica, 4, 129-138.
3.Gassman, M. L. (1973) The conversion of
photoinactive protochlorophyll(ide)633 to phototransformable
protochlorophyll(ide)650 in etiolated bean leaves, treated
with δ-aminolevulinic acid, Plant Physiol., 52,
590-594.
4.Virgin, H. I. (1975) In vivo absorption
spectra of protochlorophyll650 and
protochlorophyll636 within the region 530-700 nm,
Photosynthetica, 9, 84-92.
5.Litvin, F. F., and Stadnichuk, I. N. (1980) Long
wave chlorophyll precursors in etiolated leaves and the system of
protochlorophyll native forms, Plant Physiol., 27,
1024-1032.
6.Boddi, B., Lindsten, A., Ryberg, M., and Sundqvist,
C. (1989) On the aggregational states of protochlorophyllide and its
protein complexes in wheat etioplasts, Physiol. Plant.,
76, 135-143.
7.Boddi, B., Ryberg, M., and Sundqvist, C. (1992)
Identification of four universal protochlorophyllide forms in
dark-grown leaves by analyses of the 77 K fluorescence emission
spectra, J. Photochem. Photobiol., 12, 389-401.
8.Boddi, B., and Franck, F. (1997) Room temperature
fluorescence spectra of protochlorophyllide and chlorophyllide forms in
etiolated bean leaves, J. Photochem. Photobiol. B Biol.,
41, 73-82.
9.Schoefs, B., Bertrand, M., and Franck, F. (2000)
Spectroscopic properties of protochlorophyllide analyzed in situ
in the course of etiolation and in illuminated leaves, Photochem.
Photobiol., 72, 85-93.
10.Stadnichuk, I. N., Amirjani, M. R., and
Sundqvist, C. (2005) Identification of spectral forms of
protochlorophyllide in the region 670-730 nm, Photochem. Photobiol.
Sci., 4, 230-238.
11.Belyaeva, O. B., and Litvin, F. F. (1989)
Photobiosynthesis of Chlorophyll [in Russian], MGU Publishers,
Moscow, p. 102.
12.Belyaeva, O. B. (2009) Light-Independent
Biosynthesis of Chlorophyll [in Russian], Binom, Moscow, p.
232.
13.Belyaeva, O. B., and Litvin, F. F. (2009)
Pathways of pigment forms synthesis at terminal photochemical stage of
chlorophyll biosynthesis, Adv. Biol. Chem., 49,
319-340.
14.Belyaeva, O. B., and Litvin, F. F. (2014)
Mechanisms of phototransformation of protochlorophyllide into
chlorophyllide, Biochemistry (Moscow), 79, 337-348.
15.Ignatov, N. V., and Litvin, F. F. (1996)
Photoconversion of long-wavelength protochlorophyll native form Pchl
682/672 into chlorophyll Chl 715/696 in Chlorella vulgaris B-15,
Photosynth. Res., 50, 271-283.
16.Schoefs, B., and Franck, F. (2008) The
photoenzymatic cycle of NADPH:protochlorophyllide oxidoreductase in
primary bean leaves (Phaseolus vulgaris) during the first days
of photoperiodic growth, Photosynth. Res., 96, 15-26.
17.Belyaeva, O. B., and Litvin, F. F. (2007)
Photoactive pigment–enzyme complexes of chlorophyll precursor in
plant leaves, Biochemistry (Moscow), 72, 1458-1477.
18.Rubin, A. B., Minchenkova, L. E., Krasnovsky, A.
A., and Tumerman, L. A. (1962) Study of the duration of
protochlorophyllide fluorescence during the etiolated leaves greening,
Biofizika, 7, 571-577.
19.Goedheer, J., and Verhulsdonk, C. (1970)
Fluorescence and phototransformation of protochlorophyll with etiolated
bean leaves from –196 to +20°C, Biochem. Biophys. Res.
Commun., 39, 260-266.
20.Sironval, C., and Kuyper, P. (1972) The reduction
of protochlorophyllide into chlorophyllide. IV. The nature of the
intermediate P688-676 species, Photosynthetica,
6, 254-275.
21.Raskin, V. I. (1976) Mechanisms of
protochlorophyllide photoreduction in intact etiolated leaves, News
Acad. Sci. BSSR, 5, 43-46.
22.Dujardin, E., and Correia, M. (1979)
Long-wavelength absorbing pigment protein complexes as fluorescence
quenchers in etiolated leaves illuminated in liquid nitrogen,
Photobiochem. Photobiophys., 1, 25-32.
23.Correia, M., and Dujardin, E. (1983) Kinetics of
the action of intrinsic fluorescence quenchers in etiolated, greening
and green leaves illuminated at 77 K, Photobiochem.
Photobiophys., 5, 281-292.
24.Dujardin, E. (1984) The long-wavelength-absorbing
quenchers formed during illumination of protochlorophyllide-proteins,
in Protochlorophyllide Reduction and Greening (Sironval, C., and
Brouers, M., eds.) Martinus Nijhoff/Dr. W. Junk Publisher, The Hague,
pp. 87-98.
25.Losev, A. P., and Lyalkova, N. D. (1979) Primary
stages of photohydration of protochlorophyllide in etiolated leaves,
Mol. Biol., 13, 837-844.
26.Litvin, F. F., and Ignatov, N. V. (1980)
Reversibility of protochlorophyllide transformation in chlorophyllide
in etiolated plant leaves under light treatment, Dokl. AN SSSR,
250, 1463-1465.
27.Belyaeva, O. B., and Litvin, F. F. (1980) New
intermediate reactions in protochlorophyllide photoreduction process,
Biofizika, 25, 617-623.
28.Belyaeva, O. B., and Litvin, F. F. (1981) Primary
reactions of protochlorophyllide into chlorophyllide
phototransformation at 77 K, Photosynthetica, 15,
210-215.
29.Litvin, F. F., Ignatov, N. V., and Belyaeva, O.
B. (1981) Photoreversibility of transformation of protochlorophyllide
into chlorophyllide, Photobiochem. Photobiophys., 2,
233-237.
30.Belyaeva, O. B., Personova, E. R., and Litvin, F.
F. (1983) Photochemical reaction of chlorophyll biosynthesis at 4.2K,
Photosynth. Res., 4, 81-85.
31.Frank, F., Dujardin, E., and Sironval, C. (1980)
Non-fluorescent, short-lived intermediate in photoenzymatic
protochlorophyllide reduction at room temperature, Plant Sci.
Lett., 18, 375-380.
32.Franck, F., and Mathis, P. (1980) A short-lived
intermediate in the photoenzymatic reduction of protochlorophyll(ide)
into chlorophyll(ide) at a physiological temperature, Photochem.
Photobiol., 32, 799-803.
33.Inoue, Y., Kobayashi, T., Ogawa, T., and Shibata,
K. (1981) A short intermediate in the photoconversion of
protochlorophyllide to chlorophyllide a, Plant Cell
Physiol., 22, 197-204.
34.Iwai, J., Ikeuchi, M., Inoue, Y., and Kobayashi,
T. (1984) Early processes of protochlorophyllide photoreduction as
measured by nanosecond and picosecond spectrophotometry, in
Protochlorophyllide Reduction and Greening (Sironval, C., and
Brouers, M., eds.) Martinus Nijhoff/Dr. W. Junk Publisher, The Hague,
pp. 99-112.
35.Belyaeva, O. B., Timofeev, K. N., and Litvin, F.
F. (1987) Study of the nature of intermediate products of
protochlorophyll(ide) photoreduction in vitro and in vivo
by optic and EPR-spectroscopy, Biofizika, 32,
104-109.
36.Belyaeva, O. B., Timofeev, K. N., and Litvin, F.
F. (1988) The primary reaction in the protochlorophyll(ide)
photoreduction as investigated by optical and ESR-spectroscopy,
Photosynth. Res., 15, 247-256.
37.Ignatov, N. V., Belyaeva, O. B., and Litvin, F.
F. (1993) Low temperature phototransformation of protochlorophyll(ide)
in etiolated leaves, Photosynth. Res., 38, 117-124.
38.Lebedev, N. N., and Timko, M. (1999)
Protochlorophyllide oxidoreductase B-catalyzed protochlorophyllide
photoreduction in vitro: insight into the mechanism of
chlorophyll formation in light-adapted plant, Proc. Natl. Acad. Sci.
USA, 96, 17954-17959.
39.Belyaeva, O. B., Griffiths, W. T., Kovalev, J.
V., Timofeev, K. N., and Litvin, F. F. (2001) Participation of free
radicals in photoreduction of protochlorophyllide to chlorophyllide in
an artificial pigment–protein complex, Biochemistry
(Moscow), 66, 173-177.
40.Heyes, D. J., Ruban, A. V., Wilks, H. M., and
Hunter, C. N. (2002) Enzymology below 200 K: the kinetics and
thermodynamics of the photochemistry catalyzed by protochlorophyllide
oxidoreductase, PNAS, 99, 11145-11150.
41.Heyes, D. J., Ruban, A. V., and Hunter, C. N.
(2003) Protochlorophyllide oxidoreductase: “Dark” reaction
of a light-driven enzyme, Biochemistry, 42, 523-528.
42.Heyes, D. J., Hunter, C. N., Van Stokkum, I. H.
M., Grondelle, R., and Groot, M. L. (2003) Ultrafast enzymatic reaction
dynamics in protochlorophyllide oxidoreductase, Nat. Struct.
Biol., 10, 491-492.
43.Heyes, D. J., Heathcote, P., Rigby, S. E. J.,
Palacios, M. A., Grondelle, R., and Hunter, C. N. (2006) The first
catalytic step of the light-driven enzyme protochlorophyllide
oxidoreductase proceeds via a charge transfer complex, J. Biol.
Chem., 281, 26847-26853.
44.Sytina, O. A., Heyes, D. J., Hunter, C. N.,
Alexandre, M. T., Van Stokkum, I. H. M., Van Grondelle, R., and Groot,
M. L. (2008) Conformational changes in an ultrafast light-driven enzyme
determine catalytic activity, Nature, 456, 1001-1005.
45.Belyaeva, O. B. (1994) Final Stages of
Chlorophyll Photosynthesis in Plant Leaves: Author’s abstract
of doctoral dissertation [in Russian], Institute of Soil Science and
Photosynthesis, Russian Academy of Sciences, Pushchino.
46.Raskin, V. I., and Schwartz, A. (2002) The
charge-transfer complex between protochlorophyllide and NADPH: an
intermediate in protochlorophyllide photoreduction, Photosynth.
Res., 74, 181-186.
47.Griffiths, W. T., McHugh, T., and Blankenship, R.
E. (1996) The light intensity dependence of protochlorophyllide
photoreduction and its significance to the catalytic mechanism of
protochlorophyllide reductase, FEBS Lett., 398,
235-238.
48.Belyaeva, O. B., and Sundqvist, C. (1998)
Comparative investigation of the appearance of primary chlorophyllide
forms in etiolated leaves, prolamellar bodies and prothylakoids,
Photosynth. Res., 55, 41-48.
49.Dobek, A., Dujardin, E., Franck, F., Sironval,
C., Breton, J., and Roux, E. (1981) The first events of
protochlorophyll(ide) photoreduction investigated in etiolated leaves
by means of the fluorescence excited by short, 610 nm laser flashes at
room temperature, Photobiochem. Photobiophys., 2,
35-44.
50.Ignatov, N. V., and Litvin, F. F. (2002) A new
pathway of chlorophyll biosynthesis from long-wavelength
protochlorophyllide Pchld 686/676 in juvenile etiolated plants,
Photosynth. Res., 71, 195-207.
51.Schoefs, B., and Franck, F. (1993) Photoreduction
of protochlorophyllide to chlorophyllide in 2-d-old dark-grown bean
(Phaseolus vulgaris cv. Commodore) leaves. Comparison with
10-d-old dark-grown (etiolated) leaves, J. Exp. Bot., 44,
1053-1057.
52.Franck, F., and Strzalka, K. (1992) Detection of
the photoactive protochlorophyllide–protein complex in the light
during the greening of barley, FEBS Lett., 309,
73-77.
53.Belyaeva, O. B., Boddi, B., Ignatov, N. V., Lang,
F., and Litvin, F. F. (1984) The role of a long-wavelength pigment
forms in the chlorophyll biosynthesis, Photosynth. Res.,
5, 263-271.
54.Ignatov, N. V., Belyaeva, O. B., and Litvin, F.
F. (1983) Sensibilization of photochemical stage of chlorophyll
formation by long wave forms of protochlorophyll(ide) and
chlorophyllide under laser treatment, Proc. Acad. Sci. USSR,
273, 737-740.
55.Litvin, F. F., Krasnovsky, A. A., and Rikhireva,
G. T. (1959) Formation and transformation of protochlorophyll in green
plant leaves, Proc. Acad. Sci. USSR, 127, 699-701.
56.Garab, G. T., Sundqvist, C., and Faludi-Daniel,
A. (1980) Detection of protochlorophyllide forms in green leaves,
Photochem. Photobiol., 31, 491-503.
57.Lebedev, N. N., Siffel, P., and Krasnovsky, A. A.
(1985) Spectral characteristics of protochlorophyllide and chlorophyll
fluorescence in green leaves and isolated chloroplasts,
Biofizika, 30, 44-49.
58.Ignatov, N. V., and Litvin, F. F. (2002)
Biosynthesis of chlorophyll from protochlorophyll(ide) in green plant
leaves, Biochemistry (Moscow), 67, 949-955.