Participation of Two Carbonic Anhydrases of the Alpha Family in Photosynthetic Reactions in Arabidopsis thaliana
E. M. Zhurikova, L. K. Ignatova, N. N. Rudenko, V. A. Mudrik, D. V. Vetoshkina, and B. N. Ivanov*
Institute of Basic Biological Problems, Russian Academy of Sciences, 142290 Pushchino, Moscow Region, Russia; E-mail: ivboni@rambler.ru* To whom correspondence should be addressed.
Received July 11, 2016; Revision received July 20, 2016
The expression of genes of two carbonic anhydrases (CA) belonging to the α-family, α-CA2 and α-CA4 (according to the nomenclature in N. Fabre et al. (2007) Plant Cell Environ., 30, 617-629), was studied in arabidopsis (Arabidopsis thaliana, var. Columbia) leaves. The expression of the At2g28210 gene coding α-CA2 decreased under increase in plant illumination, while the expression of the At4g20990 gene coding α-CA4 increased. Under conditions close to optimal for photosynthesis, in plants with gene At2g28210 knockout, the effective quantum yield of photosystem 2 and the light-induced accumulation of hydrogen peroxide in leaves were lower than in wild type plants, while the coefficient of non-photochemical quenching of leaf chlorophyll a fluorescence and the rate of CO2 assimilation in leaves were higher. In plants with At4g20990 gene knockout, the same characteristics changed in opposite ways relative to wild type. Possible mechanisms of the participation of α-CA2 and α-CA4 in photosynthetic reactions are discussed, taking into account that protons can be either consumed or released in the reactions they catalyze.
KEY WORDS: photosynthesis, carbonic anhydrase, arabidopsis, gene expression, mutantsDOI: 10.1134/S0006297916100151
Abbreviations: CA, carbonic anhydrase; Chl, chlorophyll; PAR, photosynthetically active radiation; PETC, photosynthetic electron-transport chain; PS1, photosystem 1; PS2, photosystem 2.
Carbonic anhydrase (CA) catalyzes the interchange of two forms of
inorganic carbon according to the reaction: CO2 +
H2O ↔ HCO3− +
H+. Carbonic anhydrase genes found in living organisms are
divided into several families based on their nucleotide sequences [1]. Nineteen CA genes from three families have been
found in the Arabidopsis thaliana genome [2]: there are eight genes from the α-family, six
genes from the β-family, and five genes from the γ-family.
All CAs from the γ-family are located in mitochondria, where they
are incorporated into protein supercomplex I [3].
β-CAs were found in cell membrane, mitochondria, cytoplasm, and
chloroplasts [2]. β-CA1 (according to the
nomenclature suggested in [2]) is located in
chloroplast stroma, and it is the second most abundant protein in
leaves after Rubisco. Soluble CA, which apparently belongs to the
β-family, was found in the thylakoid lumen [4]. The location of only two α-CAs is known:
α-CA1 is presumably located in chloroplast stroma [5], and α-CA4 is located in thylakoids [6].
The function of CA in the metabolism of algae and cyanobacteria is well-described [7], whereas in higher plants it is not understood as clearly. Current views on the functions of CAs in higher plants are presented in detail in reviews [8, 9]. It should be noted that even the function of β-CA1 is not totally understood, despite the obvious necessity of such enzyme in chloroplast stroma. The main substrate for Rubisco is CO2, but the predominant form of inorganic carbon in stroma is bicarbonate ions, and the fast conversion from HCO3− to CO2 for Rubisco was suggested as the main function of β-CA1 in stroma. However, it was found that manipulating the amount of β-CA1 by changing its gene expression had no effect on the rate of photosynthesis [10, 11]. Still, the very existence of 19 genes encoding CAs in the small genome of arabidopsis suggests their importance in the metabolism of higher plants. It is mostly photosynthesis that requires interchange of the forms of inorganic carbon not only to transport it through membranes, but also to increase the rate of supply or removal of the products of the CA reaction – first of all protons. The presence of several CAs in chloroplasts also supports this idea.
The main purpose of this study was to elucidate the possible roles of two α-CAs in photosynthesis, namely α-CA4 that was found in thylakoids, and α-CA2 whose location was only speculated based on its gene sequence. We compared a variety of properties of A. thaliana (var. Columbia) wild type plants with At2g28210 or At4g20990 knockout mutants, lacking α-CA2 and α-CA4, respectively.
MATERIALS AND METHODS
Wild type plants, two homozygous lines of At4g20990 knockouts (α-CA4 knockouts) 9-12 and 8-8, and two homozygous lines of At2g28210 knockouts (α-CA2 knockouts) 9-11 and 8-3 were grown under 100-120 µmol quanta PAR·m−2·s−1 at 19°C and 450 ppm CO2 concentration. The seeds were kindly supplied by Prof. J. V. Moroney, Louisiana State University, USA. The photoperiod was either 8 or 16 h a day. All experiments were performed 55-65 days after sowing. Measurements were performed on at least six leaves from four crop cycles. Most illustrations show typical results.
RNA was extracted from leaves previously frozen in liquid nitrogen using an Aurum total RNA Mini Kit (BioRad, USA). RNA concentration was measured spectrophotometrically at 260 nm wavelength. Reverse transcription was performed using an iScript Reverse Transcription Supermix Kit (BioRad). Single-stranded cDNA and specific primers for the At4g20990 gene encoding α-CA4 (forward 5′-TCCTCACCAAGCTACTAAATGGAATAAA-3′ and reverse 5′-TTGACGACAGTCCAAATGACGC-3′) and for the At2g28210 gene encoding α-CA2 (forward 5′-GGCGGTTCACGATAATTCAGACACA-3′ and reverse 5′-ACAAAAAAATTAAATAAATAGTACACTGTGGTTGTA-3′) obtained after reverse transcription were used for quantitative PCR (qPCR) with the iQ5 BioRad system and reagents from Evrogen (Russia). qPCR was performed at least three times for each sample. Primers for the actin 7 gene At5g09810 (forward 5′-GAAGGCTGGTTTTGCTGGTGAT-3′ and reverse 5′-CCATGTCATCCCAGTTACTTACAATACC-3′) were used as a positive control.
Chlorophyll (Chl) a fluorescence measurements were performed on intact attached leaves using a leaf-clip connected to a gas chamber with constant CO2 concentration, Mini-PAM fluorometer (Walz, Germany), and Wincontrol-3 software. All plants were kept in darkness for 30 min prior to measurements. After turning on the modulating (measuring) light and triggering saturating flash, the fluorescence levels F0 and Fm were measured. After that, the actinic white light was turned on, and after reaching the stationary level of fluorescence Fs, another saturating flash was triggered to measure Fm′. The effective quantum yield of photosystem 2 (PS2) Y (= (Fm′ − FS)/Fm′) and the rate of non-photochemical quenching qN (= (Fm − Fm′)/(Fm − F0′)) were calculated. Non-photochemical quenching relaxation during the first minute after the end of actinic light (qN1) and between 1st and 15th minutes of darkness (qN15) were calculated as qN1 = (Fm1 − Fm′)/(Fm − F0′) and qN15 = (Fm15 − Fm1)/(Fm − F0′), where Fm1 and Fm15 are the maximum levels of fluorescence at saturating flashes on the 1st and 15th minutes of darkness.
The rate of CO2 assimilation by leaves was measured with a GFS-3000 gas analyzer at 700 ppm CO2 concentration and light intensity of 460 µmol quanta PAR·m−2·s−1 over a period of 7 min.
Hydrogen peroxide content in leaves was measured before and after illumination of the whole plant (500 µmol quanta PAR·m−2·s−1). Leaf samples were frozen in liquid nitrogen and ground in 0.4 ml of 2 M TCA. The homogenate was added to 3 ml of 0.05 M potassium-phosphate buffer with activated charcoal for chlorophyll and carotenoid absorption. After 1 h of incubation, the mixture was centrifuged for 10 min at 10,000g. The pH of the supernatant was normalized to 7.0 with KOH. H2O2 content was measured using a Lum-100 chemiluminometer (DISoft LLC, Russia) by adding 1 ml of peroxidase (5·10−6 M) and luminol (5·10−4 M) mixture to 50 µl of clear supernatant. A standard curve was obtained using 50 µl aliquots of H2O2 solutions of known concentration instead of the supernatant.
PAR intensity was measured with an LI-190SA Quantum Sensor (Li-Cor Biosciences, USA), which is sensitive to light in 400-700 nm range.
RESULTS
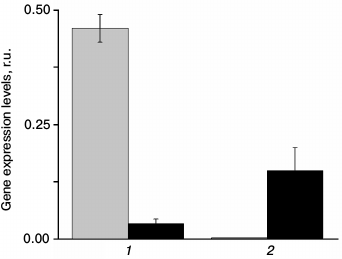
The measurement of At2g28210 and At4g20990 gene expression in wild type plants shows that these genes are indeed expressed, and that their expression level depends on light intensity (Fig. 1). Even though the level of expression does not necessarily correlate with protein concentration, it means that light intensity can affect the amount of these enzymes. Change in light intensity within the range used in this study affects the rate of photosynthesis, suggesting that the changes in the expression of the CA genes can be attributed to the need for fine-tuning some photosynthetic reactions in the altered light conditions. It is also seen from Fig. 1 that the expression of the α-CA2 and α-CA4 genes changes in opposite ways in response to light intensity: under high light, the expression of α-CA2 sharply decreases, whereas the expression of α-CA4 increases.
Fig. 1. Expression levels of genes At2g28210 (1) and At4g20990 (2) encoding α-CA2 and α-CA4, respectively, in leaves of Arabidopsis under different light intensity. Gray bars – light intensity 80 µmol quanta PAR·m−2·s−1; black bars – 5 days after transition to 360 µmol quanta PAR·m−2·s−1.
We found previously [12] that α-CA4 knockouts had larger leaves, larger crown, 10% greater weight, and higher starch content compared to wild type. Electron microscopy showed that starch granules in chloroplasts of α-CA4 knockouts were much larger than in wild type [12]. At the same time, α-CA2 knockouts displayed opposite changes, i.e. their weight and starch content was lower than in the wild type (data not shown).
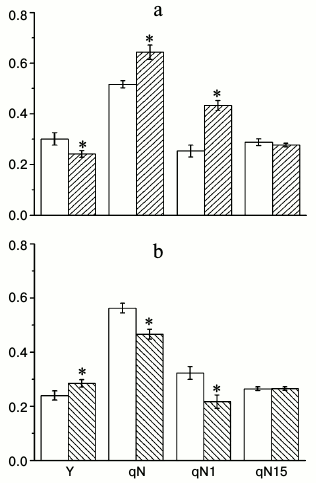
To find conditions in which the differences between wild type and knockouts can be seen, we measured Chl a fluorescence parameters that characterize the properties of the photosynthetic apparatus under different CO2 concentrations (100, 800, and 1500 ppm) and light intensities (70 and 500 µmol quanta PAR·m−2·s−1). Under low CO2 concentration and low light intensity, the difference between the wild type and knockouts was not noticeable (data not shown), probably because the conditions themselves were limiting photosynthesis. Because the results from different homozygous lines showed very similar results, we show the data from only one line for each knockout for simplicity. Figure 2 shows the effective quantum yield (Y), total coefficient of non-photochemical quenching (qN), and its components under 500 µmol quanta PAR·m−2·s−1 light and 800 ppm CO2 concentration, which is close to the optimal levels for photosynthesis. It is seen that Y is higher and qN is lower in α-CA2 knockout compared to wild type, whereas in α-CA4 knockout these properties are changing in the opposite direction. The data about qN changes in α-CA4 knockouts matches our previous assessments of non-photochemical quenching using parameter NPQ [12], significantly supplementing them. Figure 2 reveals that the changes in qN in both mutants are mostly attributed to changes in the qN1 component, which is often described as energy-dependent, appearing because of light-induced pH decrease in the thylakoid lumen. The relaxation of this component happens during the first minute of darkness after illumination. There was no difference in qN15 (relaxation between 1st and 15th minutes of darkness after illumination) between wild type and knockouts. This component was previously attributed to relaxation due to state-transition [13], but now it is considered to be the result of conversion of zeaxanthin into violaxanthin, and sometimes it is even labeled as qZ [14].
Fig. 2. Effects of At2g28210 knockout, lacking α-CA2, line 9-11 (a), and At4g20990 knockout, lacking α-CA4, line 9-12 (b) on effective quantum yield Y under illumination, and coefficients of non-photochemical quenching qN, qN1, and qN15 (see explanation in text). White bars – wild type plants; dashed bars – knockout mutants. Light intensity was 500 µmol quanta PAR·m−2·s−1, CO2 concentration in the air was 800 ppm. The data are presented as mean ± standard error, * p < 0.05.
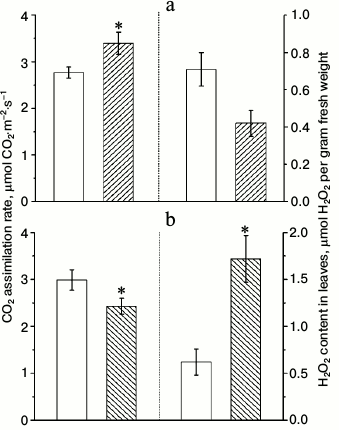
Measurements of CO2 assimilation rates in similar conditions as for Chl a fluorescence measurements yielded unexpected results (Fig. 3). Despite that the effective quantum yield of PS2 in α-CA2 knockouts was lower than in wild type, and in α-CA4 knockouts it was higher than in wild type, the rates of CO2 assimilation changed in opposite direction relative to wild type. This apparent discrepancy may be explained by the fact that effective quantum yield of PS2 reflects the total electron flow through PS2 from water to terminal electron acceptors, whereas CO2 assimilation rate reflects only the flow of electrons to NADP+ and finally CO2. The main alternative electron flow is the flow to oxygen as the terminal acceptor. This flow may account for up to 60% of total electron flow through the PETC [15].
Fig. 3. Effects of At2g28210 knockout, lacking α-CA2, line 9-11 (a), and At4g20990 knockout, lacking α-CA4, line 9-12 (b) on the rate of CO2 assimilation and H2O2 content in leaves. White bars – wild type plants; dashed bars – knockout mutants. Light intensity was 500 µmol quanta PAR·m−2·s−1, CO2 concentration in the air was 700 ppm. The data are presented as mean ± standard error, * p < 0.05.
Figure 3 shows that 5-min illumination of dark-adapted leaves leads to lower accumulation of H2O2 in α-CA2 knockouts and to higher accumulation of H2O2 in α-CA4 knockouts compared to wild type. Since these changes appear because of short-term illumination, they probably indicate that knockout mutants have some changes in PETC functions compared to wild type.
DISCUSSION
The results of this study suggest that functions of two carbonic anhydrases of the α-family, α-CA2 and α-CA4, are strongly connected to photosynthetic reactions. First, the expression of the CA genes is affected by light intensity (Fig. 1). We have shown previously that α-CA4 gene expression increases under high CO2 concentration in the air [12]. The expression of the gene encoding α-CA2, on the contrary, decreases under the same conditions (data not shown). Therefore, changes in two main environmental factors that define the rate of photosynthesis affect the expression of both α-CA2 and α-CA4 genes, but in opposite directions.
The comparison of wild type plants with knockout mutants lacking α-CA2 or α-CA4 revealed the same opposite effect on PETC characteristics – effective quantum yield of PS2 (Y) and non-photochemical quenching of Chl a fluorescence (qN). The maximum quantum yield of PS2 in dark-adapted leaves of both knockouts did not differ from wild type (data not shown). Together with the changes in Y in the light, this means that the lack of CAs does not change the structure of PS2, but it suggests that they are involved in PETC functioning under illumination.
The changes in the effective quantum yield of PS2 in knockouts, which can be multiplied by light intensity to get the rate of electron flow through PETC, gives no basis for speculation about functions of CAs in chloroplasts because the value of Y depends on both the rate of influx and the rate of efflux of electrons from PS2. On the other hand, the molecular mechanisms of non-photochemical quenching of Chl a fluorescence and the factors that can affect it are now well described ([16] and references therein). The increase in proton concentration in the lumen under illumination leads to increase in non-photochemical quenching in two ways: (i) as a result of conformational changes in light-harvesting antenna because of protonation of PsbS protein; (ii) as a result of zeaxanthin accumulation when violaxanthin deepoxidase is activated. The zeaxanthin epoxidation process in darkness is slow, and it takes 10-60 min in different plants [14]. The slow component of the relaxation of non-photochemical quenching (qN15) was nearly the same in wild type and in the knockouts. The increase of qN in α-CA2 knockout and the decrease in α-CA4 knockout were in accordance with the changes in the qN1 component (Fig. 2), which reflects fast relaxation of non-photochemical quenching due to efflux of protons from the lumen after turning off the light. This suggests that it is protonation of PsbS protein, and its influence on total non-photochemical quenching, that increases or decreases in these knockouts. Taking into account that a proton is one of the products of the CA reaction, localization of CA near PsbS or near PS2 can provide controlled protonation and deprotonation of PsbS.
Two sources of CA activity were found in PS2-enriched thylakoid membrane fragments from higher plants [17-20]; however, the genes encoding CAs located near PS2 were not determined yet. Carbonic anhydrase Cah 3 found in alga Chlamydomonas reinhardtii near the donor side of PS2 is believed to stabilize the Mn cluster and support the active state of the water-oxidizing complex by removing the excess of protons, preventing photoinhibition of PS2 [21, 22]. It was shown that bicarbonate may have a role as mobile proton carrier in PS2 of higher plants, and it is assumed that CA may be involved in this process [23].
Taking into account the literature data, the data obtained in this study indicates that α-CA2 and α-CA4 take part, competitively, in proton exchange near PS2, thus regulating the level of PsbS protonation, and as a result, the non-photochemical quenching of Chl a fluorescence, which reflects the level of excess energy dissipation. The decrease in this quenching without α-CA4 means that this CA provides protons for PsbS, which fits the fact that its expression increases under high light (Fig. 1) when the defense against photoinhibition by means of dissipation of excess energy is the most important. α-CA2 has an opposite role providing fast deprotonation of PsbS under low light to prevent energy dissipation when the supply of light energy becomes low; this is also in accordance with the data about high expression of its gene under low light (Fig. 1).
To explain opposite actions of the studied CAs, as a working hypothesis we assume that they have different orientation of their reaction centers relative to the lumenal side of the thylakoid membrane. The direction of the reaction center towards the lumen in α-CA2 gives preference to catalysis of the reaction between H+ and HCO3− diffusing from stroma into lumen along the concentration gradient. On the other hand, contact of the reaction center of α-CA4 with thylakoid membrane will make it consume CO2 accumulated inside the membrane (1 µmol/mg Chl) [24], and release H+.
The changes in the effective quantum yield (Y) in both mutants compared to wild type correlate with the changes in non-photochemical quenching: in α-CA2 knockout, Y is lower because of the larger rate of light energy dissipation, whereas in α-CA4 the situation is the opposite. At the same time, changes in the rate of CO2 fixation were opposite to changes in Y (Fig. 2 vs. Fig. 3). Measurements of H2O2 accumulation in wild type and knockouts showed that both α-CA2 and α-CA4 knockouts have altered electron flow to oxygen (Fig. 3). Without α-CA2, the fraction of electron flow to O2 (H2O2) decreased and the total electron flow through PETC decreased, whereas without α-CA4 both these increased. The contribution of electron flow to O2 in total electron flow can be quite high in some plants [15], but data about its share in arabidopsis is lacking. In plants that normally grow under low light, like arabidopsis, the fraction of electron flow to O2 is usually higher than in plants that grow under high light [25]. Noticeable production of H2O2 in chloroplasts from arabidopsis leaves [26] may indicate a substantial electron flow to O2 through the PETC in this species.
Since both α-CA2 and α-CA4 have an impact on proton concentration in the lumen, opposite effects on electron flow towards NADP+ (CO2) and O2 (H2O2) when one or the other CA is missing may be attributed to the changes in Calvin cycle turnover due to ATP availability. The decrease in proton concentration decreases the rate of ATP synthesis and, accordingly Calvin cycle turnover, increasing the electron flow to O2 due to shortage of NADP+; and vice versa, the increase in ATP synthesis increases Calvin cycle turnover, i.e. regeneration of NADP+, that in turn decreases the electron flow to O2.
Acknowledgements
Authors express their gratitude to Dr. V. S. Sukhov from Lobachevsky State University of Nizhni Novgorod for assistance in CO2 assimilation rate measurements and to T. P. Fedorchuk from the Institute of Basic Biological Problems, Russian Academy of Sciences, for helpful discussion.
This study was conducted with financial support by the Russian Foundation for Basic Research (project No. 15-04-03883).
REFERENCES
1.Hewett-Emmett, D., and Tashian, R. E. (1996)
Functional diversity, conservation, and convergence in the evolution of
the α-, β-, and γ-carbonic anhydrase gene families,
Mol. Phylogenet. Evol., 5, 50-77.
2.Fabre, N., Reiter, I. M., Becuwe-Linka, N., Genty,
B., and Rumeau, D. (2007) Characterization and expression analysis of
genes encoding α and β carbonic anhydrases in
Arabidopsis, Plant Cell Environ., 30, 617-629.
3.Sunderhaus, S., Dudkina, N. V., Jansch, L.,
Klodmann, J., Heinemeyer, J., Perales, M., Zabaleta, E., Boekema, E.
J., and Braun, H.-P. (2006) Carbonic anhydrase subunits form a
matrix-exposed domain attached to the membrane arm of mitochondrial
complex I in plants, J. Biol. Chem., 281, 6482-6488.
4.Fedorchuk, T., Rudenko, N., Ignatova, L., and
Ivanov, B. (2014) The presence of soluble carbonic anhydrase in the
thylakoid lumen of chloroplasts from Arabidopsis leaves, J.
Plant Physiol., 171, 903-906.
5.Villarejo, A., Buren, S., Larsson, S., Dejardin,
A., Monne, M., Rudhe, Ch., Karlsson, J., Jansson, S., Lerouge, P.,
Rolland, N., von Heijne, G., Grebe, M., Bako, L., and Samuelsson, G.
(2005) Evidence for a protein transported through the secretory pathway
en route to the higher plant chloroplast, Nat. Cell
Biol., 7, 1224-1231.
6.Friso, G., Giacomelli, L., Ytterberg, A. J.,
Peltier, J.-B., Rudella, A., Sun, Q., and Van Wijka, K. J. (2004)
In-depth analysis of the thylakoid membrane proteome of Arabidopsis
thaliana chloroplasts: new proteins, new functions, and a plastid
proteome database, Plant Cell, 16, 478-499.
7.Moroney, J. V., Ma, Y., Frey, W. D., Fusilier, K.
A., Pham, T. T., Simms, T. A., DiMario, R. J., Jing, J., and Mukherjee,
B. (2011) The carbonic anhydrase isoforms of Chlamydomonas
reinhardtii: intracellular location, expression, and physiological
roles, Photosynth. Res., 109, 133-149.
8.Ivanov, B. N., Ignatova, L. K., and Romanova, A. K.
(2007) Diversity in forms and functions of carbonic anhydrase in
terrestrial higher plants, Russ. J. Plant Physiol., 54,
143-162.
9.Rudenko, N. N., Ignatova, L. K., Fedorchuk, T. P.,
and Ivanov, B. N. (2015) Carbonic anhydrases in photosynthetic cells of
higher plants, Biochemistry (Moscow), 80, 674-687.
10.Majeau, N., Arnoldo, M. A., and Coleman, J. R.
(1994) Modification of carbonic anhydrase activity by antisense and
over-expression constructs in transgenic tobacco, Plant Mol.
Biol., 25, 377-385.
11.Price, G. D., Von Caemmerer, S., Evans, J. R.,
Yu, J.-W., Lloyd, J., Oja, V., Kell, P., Harrison, K., Gallagher, A.,
and Badger, M. R. (1994) Specific reduction of chloroplast carbonic
anhydrase activity by antisense RNA in transgenic tobacco plants has a
minor effect on photosynthetic CO2 assimilation,
Planta, 193, 331-340.
12.Zhurikova, E. M., Ignatova, L. K., Semenova, G.
A., Rudenko, N. N., Mudrik, V. A., and Ivanov, B. N. (2015) Effect of
knockout of α-carbonic anhydrase 4 gene on photosynthetic
characteristics and starch accumulation in leaves of Arabidopsis
thaliana, Russ. J. Plant Physiol., 62, 564-569.
13.Quick, W. P., and Stitt, M. (1989) An examination
of factors contributing to non-photochemical quenching of chlorophyll
fluorescence in barley leaves, Biochim. Biophys. Acta,
977, 287-296.
14.Nilkens, M., Kress, E., Lambrev, P., Miloslavina,
Y., Muller, M., Holzwarth, A. R., and Jahns, P. (2010) Identification
of a slowly inducible zeaxanthin-dependent component of
non-photochemical quenching of chlorophyll fluorescence generated under
steady-state conditions in Arabidopsis, Biochim. Biophys.
Acta, 1797, 466-475.
15.Ivanov, B. N., Khorobrykh, S. A., Kozuleva, M.
A., and Borisova-Mubarakshina, M. M. (2014) The role of oxygen and its
reactive forms in photosynthesis, in Contemporary Problems of
Photosynthesis (Allakhverdiev, S. I., Rubin, A. B., and Shauvalov,
V. A., eds.) Izhevsk Institute of Computer Science, Moscow-Izhevsk, pp.
407-460.
16.Ruban, A. V. (2016) Nonphotochemical chlorophyll
fluorescence quenching: mechanism and effectiveness in protecting
plants from photodamage, Plant Physiol., 170,
1903-1916.
17.Lu, Y. K., and Stemler, A. J. (2002) Extrinsic
photosystem II carbonic anhydrase in maize mesophyll chloroplasts,
Plant Physiol., 128, 643-649.
18.Khristin, M. S., Ignatova, L. K., Rudenko, N. N.,
Ivanov, B. N., and Klimov, V. V. (2004) Photosystem II associated
carbonic anhydrase activity in higher plants is situated in core
complex, FEBS Lett., 577, 305-308.
19.Ignatova, L. K., Rudenko, N. N., Khristin, M. S.,
and Ivanov, B. N. (2006) Heterogeneous nature of carbonic anhydrase
activity in thylakoid membranes, Biochemistry (Moscow),
71, 525-632.
20.Rudenko, N. N., Ignatova, L. K., and Ivanov, B.
N. (2007) Multiple sources of carbonic anhydrase activity in pea
thylakoids: soluble and membrane bound forms, Photosynth. Res.,
91, 81-89.
21.Villarejo, A., Shutova, T., Moskvin, O., Forssen,
M., Klimov, V. V., and Samuelsson, G. (2002) A photosystem
II-associated carbonic anhydrase regulates the efficiency of
photosynthetic oxygen evolution, EMBO J., 21,
1930-1938.
22.Shutova, T., Kenneweg, H., Buchta, J., Nikitina,
J., Terentyev, V., Chernyshov, S., Andersson, B., Allakhverdiev, S. I.,
Klimov, V. V., Dau, H., Junge, W., and Samuelsson, G. (2008) The
photosystem II-associated Cah3 in Chlamydomonas enhances the
O2 evolution rate by proton removal, EMBO J.,
27, 782-791.
23.Koroidov, S., Shevela, D., Shutova, T.,
Samuelsson, G., and Messinger, J. (2014) Mobile hydrogen carbonate acts
as proton acceptor in photosynthetic water oxidation, Proc. Natl.
Acad. Sci. USA, 111, 6299-6304.
24.Stemler, A. (1977) The binding of bicarbonate to
washed chloroplast grana, Biochim. Biophys. Acta, 460,
511-522.
25.Shmeleva, V. L., Ivanov, B. N., and Red’ko,
T. P. (1982) Electron transport and photophosphorylation, coupled with
photoreduction of oxygen by chloroplasts of peas, grown under different
conditions of illumination, Biochemistry (Moscow), 47,
1104-1107.
26.Naydov, I. A., Mubarakshina, M. M., and Ivanov,
B. N. (2012) Formation kinetics and H2O2
distribution in chloroplasts and protoplasts of photosynthetic leaf
cells of higher plants under illumination, Biochemistry
(Moscow), 77, 143-151.