Effects of Ouabain on Proliferation of Human Endothelial Cells Correlate with Na+,K+-ATPase Activity and Intracellular Ratio of Na+ and K+
A. M. Tverskoi1, S. V. Sidorenko1, E. A. Klimanova1, O. A. Akimova1, L. V. Smolyaninova1, O. D. Lopina1, and S. N. Orlov1,2,3*
1Lomonosov Moscow State University, Faculty of Biology, 119991 Moscow, Russia; E-mail: sergeinorlov@yandex.ru2Tomsk National Research State University, 634050 Tomsk, Russia
3Siberian State Medical University, 634050 Tomsk, Russia
* To whom correspondence should be addressed.
Received March 10, 2016; Revision received May 3, 2016
Side-by-side with inhibition of the Na+,K+-ATPase ouabain and other cardiotonic steroids (CTS) can affect cell functions by mechanisms other than regulation of the intracellular Na+ and K+ ratio ([Na+]i/[K+]i). Thus, we compared the dose- and time-dependences of the effect of ouabain on intracellular [Na+]i/[K+]i ratio, Na+,K+-ATPase activity, and proliferation of human umbilical vein endothelial cells (HUVEC). Treatment of the cells with 1-3 nM ouabain for 24-72 h decreased the [Na+]i/[K+]i ratio and increased cell proliferation by 20-50%. We discovered that the same ouabain concentrations increased Na+,K+-ATPase activity by 25-30%, as measured by the rate of 86Rb+ influx. Higher ouabain concentrations inhibited Na+,K+-ATPase, increased [Na+]i/[K+]i ratio, suppressed cell growth, and caused cell death. When cells were treated with low ouabain concentrations for 48 or 72 h, a negative correlation between [Na+]i/[K+]i ratio and cell growth activation was observed. In cells treated with high ouabain concentrations for 24 h, the [Na+]i/[K+]i ratio correlated positively with proliferation inhibition. These data demonstrate that inhibition of HUVEC proliferation at high CTS concentrations correlates with dissipation of the Na+ and K+ concentration gradients, whereas cell growth stimulation by low CTS doses results from activation of Na+,K+-ATPase and decrease in the [Na+]i/[K+]i ratio.
KEY WORDS: proliferation, endothelium, Na+,K+-ATPase, ouabain, intracellular Na+ and K+DOI: 10.1134/S0006297916080083
Abbreviations: CTS, cardiotonic steroids; HUVEC, human umbilical vein endothelial cells; IEG, immediate early genes.
Na+,K+-ATPase is a heterodimer protein composed of
a catalytic α-subunit and a regulatory β-subunit.
Na+,K+-ATPase plays a key role in maintaining the
electrochemical gradient of monovalent cations across the plasma
membrane in all studied animal cells. Ouabain and other cardiotonic
steroids (CTS) inhibit Na+,K+-ATPase by
interacting with its ubiquitous α1- and tissue-specific
α2-α4 subunits. This inhibition results in the dissipation
of Na+ and K+ transmembrane gradients, which in
turn affects cell mechanisms and parameters regulated by the ratio
between intracellular concentrations of Na+ and
K+
([Na+]i/[K+]i), such as
plasma membrane electric potential, cell volume, transepithelial
transfer of salts and osmotically bound water, Na+ symport
with glucose, amino acids, and nucleotides, etc. [1-3]. More recently, it was shown
that long-term treatment of cells with CTS activates expression of
c-Fos and other immediate early genes (IEG), as well as genes
for myosin light chain, actin, atrial natriuretic factor, mortalin, and
other tissue-specific late response genes [4-6]. Using Affymetrix technology, we demonstrated that
in human umbilical vein endothelial cells (HUVEC) and in several other
mammalian cell cultures incubated in a K+-free medium,
inhibition of Na+,K+-ATPase shifts RNA expression
into a pattern similar to that observed after treatment of cells with
ouabain [7]. These data suggest the existence of a
new mechanism for coupling of cell excitability and gene transcription
that is mediated by increase in the
[Na+]i/[K+]i ratio [8].
Experimental data obtained during the last 20 years suggest that, in addition to regulating Na+i,K+i-sensitive cell mechanisms, CTS can also affect cell functions independently of inhibition of the Na+,K+-pump. This makes CTS a new class of steroid hormones. Thus, for example, relatively low concentrations of ouabain and other CTS caused binding of Na+,K+-ATPase α-subunit to the non-receptor Src tyrosine protein kinase, phosphatidylinositol 3-kinase, and inositol-1,4,5-trisphosphate receptor [9-11]. It should be noted that in most studies, CTS-induced changes in ion balance and the above-mentioned functional responses were studied within different time intervals. However, the time course of such experiments should be of particular importance because of extremely slow kinetics of the interaction of ouabain with the human house-keeping α1-isoform of Na+,K+-ATPase [12]. For this reason, we studied the dose- and time-dependences of effects of ouabain on HUVEC proliferation and intracellular K+ and Na+ content in these cells.
MATERIALS AND METHODS
Reagents. 22NaCl and 86RbCl were purchased from Perkin Elmer (USA). All other reagents were from Gibco BRL (USA), Calbiochem (USA), Sigma (USA), and Anachemia (Canada).
Cell culture. Human umbilical vein endothelial cells (HUVEC) were purchased from Lonza (USA). The cells were cultured in Endothelial Cell Growth Medium 2 (EGM-2 BulletKit, CC3162; Lonza) supplemented with 10% fetal bovine serum (FBS) in 5% CO2 at 37°C (the number of passages did not exceed 8). Before the experiment, the cells were incubated for 24 h in the medium containing 0.2% FBS, then washed with phosphate-buffered saline (PBS), and incubated for up to 72 h in EGM-2 containing 0.2% FBS and 1-3000 nM ouabain.
Cell viability was determined by the caspase-3 activity method from the fluorescence of the substrate DEVD-AMC (N-acetyl-Asp-Glu-Val-Asp-AMC); Ac-DEVD-CHO was used as caspase-3 inhibitor. The cells were grown in 6-well plates. Before the measurements, the plates were transferred onto ice, and the cells were scraped off and centrifuged at 5000g for 10 min at 4°C. The pellet was washed twice with 3 ml of cold PBS and then resuspended in 150 µl of medium containing 0.32 M sucrose, 5 mM EDTA, 10 mM Tris-HCl (pH 8.0), 1% Triton X-100, 2 mM dithiothreitol (DTT), 1 mM phenylmethanesulfonyl fluoride (PMSF), 10 µg/ml pepstatin A, and 10 µg/ml aprotinin. The lysates were centrifuged, and 100-µl aliquots of the supernatant were frozen in liquid nitrogen and stored at –80°C. To measure enzymatic activity, 20 µl of the sample was mixed with 400 µl of medium containing 5 mM MgCl2, 1 mM EGTA, 50 mM Tris-HCl (pH 7.0), 0.1% CHAPS, 1 mM DTT, 40 µM DEVD-AMC in the absence or presence of 2 µM Ac-DEVD-CHO. After the mixture was incubated for 3 h at 37°C, the reaction was stopped by adding 1 ml of 0.5 M glycine-NaOH buffer (pH 10.0). The reaction mixture was diluted with water, and fluorescence was measured at 465 nm (λex, 365 nm). The calibration curve was plotted using 0.01-0.3 µM 7-amino-4-methylcoumarin. Caspase-3 activity was determined as the difference between the rates of DEVD-AMC hydrolysis in the presence and absence of Ac-DEVD-CHO as described earlier in detail [13].
Microsomes were isolated from pig kidney outer medulla. The medulla was diced with scissors and mixed with 10 volumes (w/v) of the isolation medium containing 0.25 M sucrose, 1 mM EDTA, 20 mM Tris-HCl (pH 7.5), 1 mM DTT, 5 µM thiorphan, 0.2 mM PMSF, and protease inhibitor cocktail (see above). The suspension was disrupted with a Potter–Elvehjem tissue grinder, and the homogenate was filtered through three layers of pre-boiled gauze wetted with the isolation medium and centrifuged at 5000g for 15 min. The supernatant was collected into a separate tube and stored at 4°C; the pellet was resuspended in two volumes (w/v) of the isolation medium and centrifuged at 5000g for 15 min. The supernatants were combined and centrifuged at 45,000g for 1 h. The pellet was resuspended in a minimal volume of the isolation medium and stored at –20°C. Protein concentration in the microsomal fraction was determined by the Lowry method.
Cell proliferation was determined after the cells were plated in 24-well plates and treated with experimental agents as described below. The cells were detached from the plates with PBS containing 0.05% trypsin and 0.1% EDTA, washed with PBS supplemented with 5% FBS, and resuspended in PBS containing 0.2% Trypan Blue. Live cells (not stained by Trypan Blue) were counted with a hemocytometer.
Activity of the Na+,K+-pump was quantified as the ouabain-sensitive component of the 86Rb+ influx rate. Cells in 24-well plates were washed twice with 2 ml of medium A containing 140 mM NaCl, 5 mM KCl, 1 mM MgCl2, 1 mM CaCl2, 5 mM D-glucose, and 20 mM HEPES-Tris (pH 7.4) and incubated in the same medium for 1 h at 37°C. The medium was then replaced with medium A (0.25 ml) containing various ouabain concentrations. After 5-min incubation, 0.25 ml of medium A containing 1 µCi/ml 86Rb+ and 20 µM bumetanide (Na+,K+,2Cl– cotransporter inhibitor) were added. The kinetics of isotope uptake stayed linear up to 10 min of incubation (data not shown). In our study, isotope uptake was stopped after 5 min of incubation by adding 2 ml of ice-cold medium W containing 100 mM MgCl2 and 10 mM HEPES-Tris (pH 7.4). The cells were then washed four times with 2 ml of medium W and lysed with 1 ml of 1% sodium dodecyl sulfate containing 4 mM EDTA. The radioactivity of the incubation medium and lysates was measured with a liquid scintillation counter. The rate of 86Rb+ influx was calculated as V = A/a·m·t, where A is sample radioactivity (counts per min, cpm), a is specific radioactivity of K+ (86Rb+) in the medium (cpm/nmol), m is protein content (mg), and t is incubation time with the isotope (5 min).
Na+,K+-ATPase activity in pig kidney microsomes was measured as described in [14] from inorganic phosphate accumulation in medium containing 120 mM NaCl, 30 mM KCl, 3 mM MgCl2, 3 mM ATP, and 30 mM imidazole (pH 7.4). The protein concentration was 0.01 mg/ml. The reaction was initiated by adding ATP. For more details, see [15].
Intracellular concentrations of exchangeable K+ and Na+ were measured from the steady-state distribution of extra- and intracellular 86Rb+ and 22Na+, respectively, as described in detail in [7, 16]. Cells grown in 24- and 12-well plates (for 86Rb+ and 22Na+ measurements, respectively) were incubated for up to 72 h in the presence or absence of various ouabain concentrations. The plates were transferred onto ice, and the cells were washed four times with 2 ml of medium W. The cells were then lysed as described above, and radioactivity of the incubation medium and cell lysates was quantified. The intracellular cation content was calculated as A/a·m, where A is sample radioactivity (cpm), a is specific radioactivity of 86Rb+ (K+) or 22Na+ in the medium (cpm/nmol), and m is protein content (mg).
Statistical data analysis was performed using Student’s t-test. Differences were considered significant at p < 0.05.
RESULTS
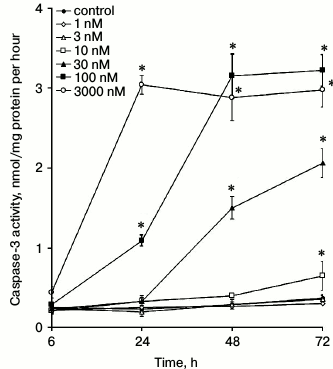
Viability of ouabain-treated cells. It was shown earlier that prolonged incubation with high CTS concentrations causes death of human and porcine endothelial cells [17, 18]. Here, we examined dose- and time-dependences of ouabain action on the viability of HUVEC. Figure 1 shows that prolonged incubation with ouabain increases its cytotoxic effect, as estimated from caspase-3 activation. Indeed, an increase in caspase-3 activity was observed after 6, 24, 48, and 72 h of incubation with 3000, 100, 30, and 10 nM ouabain, respectively. Based on these results, for further studies of cell proliferation and intracellular contents of monovalent cations, we select time intervals and ouabain doses that did not affect cell survival. Thus, for example, when ouabain was used in concentrations of 3000 and 30 nM, incubation times were limited to 6 and 48 h, respectively.
Fig. 1. Effect of ouabain on caspase-3 activity in HUVEC. Data from three independent experiments (four measurements per experiment) are presented as mean ± SE; * p < 0.05 as compared to control cells incubated for the same times in the absence of ouabain.
Intracellular contents of Na+ and K+. Ouabain in concentrations of 1 and 3 nM increased K+i and decreased Na+i contents by 20-30% (table). These differences were significant after 6, 24, and 48 h, but disappeared after 72 h of incubation. As expected, higher ouabain concentrations dissipated the transmembrane gradient of monovalent cations due to inhibition of the Na+,K+-ATPase. It should be noted that the apparent affinity for ouabain increased with increase in the incubation time. Thus, 100 nM ouabain increased [Na+]i to its half-maximal value after 6-h incubation, whereas after 24 and 48 h of incubation, the same effect was documented in the presence of 3 and 10 nM ouabain, respectively.
Dose- and time-dependence of ouabain effects on the Na+ and
K+ intracellular contents and
[Na+]i/[K+]i ratio in HUVEC
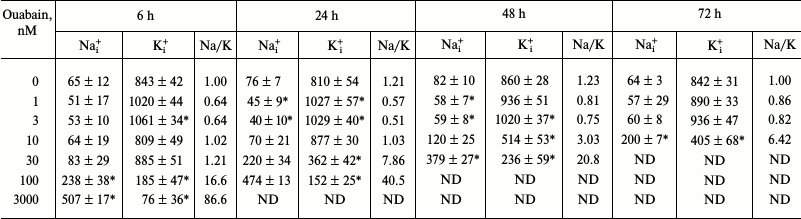
Note: Data are presented as mean ± S.E. (n = 4), *
p < 0.05;
[Na+]i/[K+]i ratio after
6 h of incubation in the absence of ouabain was taken as 1.00; ND,
not determined.
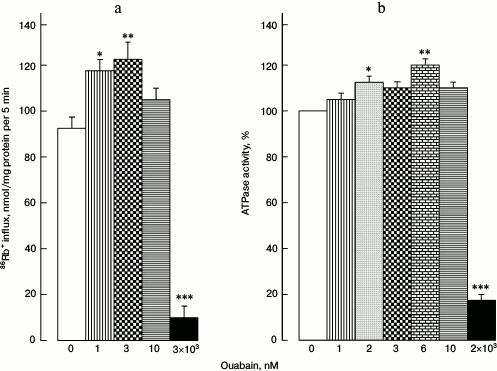
Activities of Na+,K+-pump and ATPase. The reason for increased [Na+]i/[K+]i ratio in cells treated with 1 and 3 nM ouabain might be the activation of Na+,K+-ATPase. Indeed, ouabain (1-3 nM) increased 86Rb+ influx by 25-30% when measured in the presence of the Na+,K+,2Cl–-cotransporter selective inhibitor bumetanide (Fig. 2a).
Fig. 2. 86Rb+ influx in HUVEC (a) and ATPase activity in pig kidney microsomes (b) at different ouabain concentrations. Data from four independent experiments are presented as mean ± SE; * p < 0.05, ** p < 0.01, *** p < 0.001.
To determine if Na+,K+-ATPase activation by low doses of ouabain is preserved in a cell-free system, we used pig kidney microsomes. Ouabain (1-10 nM) activated ATPase by 15-20% (Fig. 2b). As expected, further increase in ouabain concentration to 2-3 µM caused abrupt decrease in both 86Rb+ influx rate and ATPase activity.
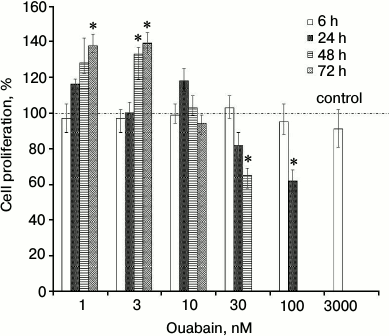
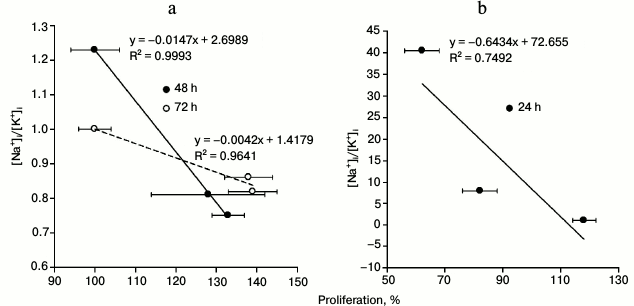
Cell proliferation. Figure 3 displays that exposure to 1 and 3 nM ouabain for 48 and 72 h increased proliferation of HUVEC by ~30-40% as determined from Trypan Blue staining (Fig. 3). Note that the number of dead cells (stained with Trypan Blue) did not exceed 2-4% of the total number of cells. This observation correlates well with the absence of caspase-3 activity increase for the incubation times and ouabain concentrations used in this experiment (Fig. 1). Prolonged incubation with higher ouabain concentrations (above 30 nM) resulted in cell growth inhibition by 20-40%, which is in a good agreement with earlier results on the opposite action of high and low ouabain doses on proliferation of rat astrocytes [19], prostate smooth muscle cells [20], canine, human, rat, and bovine vascular smooth muscle cells [21, 22], human fibroblasts [23], and human endothelial cells [24, 25]. To examine the mechanism of this phenomenon, we compared dose-dependence actions of ouabain on cell proliferation and on intracellular content of monovalent cations. We found negative correlation between [Na+]i/[K+]i ratio and activation of proliferation of HUVEC treated with low doses of ouabain for 48 and 72 h (Fig. 4a) and positive correlation between [Na+]i/[K+]i ratio and inhibition of proliferation of cells treated with high doses of ouabain for 24 h (Fig. 4b).
Fig. 3. HUVEC proliferation at different ouabain concentrations. The number of live cells after 6, 24, 48, and 72 h of incubation in the absence of ouabain was taken as 100%. Data from four independent experiments (four measurements per experiment) are presented as mean ± SE; * p < 0.05 as compared to control cells incubated for the same periods in the absence of ouabain.
Fig. 4. Correlation between [Na+]i/[K+]i ratio and HUVEC proliferation 48 and 72 h after addition of 1 and 3 nM ouabain (a) and 24 h after addition of 10, 30, and 100 nM ouabain (b). The data for [Na+]i/[K+]i and HUVEC proliferation are taken from the table and Fig. 3, respectively.
DISCUSSION
Evidence of CTS-induced Na+i,K+i-independent signaling was suggested from the fact that low CTS concentrations affect cell functions without noticeable changes in the Na+,K+-pump activity and/or the content of intracellular Na+ and K+. Nevertheless, it should be noted that, except in rare cases, CTS regulation of Na+,K+-pump activity, [Na+]i/[K+]i ratio, and cell functions has been studied during different time intervals. Thus, activation of vascular smooth muscle cell proliferation was observed 48 h after addition of 1 nM ouabain, while the independence of this effect on the [Na+]i/[K+]i ratio was suggested based on the data that demonstrated no changes in 86Rb+ influx within 10 min of incubation [21, 26]. Therefore, in this work we performed a comparison of the dose- and time-dependence of ouabain action on the [Na+]i/[K+]i ratio and HUVEC proliferation. This approach is also important because high CTS concentrations exhibit cytotoxic effect when incubated with endothelial cells for prolonged periods [17, 18, 24, 25]. For this reason, we first studied the dose-dependence and the time course of ouabain cytotoxic action (Fig. 1) and only then assessed ion balance and cell proliferation in viable cell cultures with constant caspase-3 activity levels and not stained by Trypan Blue.
Comparison of effects of different ouabain doses on the intracellular concentrations of monovalent cations at different time periods showed that the threshold of ouabain concentration required for increase in the [Na+]i/[K+]i ratio decreases more than 10 times with an increase in the incubation time from 6 to 72 h (table). It should be noted the intracellular K+ concentrations were calculated using 86Rb+. The atomic radii of K+ and 86Rb+ differ slightly – 1.33 and 1.49 Å, respectively. It is known that the Na+,K+-pump displays no selectivity toward these ions; however, it remains unknown whether other transporters and potassium-binding cytoplasmic molecules can be affected by this difference. This question will be resolved by us in the near future through additional studies using atomic absorption spectroscopy. The delayed kinetics of ouabain action on intracellular Na+ and K+ concentrations might be explained by the slow kinetics of its binding to Na+,K+-ATPase [12] and/or Na+,K+-ATPase accumulation in the CTS-sensitive E2 conformation resulting from the increase in intracellular Na+ with increase in incubation time. Indeed, Holthouse and co-workers [27] found that in human kidney proximal tubule cells, low ouabain doses activate Na+,K+-ATPase by increasing activity of the ubiquitous isoform of the Na+/H+ exchanger NHE1 that mediates Na+ influx. The observed effect might also be explained by circulation of Na+,K+-ATPase between its membrane-bound and internalized forms. However, the latter hypothesis contradicts the data on the activation of Na+,K+-ATPase by low doses of ouabain in the cell-free system (pig kidney microsomes) (Fig. 2b). Irrespectively of the mechanism of the observed phenomenon, our data show the importance of measuring effects of CTS on the intracellular ion homeostasis and cell functions within the same time intervals.
We found that prolonged incubation of cells with 1 and 3 nM ouabain was accompanied by K+i accumulation and decrease in Na+i content, with corresponding 30-50% decrease in [Na+]i/[K+]i ratio (table). Also, low ouabain doses increased, rather than decreased, 86Rb+ influx rate (Fig. 2), thereby indicating that the [Na+]i/[K+]i increase is mediated by Na+,K+-pump activation. This conclusion correlates well with data on Na+,K+-ATPase activation by low CTS concentrations observed by other researchers. Thus, it was shown that ouabain in concentrations below 10 nM decreased Na+i content in guinea pig atrial [28, 29] and attenuated Na+,K+-pump-mediated ion flux in single cardiomyocytes from guinea pig, dog, and human [30]. In human erythrocytes, activation of 86Rb+ uptake occurred at ouabain concentration of 0.1 nM [31], whereas in opossum kidney proximal tubule cells and human endothelial cells, similar activation was observed at ouabain concentrations of 1-10 nM [32, 33]. Ouabain and other CTS also activated 86Rb+ uptake in hippocampal slice cultures [34].
To explain this phenomenon, Gao and co-workers suggested that ouabain activates Na+,K+-ATPase by interacting with its α2- and α3-subunits, but not with the α1-subunit [30]. However, the α1-isoform is the only Na+,K+-ATPase isoform found in endothelial cells [35]. We showed that 1-10 nM ouabain caused ~30% activation of highly purified Na+,K+-ATPase from dog kidneys and duck salt glands (unpublished data), and this enzyme is known to contain α1-subunit only. Based on the analysis of kinetics of 3H-labeled ouabain binding, Ghysel-Burton and Godfraind proposed that activation and inhibition of sodium pump by low and high doses of ouabain, respectively, can be due to the presence of two different ouabain-binding sites within the same α-subunit of Na+,K+-ATPase [29]. However, the results of X-ray analysis disprove the existence of a second binding site in the same subunit [36]. Considering our data on the activation of Na+,K+-ATPase in pig kidney microsomes (Fig. 2b) containing α1-subunit only, as well as numerous evidence on the existence of Na+,K+-ATPase in a form of α2β2-oligomer with functionally different α-subunits [37], we suggest the following. Two α1-subunits of the α2β2-oligomer have ouabain-binding sites with different affinities. Ouabain binding to the high-affinity site in one of the oligomer α-subunits activates the enzyme, while saturation with ouabain of both sites located in different subunits inhibits Na+,K+-ATPase.
Incubation of HUVEC with low ouabain concentrations of for 48-72 h increased cell count by 20-40%, whereas ouabain concentrations over 30 nM decreased HUVEC proliferation (Fig. 3). These results correlate well with the data on the activation and inhibition of cell growth by low and high doses, respectively, of ouabain and other CTS in endothelial cells [33], vascular smooth muscle cells [21, 26], kidney proximal tubule cells [32, 38], several neuronal cell lines [39], and primary kidney cell cultures from renal cysts of patients with autosomal dominant polycystic kidney disease [40]. The correlation between ouabain dose-dependence of [Na+]i/[K+]i ratio and cell growth (Fig. 4) convincingly shows that activation and inhibition of cell proliferation is caused by increase and decrease in the monovalent cation transmembrane gradient due to activation and inhibition, respectively, of the Na+,K+-pump.
Our hypothesis on the key role of Na+,K+-ATPase activation and the corresponding decrease in the [Na+]i/[K+]i ratio in the regulation of cell growth is in good agreement with numerous studies that demonstrated Na+,K+-ATPase activation by proliferation inducers. For example, Na+,K+-ATPase activity increased in canine renal epithelial cell in response to serum-derived growth factors [41] and in murine macrophages treated with hemopoietic growth factors [42]. Na+,K+-ATPase activation and increase in intracellular K+ were observed in proliferating human lymphocytes [43]. Tian and co-workers demonstrated that siRNA-mediated knockdown of Na+,K+-ATPase α1-subunit decreases basal proliferation of LLC-PK1 cells and abolishes the growth-stimulating effect of low ouabain doses [44].
It is well documented that long-term elevation of [Na+]i drastically changes the cell transcriptome via activation of both Ca2+i-mediated and Ca2+i-independent mechanisms of transcription regulation. At the same time, [K+]i decrease suppresses translation at the elongation stage without affecting ribosome assembly from its subunits [8]. Elucidation of the role of these two mechanisms, as well as of other signaling pathways, in the activation and inhibition of cell proliferation by low and high CTS doses, respectively, requires further study.
Acknowledgements
This work was supported by the Canadian Institutes of Health Research, the Russian Foundation for Basic Research (projects Nos. 14-04-31705, 15-04-00101, and 15-04-08832), and by the Russian Science Foundation (projects Nos. 14-15-00006 and 16-15-10026).
REFERENCES
1.Blanco, G., and Mercer, R. W. (1998) Isozymes of
the Na-K-ATPase: heterogeneity in structure, diversity in function,
Am. J. Physiol., 275, F633-F650.
2.Scheiner-Bobis, G. (2002) The sodium pump. Its
molecular properties and mechanisc of ion transport, Eur. J.
Biochem., 269, 2424-2433.
3.Clarke, R. J., and Fan, X. (2011) Pumping ions,
Clin. Exp. Pharmacol. Physiol., 38, 726-733.
4.Taurin, S., Hamet, P., and Orlov, S. N. (2003) Na/K
pump and intracellular monovalent cations: novel mechanism of
excitation–transcription coupling involved in inhibition of
apoptosis, Mol. Biol., 37, 371-381.
5.Orlov, S. N., and Hamet, P. (2006) Intracellular
monovalent ions as second messengers, J. Membr. Biol.,
210, 161-172.
6.Orlov, S. N., Platonova, A. A., Hamet, P., and
Grygorczyk, R. (2013) Cell volume and monovalent ion transporters:
their role in the triggereing and progression of the cell death
machinery, Am. J. Physiol. Cell Physiol., 305,
C361-C372.
7.Koltsova, S. V., Trushina, Y., Haloui, M., Akimova,
O. A., Tremblay, J., Hamet, P., and Orlov, S. N. (2012) Ubiquitous
[Na+]i/[K+]i-sensitive
transcriptome in mammalian cells: evidence for
Ca2+i-independent excitation–transcription
coupling, PLoS One, 7, e38032.
8.Orlov, S. N., and Hamet, P. (2015) Salt and gene
expression: evidence for
Na+i,K+i-mediated signaling
pathways, Eur. J. Physiol., 467, 489-498.
9.Aperia, A. (2007) New roles for an old Na,K-ATPase
emerges as an interesting drug target, J. Intern. Med.,
261, 44-52.
10.Schoner, W., and Scheiner-Bobis, G. (2007)
Endogenous and exogenous cardiac glycosides: their role in
hypertension, salt metabolism, and cell growth, Am. J. Physiol. Cell
Physiol., 293, C509-C536.
11.Liu, J., and Xie, Z. (2010) The sodium pump and
cardiotonic steroids-induced signal transduction protein kinases and
calcium-signaling microdomain in regulation of transporter traficking,
Biochim. Biophys. Acta, 1802, 1237-1245.
12.Segel, G. B., and Lichtman, M. A. (1980) The
apparent discrepancy of ouabain inhibition of cation transport and
lymphocyte proliferation is explained by time-dependecy of ouabain
binding, J. Cell. Physiol., 104, 21-26.
13.Orlov, S. N., Thorin-Trescases, N., Dulin, N. O.,
Dam, T.-V., Fortuno, M. A., Tremblay, J., and Hamet, P. (1999)
Activation of cAMP signaling transiently inhibits apoptosis in vascular
smooth muscle cells in a site upstream of caspase 3, Cell Death
Differ., 6, 661-672.
14.Rathbun, W. B., and Betlach, M. V. (1969)
Estimation of enzymatically produced orthophosphate in the presence of
cysteine and adenosine triphosphate, Anal. Biochem., 28,
436-445.
15.Klimanova, E. A., Petrushenko, I. Y., Mitkevich,
V. A., Anashkina, A. A., Orlov, S. N., Makarov, A. A., and Lopina, O.
D. (2015) Binding of ouabain and marinobufagenin leads to different
structural changes in Na,K-ATPase and depends on the enzyme
conformation, FEBS Lett., 589, 2668-2674.
16.Orlov, S. N., Thorin-Trescases, N., Kotelevtsev,
S. V., Tremblay, J., and Hamet, P. (1999) Inversion of the
intracellular Na+/K+ ratio blocks apoptosis in
vascular smooth muscle at a site upstream of caspase-3, J. Biol.
Chem., 274, 16545-16552.
17.Orlov, S. N., Thorin-Trescases, N., Pchejetski,
D., Taurin, S., Farhat, N., Tremblay, J., Thorin, E., and Hamet, P.
(2004) Na+/K+ pump and endothelial cell survival:
[Na+]i/[K+]i-independent
necrosis triggered by ouabain, and protection against apoptosis
mediated by elevation of [Na+]i, Pflugers
Arch., 448, 335-345.
18.Akimova, O. A., Tverskoi, A. M., Smolyaninova, L.
V., Mongin, A. A., Lopina, O. D., La, J., Dulin, N. O., and Orlov, S.
N. (2015) Critical role of the
α1-Na+,K+-ATPase subunit in insensitivity
of rodent cells to cytotoxic action of ouabain, Apoptosis,
20, 1200-1210.
19.Murata, Y., Matsuda, T., Tamada, K., Hosoi, R.,
Asano, S., Takuma, K., Tanaka, K., and Baba, A. (1996) Ouabain-induced
cell proliferation in cultured rat astrocytes, Jpn. J.
Pharmacol., 72, 347-353.
20.Chueh, S.-C., Guh, J.-H., Chen, J., Lai, M.-K.,
and Teng, C.-M. (2001) Dual effect of ouabain on the regulation of
proliferation and apoptosis in human prostatic smooth muscle cells,
J. Urol., 166, 347-353.
21.Abramowitz, J., Dai, C., Hirschi, K. K.,
Dmitrieva, R. I., Doris, P. A., Liu, L., and Allen, J. C. (2003)
Ouabain- and marinobufagenin-induced proliferation of human umbilical
vein smooth muscle cells and a rat vascular smooth muscle cell line,
A7r5, Circulation, 108, 1049-1054.
22.Li, M., Wang, Q., and Guan, L. (2007) Effects of
ouabain on proliferation, intracellular free calcium and c-myc mRNA
expression in vascular smooth muscle cells, J. Comp. Physiol.,
177, 589-595.
23.Winnicka, K., Bielawski, K., Bielawska, A., and
Miltyk, W. (2010) Dual effects of ouabain, digoxin and proscillaridin A
on the regulation of apoptosis in human fibroblasts, Nat. Prod.
Res., 24, 274-285.
24.Qui, J., Gao, H.-Q., Li, B.-Y., and Shen, L.
(2008) Proteomics investigation of protein expression changes in
ouabain-induced apoptosis in human umblical vein endothelial cells,
J. Cell. Biochem., 104, 1054-1064.
25.Ren, Y. P., Zhang, M. J., Zhang, T., and Huang,
R. W. (2014) Dual effects of ouabain on the regulation of proliferation
and apoptosis in human umbelical vein endothelial cells: involvement of
Na+,K+-ATPase α-subinits and NF-κB,
Int. J. Clin. Exp. Med., 7, 1244-1222.
26.Aydemir-Koksoy, A., Abramowitz, J., and Allen, J.
C. (2001) Ouabain-induced signaling and vascular smooth muscle cell
proliferation, J. Biol. Chem., 276, 46605-46611.
27.Holthouse, K. A., Mandal, A., Merchant, M. L.,
Schelling, J. R., Delamere, N. A., Valdes, R. R., Tyagi, S. C.,
Lederer, E. D., and Khundmiri, S. J. (2010) Ouabain stimulates
Na-K-ATPase through a sodium/hydrogen exchanger-1 (NHE1)-dependent
mechanism in human kidney proximal tubule cells, Am. J. Physiol.
Renal Physiol., 299, F77-F90.
28.Li, J., Zelenin, S., Aperia, A., and Aizman, O.
(2006) Low doses of ouabain protect from serum deprivation-triggered
apoptosis and stimulate kidney cell proliferation via activation of
NF-kappaB, J. Am. Soc. Nephrol., 17, 1848-1857.
29.Ghysel-Burton, J., and Godfraind, T. (1979)
Stimulation and inhibition of the sodium pump by cardiotonic steroids
in relation to their binding sites and ionotropic effect, Br. J.
Pharmacol., 66, 175-184.
30.Gao, J., Wymore, R. S., Wang, Y., Gaudette, G.
R., Krukenkamp, I. B., Cohen, I. S., and Mathias, R. T. (2002)
Isoform-specific stimulation of cardiac Na/K pumps by nanomolar
concentrations of glycosides, J. Gen. Physiol., 119,
297-312.
31.Balzan, S., D’Urso, G., Nicolini, G.,
Forini, F., Pellegrino, M., and Montali, U. (2007) Erythrocyte sodium
pump stimulation by ouabain and an endogenous ouabain-like factor,
Cell Biochem. Funct., 25, 297-303.
32.Khundmiri, S. J., Metzler, M. A., Ameen, M.,
Amin, V., Rane, M. J., and Delamere, N. A. (2006) Ouabain induces cell
proliferation through calcium dependent phosphorylation of Akt (protein
kinase B) in opossum kidney proximal tubule cells, Am. J. Physiol.
Cell Physiol., 291, C1247-1257.
33.Saunders, R., and Scheiner-Bobis, G. (2004)
Ouabain stimulates endothelin release and expression in human
endothelial cells without inhibiting the sodium pump, Eur. J.
Biochem., 271, 1054-1062.
34.Oselkin, M., Tian, D., and Bergold, P. J. (2010)
Low-dose cardiotonic steroids increase sodium-potassium ATPase activity
that protects hippocampal slice cultures from experimental ischemia,
Neurosci. Lett., 473, 67-71.
35.Pierre, S., Compe, E., Grillasca, J. P.,
Plannells, R., Sampol, J., Pressley, T. A., and Maixent, J. M. (2001)
RP-PCR detection of Na,K-ATPase subunit isoforms in human unbilical
vein endothelial cells (HUVEC): evidence for the presence of a1 and b3,
Cell. Mol. Biol., 47, 319-324.
36.Laursen, M., Yatime, L., Nissen, P., and
Fedosova, N. U. (2013) Crystal structure of the high-affinity
Na+,K+-ATPase-ouabain complex with
Mg2+ bound in cation binding site, Proc. Natl. Acad. Sci.
USA, 110, 10958-10963.
37.Askari, A. (1987)
Na+,K+-ATPase: on the number of the ATP sites of
the functional unit, J. Bioenerg. Biomembr., 19,
359-374.
38.Dmitrieva, R. I., and Doris, P. A. (2004) Ouabain
is a potent promoter of growth and activator of ERK1/2 in
ouabain-resistant rat renal epithelial cells, J. Biol. Chem.,
278, 28160-28166.
39.Dvela, M., Rosen, H., Ben-Ami, H. C., and
Lichtstein, D. (2012) Endogenous ouabain regulates cell viability,
Am. J. Physiol. Cell Physiol., 302, C442-C452.
40.Nguyen, A. N., Wallace, D. P., and Blanco, G.
(2007) Ouabain binds with high affinity to the
Na+,K+-ATPase in human polycystic kidney cells
and induces extracellular signal-regulated kinase activation and cell
proliferation, J. Am. Soc. Nephrol., 18, 46-57.
41.Reznik, V. M., Villela, J., and Mendoza, S. A.
(1983) Serum stimulates Na+ entry and the Na-K pump in
quiescent cultures of epithelial cells (MDCK), J. Cell.
Physiol., 117, 211-214.
42.Vairo, G., and Hamilton, J. A. (1988) Activation
and proliferation signals in murine macrophases: stimulation of
Na+,K+-ATPase activity by hemopoietic growth
factors and other agents, J. Cell. Physiol., 134,
13-24.
43.Marakhova, I. I., Vereninov, A. A., Toropova, F.
V., and Vinogradova, T. A. (1998) Na,K-ATPase pump in activated human
lymphocytes: on the mechanisms of rapid and long-term increase in K
influxes during the initiation of phytohemagglutinin-induced
proliferation, Biochim. Biophys. Acta, 1368, 61-72.
44.Tian, J., Li, X., Liang, M., Liu, L., Xie, J. X.,
Ye, Q., Kometiani, P., Tillekeratne, M., Jin, R., and Xie, Z. (2009)
Changes in sodium pump expression dictate the effects of ouabain on
cell growth, J. Biol. Chem., 284, 14921-14929.