REVIEW: Neurotrophic Factors (BDNF and GDNF) and the Serotonergic System of the Brain
N. K. Popova*, T. V. Ilchibaeva, and V. S. Naumenko
Federal Research Center Institute of Cytology and Genetics, Siberian Branch of the Russian Academy of Sciences, 630090 Novosibirsk, Russia; E-mail: npopova@bionet.nsc.ru, rbicehok@mail.ru, naumenko2002@mail.ru* To whom correspondence should be addressed.
Received October 13, 2016; Revision received November 16, 2016
Neurotrophic factors play a key role in development, differentiation, synaptogenesis, and survival of neurons in the brain as well as in the process of their adaptation to external influences. The serotonergic (5-HT) system is another major factor in the development and neuroplasticity of the brain. In the present review, the results of our own research as well as data provided in the corresponding literature on the interaction of brain-derived neurotrophic factor (BDNF) and glial cell line-derived neurotrophic factor (GDNF) with the 5-HT-system of the brain are considered. Attention is given to comparison of BDNF and GDNF, the latter belonging to a different family of neurotrophic factors and being mainly considered as a dopaminergic system controller. Data cited in this review show that: (i) BDNF and GDNF interact with the 5-HT-system of the brain through feedback mechanisms engaged in autoregulation of the complex involving 5-HT-system and neurotrophic factors; (ii) GDNF, as well as BDNF, stimulates the growth of 5-HT neurons and affects the expression of key genes of the brain 5-HT-system – those coding tryptophan hydroxylase-2 and 5-HT1A and 5-HT2A receptors. In turn, 5-HT affects the expression of genes that control BDNF and GDNF in brain structures; (iii) the difference between BDNF and GDNF is manifested in different levels and relative distribution of expression of these factors in brain structures (BDNF expression is highest in hippocampus and cortex, GDNF expression in the striatum), in varying reaction of 5-HT2A receptors on BDNF and GDNF administration, and in different effects on certain types of behavior.
KEY WORDS: neurotrophic factors, serotonergic system, BDNF, GDNF, interaction between 5-HT-system and neurotrophic factorsDOI: 10.1134/S0006297917030099
Abbreviations: BDNF, brain-derived neurotrophic factor; DA, dopamine; GDNF, glial cell line-derived neurotrophic factor; GFRα1–4, glycosylphosphatidylinositol (GPI)-linked cell surface receptors; 5-HT, serotonin or 5-hydroxytryptamine; 5-HT-system, serotonergic system of the brain; SERT, serotonin transporter; TGFβ, transforming growth factor β; TPH-2, tryptophan hydroxylase-2; TrkB, tropomyosin-related kinase B receptor; UTR, untranslated region.
Neurotrophic factors, a group of large polypeptides (up to 200 amino
acids) organized in single- and double-stranded forms, play a key role
in developing and maintaining structures of both the central and the
peripheral nervous systems. They are involved in regulation of growth,
development, differentiation, and survival of cell populations as well
as their adaptation to environmental influences [1-4].
At least eight families of neurotrophic factors are now differentiated, although different authors provide varying classifications [5, 6].
The first neurotrophic factor, nerve growth factor (NGF), was discovered in the early 1950s [7], and the brain-derived neurotrophic factor (BDNF) was found 30 years later [8]. The discovery of BDNF caused keen interest and attracted attention to all neurotrophic factors. Interest in BDNF was linked to, as it soon became clear, its remarkable property of stimulating growth of neurons, axons and dendrites, synapse formation, and other processes of neuroplasticity not only in early ontogeny, but also in the brain of adult organisms [9, 10], which was previously thought to be impossible. Now BDNF is one of the best-studied neurotrophic factors of the central nervous system.
Neurotrophic factors from different families share common characteristics, but special attention is drawn to those that affect the functioning of the neurotransmitter systems of the brain. BDNF is closely linked with the serotonergic (5-HT) system of the brain, and the glial cell line-derived neurotrophic factor (GDNF) demonstrates a marked protective effect on the nigrostriatal and mesolimbic dopamine (DA) system of the brain and is considered dopaminergic [11].
The question on which we concentrate the attention of this review is how mediator-specific are the properties of the neurotrophic factors of different families. This question is primarily addressed to the 5-HT-system, which is evolutionarily the most ancient and expansive neurotransmitter system. A great number of studies have established the participation of 5-HT in the regulation of various forms of behavior: sleep and wakefulness, aggressive behavior, sexual motivation [12], and neuroendocrine regulation, including the regulation of the hypothalamo–pituitary–adrenal system [13] being the main system responding to stress. 5-HT is involved in the mechanism of action of all currently used groups of antidepressants (inhibitors of the 5-HT reuptake, tricyclic antidepressants, monoamine oxidase inhibitors), and depression and suicide are associated with insufficiency of the functional activity of the 5-HT-system [14, 15].
5-HT and BDNF are the main players in the mechanisms of neurogenesis and neuroplasticity [4]. There is much less data on the relation between 5-HT and GDNF. The two neurotrophic factors belong to different families: BDNF to the family of neurotrophins, GDNF to the family of transforming growth factors β (TGFβ). In this review, we analyze and compare data on features of these neurotrophic factors and their interaction with the 5-HT-system of the brain.
BRAIN-DERIVED NEUROTROPHIC FACTOR (BDNF)
BDNF is the dominant factor (compared to other neurotrophic factors) in the brain. This holds true not only to the variety of brain structures, but also to the BDNF expression level. According to our data [16, 17], BDNF expression in the brain structures of rats is much higher than that of GDNF. According to current estimations, the human BDNF gene is located in the p14 region of chromosome 11 (in rats and mice these are chromosomes 3q33 and 2qE3, respectively) and contains 12 exons, nine of which have specific promoters (I-VIII 5′ exons spliced to the common 3′ IX exon). This gene structure is observed both in humans [18] and in rodents [19], but the number of exons varies (9 in mice and 10 in rats). Transcripts of template RNA as well as BDNF protein are widely present in the neocortex, hippocampus, amygdala, and cerebellum [20].
BDNF is notable by structural and functional complexity, which is based on (i) the presence of several promoters in the encoding gene; (ii) expression of a multitude of transcripts subjected to alternative splicing or having various patterns of polyadenylation; (iii) several isoforms of the precursor, but only one form of a mature molecule; (iv) existence of two different receptors (TrkB and p75), whose activation causes opposite effects. All these features determine the complexity of the molecular mechanism regulating BDNF production and functional activity [21].
The complex regulation of BDNF transcription involves epigenetic factors. Promoters I, II, and IV of the human BDNF gene and I, II, IV-VI, and IX of the BDNF rodent gene are saturated by CpG islands (CG-rich regions of DNA) that makes them targets for methylation/demethylation processes [21]. There is numerous evidence for the participation of Bdnf gene methylation in the regulation of normal neuron activity and in pathological processes [22]. Histone deacetylation significantly contributes to the epigenetic control of BDNF expression. Histones H3 and H4 in promoters I and IV are most frequently subjected to modifications. Such phenomenon can be critical for the implementation of neuroplastic processes and effects of antidepressants and mood stabilizers [22]. It was recently found that Bdnf expression can also be regulated by anti-bdnf transcripts (miRNAs) [21].
BDNF transcription in neurons is positively regulated by membrane depolarization induced by sensory stimuli, as well as NMDA activation of glutamate receptors. The presence of multiple promoters and alternative splicing gives rise to at least 17 transcripts (in humans) with different 5′- and 3′-untranslated regions (UTR). However, they all have a common coding region that includes exon IX, which in turn contains the complete sequence of a precursor molecule – proBDNF. Furthermore, this exon contains two polyadenylation sites producing transcripts with long or short 3′-UTR [18]. This is of great functional significance, since mRNA with the long 3′-UTR is predominantly located in dendritic spines [23], and it is transmitted in response to the activation of neurons. On the contrary, mRNA with the short 3′-UTR is actively transmitted into neuronal bodies to maintain the basal level of BDNF protein [24].
BDNF is made “on demand” in response to neuronal activity from the protein precursor pre-proBDNF, which is cleaved to proBDNF in the Golgi apparatus [20]. In the brain, there are three possible outcomes for proBDNF: (i) secrete and function in the form of proBDNF; (ii) be subjected to editing in the Golgi complex and secreted as a mature BDNF molecule; (iii) secreted in the form of proBDNF and be converted to BDNF in the synaptic space [25].
Effects of mature BDNF are realized through activation of two types of receptors – tropomyosin-related kinase B receptor (TrkB) and the nonspecific p75 receptor. However, expression of the TrkB receptor is much higher, TrkB/p75 ratio in most brain structures being 8-10 [26]. This determines the prevailing role of the TrkB receptor that initiates a cascade of phosphorylation, which in turn leads to protein synthesis, axonal growth, and maturation of dendrites increasing synaptic plasticity [20]. In contrast to BDNF, proBDNF binds to the p75 receptor common for neurotrophins and inhibits neurite growth, reduces the size of neurons, and triggers apoptosis [27-29].
The ratio of the mature BDNF form and its predecessor proBDNF is of interest. Opposing functions of BDNF and its proBDNF predecessor give reason to consider the ratio of BDNF/proBDNF (some researchers use proBDNF/BDNF, which with the significant predominance of BDNF in the brain is less convenient) to be the most important autoregulatory mechanism of synaptic plasticity, and its decrease caused by the increase in predecessor or a decrease in the BDNF level to be one of the pathogenetic factors of neuropathologies and psychopathologies. Detailed information on BDNF and proBDNF functions and analysis of their role in the mechanisms of synaptic plasticity are presented in a review of Borodinova and Salozhin [29]. However, the use of the ratio as an informatively important indicator of brain neuroplasticity is complicated by the fact that changes in the expression of BDNF as well as proBDNF are structure-specific, and in different brain structures may change in the opposite way. Thus, we found marked differences between genetically predisposed to high levels of aggression rats and “tame” rats in BDNF expression, proBDNF, and BDNF/proBDNF ratio. However, in this case in the frontal cortex of aggressive rats the proBDNF level is lower compared with non-aggressive rats, but higher in the hippocampus. Accordingly, the ratio of BDNF/proBDNF in the frontal cortex of highly aggressive rats is high and in the hippocampus, it is low [16].
BDNF expression is sensitive to such influences as stress, trauma, hypoglycemia, ischemia, and brain damage. Many pharmacological agents targeted at various neurotransmitter systems are also modulators of BDNF expression [30]. It is believed that violation of genetic and epigenetic control of metabolism, transport, or transfer of BDNF signal contributes to the development of many neurological and psychiatric disorders, including Alzheimer’s disease [31-33], Huntington’s disease [34-36], Parkinson’s disease [37], neuropathic pain [38, 39], schizophrenia [40, 41], severe depressive disorders [42, 43], and addiction [44, 45].
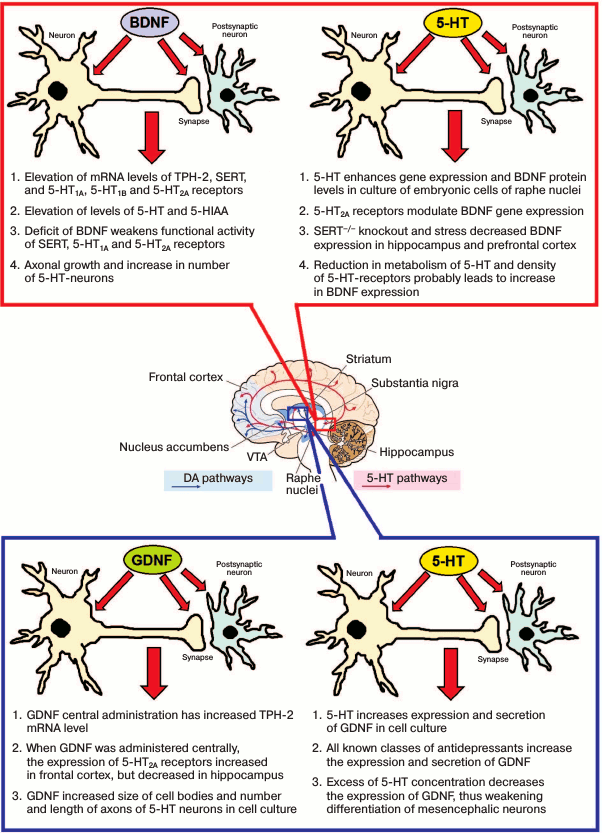
BDNF and the 5-HT brain system. The close relationship of two main factors (BDNF and 5-HT) in the development and neuroplasticity of the brain has been shown in numerous studies, and it is practically assured. Distinct effect of BDNF on the 5-HT-system has been identified in experiments not only in cell cultures, but also in vivo. On the raphe nuclei cell culture of a 14-day-old rat embryo, it has been found that 18-h exposure to BDNF was sufficient to nearly double the number of 5-HT neurons and axonal growth [46]. This striking effect was combined with an increase in the expression of the genes encoding the 5-HT transporter (SERT) and 5-HT1A and 5-HT1B receptors, and it was carried through TrkB BDNF receptors being prevented by tyrosine kinase blocker – genistein.
The effect of BDNF on the brain 5-HT-system is confirmed in experiments in vivo. Chronic administration of BDNF locally into the main cluster of cell bodies of 5-HT neurons – dorsal raphe nuclei – altered the electrophysiological activity of 5-HT neurons [47]. BDNF administered into the midbrain or intraventricularly increased the level of 5-HT and its major metabolite 5-hydroxyindole acetic acid in all five studied brain structures [48]. The same authors noticed increased level of DA, but it was local and limited to the striatum. BDNF demonstrated protective effect when 5-HT neurons were damaged by neurotoxin, primarily by increasing the number of 5-HT axons [49]. Significant long-lasting increase in expression of a key gene of 5-HT synthesis in brain, tryptophan hydroxylase-2 (tph-2), and the genes encoding 5-HT1A and 5-HT2A receptors was observed after a single central injection of BDNF [50]. These changes were observed in predisposed to depression mice of the ASC (Antidepressants Sensitive Cataleptics) line, but not in related mice of the “non-depressive” CBA line. Such changes suggest a significant role of the genotype in BDNF effects.
Unusually long duration of drug action is characteristic of BDNF (as well as of GDNF). In earlier-cited studies, the decrease of the 5-HT-system genes expression was found 21 days after a single injection of BDNF into the lateral ventricle of the brain. We previously established the preservation of BDNF positive effect (restoration of reduced prepulse-inhibition of the startle reflex in DBA/2 mice) 1.5 months after its single central administration [51]. This unique feature of the effect of neurotrophic factors supports the idea of morphological changes in the synaptic connections and neurogenesis that they cause.
Evidence for the effect of BDNF on the 5-HT-system of the brain was also obtained in animals with genetic knockout of bdnf. Since both mice and rats with complete knockout of the BDNF gene (BDNF–/–) are not viable, experiments were conducted on BDNF+/– heterozygotes. Significant changes in the level of 5-HT in brain structures of young mice were not reported, but two-fold decreased expression of BDNF led to significant abnormalities in the 5-HT brain system, which were manifested in decreased sensitivity to a 5-HT reuptake inhibitor, early extinction of its functional activity, and increase in aggressiveness. It was concluded that endogenous BDNF is crucial for normal development and functioning of the 5-HT-system of the brain [52]. This conclusion was confirmed by other researchers. Reduced BDNF level in BDNF+/– mice leads to a decrease in SERT and 5-HT1A receptor functional activity in the hippocampus and expressed deficiency of 5-HT2A receptors in the prefrontal cortex and dorsal raphe nuclei of the midbrain [4].
A special role in the mechanism of BDNF action on the 5-HT-system is played by 5-HT2A receptors. This type of 5-HT receptor is involved in the mechanism of action of antipsychotic drugs and hallucinogens. A 7-day exposure to BDNF on hippocampal cell culture lowered the level of 5-HT2A receptor, but not that of 5-HT1A receptor. At the same time, the level of 5-HT2A receptors increased in heterozygous BDNF+/– mice [53].
Conditional bdnf knockout allowed “turning off” the BDNF gene after birth; inhibiting BDNF expression during the two weeks of postnatal development demonstrated that BDNF deficiency in the postnatal period leads to increased aggressiveness [54] and to disruption of expression and density of 5-HT2A receptors as well as 5-HT2A receptor-mediated neurotransmission [55, 56].
In turn, numerous data indicate that not only BDNF has impact on the 5-HT-system of the brain, but 5-HT is also implicated into regulation of BDNF. The main approaches used to determine the effect of 5-HT on BDNF were the following: (i) pharmacological analysis applying selective agonists and antagonists of 5-HT receptors, or 5-HT itself, and (ii) genetic models with modified characteristics of 5-HT-system knockouts in which one of the key elements of the 5-HT-system (usually SERT) is completely or partially turned off.
It was found that 5-HT increases gene expression and BDNF protein levels in cultures of embryonic cells of raphe nuclei [57]. Pharmacological analysis using agonists and antagonists of 5-HT2A and 5-HT2C receptors indicated the modulating participation of 5-HT2A receptors in the regulation of Bdnf gene expression [58]. It is noteworthy that an agonist of 5-HT2A receptors in different ways affected BDNF mRNA in different brain structures: the mRNA level of this gene decreased in the hippocampus, but increased in the neocortex.
Participation of 5-HT in the regulation of BDNF was also demonstrated in mice and rats with knockout of the gene encoding the SERT. It was found that the absence of SERT in knockout animals affected not only the 5-HT level and metabolism in the brain, but also BDNF. Expression of BDNF in the hippocampus and the prefrontal cortex of SERT–/– rats is lowered [59]. Already in early ontogeny, mutant SERT–/– rats were different from control animals by decreased BDNF expression and reduced level of its transcription factor [60]. Later, it was found out that chronic stress caused by maternal separation of pups had similar effect on BDNF. SERT–/– knockout and stress decreased expression of BDNF in the ventral hippocampus and the ventromedial prefrontal cortex, although stress caused increased BDNF expression in the dorsal hippocampus and dorsomedial cortex of SERT+/– heterozygous rats [61].
Interestingly, deviations in 5-HT-system functioning were elicited in 5-week-old and 5-month-old highly aggressive mice of the ABH line. They manifested themselves through reduction of 5-HT metabolism and imbalance in the density of 5-HT1A and 5-HT2A receptors [62], while the level of BDNF protein in the hippocampus, cortex, and striatum of ABH mice was much higher than that of non-aggressive mice of the ABG line [63]. It is difficult to say what is primary and what is secondary. However, the role of brain 5-HT as an important regulator of aggressive behavior is of no doubt [64]. Moreover, selection based on aggressive behavior is associated with profound changes in the 5-HT-system of the brain [65, 66]. All of this suggests that lowering 5-HT metabolism and changes in the density of 5-HT1A receptors increased the level of BDNF.
GLIAL CELL LINE-DERIVED NEUROTROPHIC FACTOR (GDNF)
GDNF was originally isolated from glioma cell culture, and it was predominantly found in astrocytes, being the major producer of cells. It should be noted that significant importance is recently attributed to astrocyte pathology in causing degenerative processes in the central nervous system [67, 68]. The trophic effect of GDNF on cultures of DA neurons [11] was immediately shown, and currently GDNF is recognized as a necessary factor for the development, maintenance, and protection of the nigrostriatal DA neurons, being a potential factor that protects and restores DA neurons affected in Parkinson’s disease [69, 70]. Alongside three other structurally related factors – neurturin, artemin, and persephin – GDNF forms a family of neurotrophic factors, being a part of the TGFβ superfamily [71]. Members of the GDNF family transmit signals through extracellular receptors (GFRα1–4), each of which is selective for the respective family member. GDNF displays highest affinity for GFRα1. The receptor complex GDNF–GFRα1 binds to the extracellular domain of the receptor tyrosine kinase modulating various intracellular signaling cascades [72]. In addition, GDNF may communicate directly with neural cellular adhesion molecules (NCAM) with subsequent activation of Src-like kinases and MAP kinases.
GDNF has a biologically active pro-form (proGDNF), which is expressed in most parts of the brain and is found in astrocytes as well as in DA neurons [73]. Besides GDNF, peptides formed by proGDNF cleavage, known as DNSP-11 (in humans) and BEP (in rats), also have biological activity. BEP enhances synaptic excitation in the hippocampal pyramidal neurons [74], and DNSP-11 as effectively protects DA neurons as the mature GDNF form [75].
The distribution of GDNF and its receptor is not restricted to the area of midbrain DA neurons. GDNF receptors, as well as its transcripts and protein, were detected in many structures of the brain, indicating that GDNF may have multiple functions [76]. Among them is participation in synaptogenesis in the hippocampus, where GDNF and GFRα1 play an instructive role in synapse formation to induce ectopic presynaptic sites [77]. It is worth mentioning that GDNF improves spatial learning in ASC mice predisposed to depressive-like behavior [78]. It was found two weeks after a single administration of GDNF into the lateral ventricle of the brain and may be related to synaptic remodeling that occurs under the control of GDNF. Several studies have reported that GDNF/GFRα1 signaling may be important in the development and functioning of the various types of GABAergic neurons in the mammalian brain [70]. GDNF is involved in maintaining cell elements in the blood–brain barrier [79-81]. ProGDNF increased expression after the administration of bacterial lipopolysaccharide [73], and GDNF increased formation by astrocytes and microglial cells observed in inflammation indicate that GDNF is an activator of microglia and an inhibitor of neuronal inflammation [76, 82].
Due to the variety of its functions, GDNF, on one hand, is involved in many physiological processes, and, on the other hand, in the pathogenesis of a variety of neurological and psychiatric disorders. GDNF is known for being involved in the mechanisms of aging [73], Parkinson’s, Alzheimer’s, and Huntington’s diseases, amyotrophic lateral sclerosis [83], epilepsy [70], and several neuropsychiatric disorders such as bipolar disorder [43] and unipolar depression [84].
Various factors affect GDNF expression. One is chronic stress. It is well known that prolonged exposure to stress is a risk factor for the development of mental disorders, including depression. Rats exposed to chronic unpredictable stress exhibit depressive-like behavior, and at the same time demonstrate a significant decrease in GDNF expression in the hippocampus [85]. Effects of chronic but very mild stress on mice of the stress-sensitive BALB/c line include significantly reduced GDNF expression in the hippocampus and striatum [86]. “Stress hormones” glucocorticoids can suppress GDNF expression and secretion [87-89], causing epigenetic effects in a variety of ways [90]. Thus, chronic but very mild stress increases DNA methylation linked with the modification of histones resulting in Gdnf transcription repression and formation of a mouse phenotype more susceptible to depression [86]. MicroRNA can change the neuronal response to GDNF through suppression of GFRα1a receptor (an isoform-specific form of the GFRA1 gene) as found in the basolateral amygdala of subjects suffering from depression [91]. A low-calorie diet, exercise, and environmental enrichment are also inducers of GDNF expression [75].
GDNF and the 5-HT-system. Data on GDNF and the 5-HT brain system interaction are scarce, but they are sufficient to suggest a close interaction of these systems. First, the effect of GDNF on the 5-HT-system of the brain has been verified in vitro experiments. In cell culture, GDNF increased the size of cell bodies and the number and length of 5-HT neuron axons [92]. A single central administration of GDNF reduced anxiety and manifestation of cataleptic immobility in predisposed to catalepsy and depression-like behavior mice of the ASC line. However, it increased signs of depression and stereotypic behavior [93]. These behavioral changes were concordant with a significant change in the expression of key genes of the 5-HT-system. Expression in the midbrain of the gene encoding the enzyme TPH-2 limiting synthesis of 5-HT in the brain increased. GDNF increased the expression of the gene encoding 5-HT2A receptors in the frontal cortex, but decreased it in the hippocampus [94, 95]. In turn, it was shown that in cell culture of rat C6 glioma, 5-HT (in dose- and time-dependent manner) increases GDNF expression and secretion, acting predominantly via 5-HT2A receptors [96]. This effect was achieved through transactivation via 5-HT2A receptors of fibroblast growth factor receptors [97]. At the same time, excess concentration of 5-HT decreases GDNF expression, thereby weakening mesencephalic neuronal differentiation [98].
Indirect evidence for a close connection between GDNF and the 5-HT-system is its ability to respond to the use of antidepressants, such as selective serotonin reuptake inhibitors (SSRIs). Several studies have shown that GDNF expression and secretion increase after a single or chronic administration of reuptake inhibitors in both cell cultures [99-101] and the serum of patients with depression after a course of antidepressant therapy [102].
These data demonstrate the existence of interactions in the GDNF–5-HT system, which is probably carried out with the participation of 5-HT2A receptors.
Discovery of neurotrophic brain factors has opened a new chapter in the understanding of neurogenesis and synaptic plasticity mechanisms. It also once again showed how complex regulatory systems of the brain are and how simplified current knowledge of the mechanisms of these regulations is. However, the paradigm of reductionism has significantly advanced understanding of brain functioning and highlighted reference points in the interaction of neurotransmitters and neurotrophic factors in the regulation of normal and pathological behavior.
The existence of a number of neurotrophic factors in the brain raises several issues. (i) How different are their functions – are they mostly doubles or possess individual features? (ii) What are their relations with the brain neurotransmitters, especially with 5-HT system as another regulator of neuroplasticity? The comparison of features of the “oldest” and the most studied BDNF with the data on GDNF, which is structurally different from BDNF and belongs to another family of neurotrophic factors, makes it possible to answer some of these questions.
BDNF has the highest expression and most apparent wide range of physiological effects. Accumulated evidence suggests close ties of BDNF and the 5-HT-system of the brain. It is important that this is 5-HT–BDNF interaction because not only BDNF is necessary for normal development and functioning of the 5-HT-systems, but brain 5-HT also affects BDNF.
GDNF, which is considered to be predominantly a regulator of the DA system, also interacts with brain 5-HT. As well as BDNF, it stimulates the growth of 5-HT brain neurons and affects the expression of key genes of the 5-HT-system of the brain, i.e. genes coding TPH-2 and 5-HT1A and 5-HT2A receptors. 5-HT2A receptors play an important role in the interaction mechanisms of these neurotrophic factors with the 5-HT-system. BDNF as well as GDNF effects depend on the genotype.
BDNF and GDNF are involved in the regulation of certain forms of behavior, including pathological behaviors. The basis of their impact is twofold. First, it is the ability to stimulate neurogenesis and restore functional activity that decreased during neurodegenerative pathology. Second, it is the interaction with the brain 5-HT-system – the regulator of the wide spectrum of normal and abnormal types of behavior.
The ability to stimulate neurons and synaptogenesis explains the unusually long-lasting BDNF and GDNF effects. We have found preservation of positive action of BDNF for 1.5 months after its single central administration [50]. This feature of BDNF and GDNF effects is unique among other pharmacological drugs.
However, differences have been detected. They manifested at different levels and in the structural distribution of expression of these factors in the brain: BDNF expression is much higher than that for GDNF. BDNF highest level was observed in the hippocampus and cerebral cortex, and GDNF expression – in the dopaminergic structure, the striatum.
The relative role of BDNF and GDNF in the regulation of the two neurotransmitter systems, 5-HT and DA, is also different. It is obvious that 5-HT is a priority for BDNF, while the DA brain system is more influenced by GDNF (figure). BDNF administration reduced gene expression of 5-HT2A receptors in the frontal cortex, increased it in the hippocampus, and increased functional activity of 5-HT2A receptors. In contrast, GDNF increased the expression of 5-HT2A receptors in the frontal cortex, decreased it in the hippocampus, and had no effect on their functional activity [50, 94, 95].
Interaction of neurotrophic factors and the 5-HT-system in the brain. VTA, ventral tegmental area; TPH-2, tryptophan hydroxylase-2; 5-HIAA, 5-hydroxyindoleacetic acid
Regional specificity of the BDNF and GDNF effects in the brain is apparently connected with the peculiarities of the microenvironment of different brain structures that affect the functioning of 5-HT neurons. Such peculiarities may be different density of 5-HT receptors, dimerization intensity, ratio, and interaction between various types of 5-HT receptors or with other types of receptors [103]. Regarding GDNF, an important factor is high heterogeneity of astrocytes. Astrocytes of different brain regions vary considerably in their biochemical characteristics, which undoubtedly affect their functioning, reactions, and role in different neuropathologies [104].
It is important that between the 5-HT-system and neurotrophic factors BDNF and GDNF there is a feedback mechanism, apparently autoregulating neurotransmitter–neurotrophic complexes: 5-HT–BDNF–5-HT and 5-HT–GDNF–5-HT. Different response of various structures 5-HT to the introduction of both BDNF and GDNF does not provide clear information about positive or negative feedback mechanisms in the interaction of these systems. However, this complex functional relationship is undoubtedly a factor of neuroplasticity and is a potential target for drugs that modulate the activity of neurotrophic brain factors.
Acknowledgements
The study was performed in the frames of basic research project No. 0324-2016-0002 and supported by RSF grant No. 14-25-00038.
REFERENCES
1.Gomazkov, O. A. (2007) Growth and neurotrophic
factors in the regulation of stem cell transformation and neurogenesis,
Neirokhimiya, 24, 101-112.
2.Popova, N. K., and Morozova, M. V. (2013)
Brain-derived neurotrophic factor: the influence on the genetically and
epigenetically determined behavioral disorders, Ross. Fiziol. Zh.
im. Sechenova, 99, 1125-1137.
3.Weissmiller, A. M., and Wu, C. (2012) Current
advances in using neurotrophic factors to treat neurodegenerative
disorders, Transl. Neurodegener., 1, doi:
10.1186/2047-9158-1–14.
4.Homberg, J. R., Molteni, R., Calabrese, F., and
Riva, M. A. (2014) The serotonin-BDNF duo: developmental implications
for the vulnerability to psychopathology, Neurosci. Biobehav.
Rev., 43, 35-47.
5.Nathanson, N. M. (2012) Regulation of neurokine
receptor signaling and trafficking, Neurochem. Int., 61,
874-878.
6.Voutilainen, M. H., Arumae, U., Airavaara, M., and
Saarma, M. (2015) Therapeutic potential of the endoplasmic reticulum
located and secreted CDNF/MANF family of neurotrophic factors in
Parkinson’s disease, FEBS Lett., 589,
3739-3748.
7.Levi-Montalcini, R. (1952) Effects of mouse tumor
transplantation on the nervous system, Ann. NY Acad. Sci.,
55, 330-344.
8.Barde, Y. A., Edgar, D., and Thoenen, H. (1982)
Purification of a new neurotrophic factor from mammalian brain, EMBO
J., 1, 549-553.
9.Cohen-Cory, S., Kidane, A. H., Shirkey, N. J., and
Marshak, S. (2010) Brain-derived neurotrophic factor and the
development of structural neuronal connectivity, Dev.
Neurobiol., 70, 271-288.
10.Hoyng, S. A., Tannemaat, M. R., De Winter, F.,
Verhaagen, J., and Malessy, M. J. (2011) Nerve surgery and gene
therapy: a neurobiological and clinical perspective, J. Hand. Surg.
Eur., 36, 735-746.
11.Lin, L. F., Doherty, D. H., Lile, J. D., Bektesh,
S., and Collins, F. (1993) GDNF: a glial cell line-derived neurotrophic
factor for midbrain dopaminergic neurons, Science, 260,
1130-1132.
12.Popova, N. K., Naumenko, E. V., and Kolpakov, V.
G. (1978) Serotonin and Behavior [in Russian], Nauka,
Novosibirsk, p. 304.
13.Naumenko, E. V. (1973) Central Regulation of
the Pituitary–Adrenal Complex, Plenum Publisher Corp.,
NY-London.
14.Linnoila, V. M., and Virkkunen, M. (1992)
Aggression, suicidality, and serotonin, J. Clin. Psychiatry,
53, 46-51.
15.Arango, V., Huang, Y. Y., Underwood, M. D., and
Mann, J. J. (2003) Genetics of the serotonergic system in suicidal
behavior, J. Psychiatr. Res., 37, 375-386.
16.Ilchibaeva, T. V., Kondaurova, E. M., Tsybko, A.
S., Kozhemyakina, R. V., Popova, N. K., and Naumenko, V. S. (2015)
Brain-derived neurotrophic factor (BDNF) and its precursor (proBDNF) in
genetically defined fear-induced aggression, Behav. Brain Res.,
290, 45-50.
17.Ilchibaeva, T. V., Tsybko, A. S., Kozhemyakina,
R. V., Popova, N. K., and Naumenko, V. S. (2016) Glial cell
line-derived neurotrophic factor in genetically defined fear-induced
aggression, Eur. J. Neurosci., 44, 2467-2473.
18.West, A. E., Pruunsild, P., and Timmusk, T.
(2014) Neurotrophins: transcription and translation, Handb. Exp.
Pharmacol., 220, 67-100.
19.Aid, T., Kazantseva, A., Piirsoo, M., Palm, K.,
and Timmusk, T. (2007) Mouse and rat BDNF gene structure and expression
revisited, J. Neurosci. Res., 85, 525-535.
20.Benarroch, E. E. (2015) Brain-derived
neurotrophic factor: regulation, effects, and potential clinical
relevance, Neurology, 84, 1693-1704.
21.Martinez-Levy, G. A., and Cruz-Fuentes, C. S.
(2014) Genetic and epigenetic regulation of the brain-derived
neurotrophic factor in the central nervous system, Yale J. Biol.
Med., 87, 173-186.
22.Karpova, N. N. (2014) Role of BDNF epigenetics in
activity-dependent neuronal plasticity, Neuropharmacology,
76, 709-718.
23.An, J. J., Gharami, K., Liao, G. Y., Woo, N. H.,
Lau, A. G., Vanevski, F., Torre, E. R., Jones, K. R., Feng, Y., Lu, B.,
and Xu, B. (2008) Distinct role of long 3′-UTR BDNF mRNA in spine
morphology and synaptic plasticity in hippocampal neurons, Cell,
134, 175-187.
24.Lau, A. G., Irier, H. A., Gu, J., Tian, D., Ku,
L., Liu, G., Xia, M., Fritsch, B., Zheng, J. Q., Dingledine, R., Xu,
B., Lu, B., and Feng, Y. (2010) Distinct 3′-UTRs differentially
regulate activity-dependent translation of brain-derived neurotrophic
factor (BDNF), Proc. Natl. Acad. Sci. USA, 107,
15945-15950.
25.Lu, B., Pang, P. T., and Woo, N. H. (2005) The
yin and yang of neurotrophin action, Nat. Rev. Neurosci.,
6, 603-614.
26.Naumenko, V. S., Kulikov, A. V., Kondaurova, E.
M., Tsybko, A. S., Kulikova, E. A., Krasnov, I. B., Shenkman, B. S.,
Sychev, V. N., Bazhenova, E. Y., Sinyakova, N. A., and Popova, N. K.
(2015) Effect of actual long-term spaceflight on BDNF, TrkB, p75, BAX
and BCL-XL genes expression in mouse brain regions,
Neuroscience, 284, 730-736.
27.Kenchappa, R. S., Tep, C., Korade, Z., Urra, S.,
Bronfman, F. C., Yoon, S. O., and Carter, B. D. (2010) p75 neurotrophin
receptor-mediated apoptosis in sympathetic neurons involves a biphasic
activation of JNK and up-regulation of tumor necrosis
factor-alpha-converting enzyme/ADAM17, J. Biol. Chem.,
285, 20358-20368.
28.Deinhardt, K., and Chao, M. V. (2014) Shaping
neurons: long and short range effects of mature and proBDNF signaling
upon neuronal structure, Neuropharmacology, 76,
603-609.
29.Borodinova, A. A., and Salozhin, S. V. (2016)
Diversity of proBDNF and mBDNF functions in the central nervous system,
Zh. Vyssh. Nerv. Deyat. im. Pavlova, 66, 3-23.
30.Lanni, C., Stanga, S., Racchi, M., and Govoni, S.
(2010) The expanding universe of neurotrophic factors: therapeutic
potential in aging and age-associated disorders, Curr. Pharm.
Des., 16, 698-717.
31.Sopova, K., Gatsiou, K., Stellos, K., and Laske,
C. (2014) Dysregulation of neurotrophic and hematopoietic growth
factors in Alzheimer’s disease: from pathophysiology to novel
treatment strategies, Curr. Alzheimer Res., 11,
27-39.
32.Budni, J., Bellettini-Santos, T., Mina, F.,
Garcez, M. L., and Zugno, A. I. (2015) The involvement of BDNF, NGF and
GDNF in aging and Alzheimer’s disease, Aging Dis.,
6, 331-341.
33.Beeri, M. S., and Sonnen, J. (2016) Brain BDNF
expression as a biomarker for cognitive reserve against
Alzheimer’s disease progression, Neurology, 86,
702-703.
34.He, Y. Y., Zhang, X. Y., Yung, W. H., Zhu, J. N.,
and Wang, J. J. (2013) Role of BDNF in central motor structures and
motor diseases, Mol. Neurobiol., 48, 783-793.
35.Zuccato, C., and Cattaneo, E. (2014)
Huntington’s disease, Handbook Exp. Pharmacol.,
220, 357-409.
36.Nguyen, K. Q., Rymar, V. V., and Sadikot, A. F.
(2016) Impaired TrkB signaling underlies reduced BDNF-mediated trophic
support of striatal neurons in the R6/2 mouse model of
Huntington’s disease, Front. Cell Neurosci., 10,
doi: 10.3389/fncel.2016.00037.
37.Paillard, T., Rolland, Y., and De Souto Barreto,
P. (2015) Protective effects of physical exercise in Alzheimer’s
disease and Parkinson’s disease: a narrative review, J. Clin.
Neurol., 11, 212-219.
38.Pezet, S. (2014) Neurotrophins and pain, Biol.
Aujourd’hui, 208, 21-29.
39.Khan, N., and Smith, M. T. (2015) Neurotrophins
and neuropathic pain: role in pathobiology, Molecules,
20, 10657-10688.
40.Ahmed, A. O., Mantini, A. M., Fridberg, D. J.,
and Buckley, P. F. (2015) Brain-derived neurotrophic factor (BDNF) and
neurocognitive deficits in people with schizophrenia: a meta-analysis,
Psychiatry Res., 226, 1-13.
41.Libman-Sokołowska, M., Drozdowicz, E., and
Nasierowski, T. (2015) BDNF as a biomarker in the course and treatment
of schizophrenia, Psychiatr. Pol., 49, 1149-1158.
42.Autry, A. E., and Monteggia, L. M. (2012)
Brain-derived neurotrophic factor and neuropsychiatric disorders,
Pharmacol. Rev., 64, 238-258.
43.Scola, G., and Andreazza, A. C. (2015) The role
of neurotrophins in bipolar disorder, Prog. Neuropsychopharmacol.
Biol. Psychiatry, 56, 122-128.
44.Li, X., and Wolf, M. E. (2015) Multiple faces of
BDNF in cocaine addiction, Behav. Brain Res., 279,
240-254.
45.Pitts, E. G., Taylor, J. R., and Gourley, S. L.
(2016) Prefrontal cortical BDNF: a regulatory key in cocaine- and
food-reinforced behaviors, Neurobiol. Dis., 91,
326-335.
46.Rumajogee, P., Madeira, A., Verge, D., Hamon, M.,
and Miquel, M. C. (2002) Up-regulation of the neuronal serotoninergic
phenotype in vitro: BDNF and cAMP share Trk B-dependent
mechanisms, J. Neurochem., 83, 1525-1528.
47.Celada, P., Siuciak, J. A., Tran, T. M., Altar,
C. A., and Tepper, J. M. (1996) Local infusion of brain-derived
neurotrophic factor modifies the firing pattern of dorsal raphe
serotonergic neurons, Brain Res., 712, 293-298.
48.Siuciak, J. A., Boylan, C., Fritsche, M., Altar,
C. A., and Lindsay, R. M. (1996) BDNF increases monoaminergic activity
in rat brain following intracerebroventricular or intraparenchymal
administration, Brain Res., 710, 11-20.
49.Mamounas, L. A., Altar, C. A., Blue, M. E.,
Kaplan, D. R., Tessarollo, L., and Lyons, W. E. (2000) BDNF promotes
the regenerative sprouting, but not survival, of injured serotonergic
axons in the adult rat brain, J. Neurosci., 20,
771-782.
50.Naumenko, V. S., Kondaurova, E. M., Bazovkina, D.
V., Tsybko, A. S., Tikhonova, M. A., Kulikov, A. V., and Popova, N. K.
(2012) Effect of brain-derived neurotrophic factor on behavior and key
members of the brain serotonin system in genetically predisposed to
behavioral disorders mouse strains, Neuroscience, 214,
59-67.
51.Naumenko, V. S., Bazovkina, D. V., Morozova, M.
V., and Popova, N. K. (2013) Effects of brain-derived and glial cell
line-derived neurotrophic factors on startle response and disrupted
prepulse inhibition in mice of DBA/2J inbred strain, Neurosci.
Lett., 550, 115-118.
52.Lyons, W. E., Mamounas, L. A., Ricaurte, G. A.,
Coppola, V., Reid, S. W., Bora, S. H., Wihler, C., Koliatsos, V. E.,
and Tessarollo, L. (1999) Brain-derived neurotrophic factor-deficient
mice develop aggressiveness and hyperphagia in conjunction with brain
serotonergic abnormalities, Proc. Natl. Acad. Sci. USA,
96, 15239-15244.
53.Trajkovska, V., Santini, M. A., Marcussen, A. B.,
Thomsen, M. S., Hansen, H. H., Mikkelsen, J. D., Arneberg, L., Kokaia,
M., Knudsen, G. M., and Aznar, S. (2009) BDNF downregulates 5-HT(2A)
receptor protein levels in hippocampal cultures, Neurochem.
Int., 55, 697-702.
54.Rios, M., Fan, G., Fekete, C., Kelly, J., Bates,
B., Kuehn, R., Lechan, R. M., and Jaenisch, R. (2001) Conditional
deletion of brain-derived neurotrophic factor in the postnatal brain
leads to obesity and hyperactivity, Mol. Endocrinol., 15,
1748-1757.
55.Rios, M., Lambe, E. K., Liu, R., Teillon, S.,
Liu, J., Akbarian, S., Roffler-Tarlov, S., Jaenisch, R., and
Aghajanian, G. K. (2006) Severe deficits in 5-HT2A-mediated
neurotransmission in BDNF conditional mutant mice, J.
Neurobiol., 66, 408-420.
56.Klein, A. B., Santini, M. A., Aznar, S., Knudsen,
G. M., and Rios, M. (2010) Changes in 5-HT2A-mediated behavior and
5-HT2A- and 5-HT1A receptor binding and expression in conditional
brain-derived neurotrophic factor knock-out mice, Neuroscience,
169, 1007-1016.
57.Galter, D., and Unsicker, K. (2000) Sequential
activation of the 5-HT1(A) serotonin receptor and TrkB induces the
serotonergic neuronal phenotype, Mol. Cell. Neurosci.,
15, 446-455.
58.Vaidya, V. A., Marek, G. J., Aghajanian, G. K.,
and Duman, R. S. (1997) 5-HT2A receptor-mediated regulation of
brain-derived neurotrophic factor mRNA in the hippocampus and the
neocortex, J. Neurosci., 17, 2785-2795.
59.Molteni, R., Cattaneo, A., Calabrese, F., Macchi,
F., Olivier, J. D., Racagni, G., Ellenbroek, B. A., Gennarelli, M., and
Riva, M. A. (2010) Reduced function of the serotonin transporter is
associated with decreased expression of BDNF in rodents as well as in
humans, Neurobiol. Dis., 37, 747-755.
60.Calabrese, F., Guidotti, G., Middelman, A.,
Racagni, G., Homberg, J., and Riva, M. A. (2013) Lack of serotonin
transporter alters BDNF expression in the rat brain during early
postnatal development, Mol. Neurobiol., 48, 244-256.
61.Calabrese, F., van der Doelen, R. H., Guidotti,
G., Racagni, G., Kozicz, T., Homberg, J. R., and Riva, M. A. (2015)
Exposure to early life stress regulates BDNF expression in SERT mutant
rats in an anatomically selective fashion, J. Neurochem.,
132, 146-154.
62.Schiller, L., Donix, M., Jahkel, M., and Oehler,
J. (2006) Serotonin 1A and 2A receptor densities, neurochemical and
behavioral characteristics in two closely related mice strains after
long-term isolation, Prog. Neuropsychopharmacol. Biol.
Psychiatry, 30, 492-503.
63.Lang, U. E., Gunther, L., Scheuch, K., Klein, J.,
Eckhart, S., Hellweg, R., Danker-Hopfe, H., and Oehler, J. (2009)
Higher BDNF concentrations in the hippocampus and cortex of an
aggressive mouse strain, Behav. Brain Res., 197,
246-249.
64.Popova, N. K. (2006) From genes to aggressive
behavior: the role of serotonergic system, Bioessays, 28,
495-503.
65.Popova, N. K., Voitenko, N. N., Kulikov, A. V.,
and Avgustinovich, D. F. (1991) Evidence for the involvement of central
serotonin in mechanism of domestication of silver foxes, Pharmacol.
Biochem. Behav., 40, 751-756.
66.Popova, N. K., Naumenko, V. S., Plyusnina, I. Z.,
and Kulikov, A. V. (2005) Reduction in 5-HT1A receptor density, 5-HT1A
mRNA expression, and functional correlates for 5-HT1A receptors in
genetically defined aggressive rats, J. Neurosci. Res.,
80, 286-292.
67.Maragakis, N. J., and Rothstein, J. D. (2006)
Mechanisms of disease: astrocytes in neurodegenerative disease, Nat.
Clin. Pract. Neurol., 2, 679-689.
68.Capani, F., Quarracino, C., Caccuri, R., and
Sica, R. E. (2016) Astrocytes as the main players in primary
degenerative disorders of the human central nervous system, Front.
Aging Neurosci., 8, doi: 10.3389/fnagi.2016.00045.
69.Pascual, A., Hidalgo-Figueroa, M., Gomez-Diaz,
R., and Lopez-Barneo, J. (2011) GDNF and protection of adult central
catecholaminergic neurons, J. Mol. Endocrinol., 46,
83-92.
70.Ibanez, C. F., and Andressoo, J. O. (2016)
Biology of GDNF and its receptors – relevance for disorders
of the central nervous system, Neurobiol. Dis., doi:
10.1016/j.nbd.2016.01.021.
71.Airaksinen, M. S., and Saarma, M. (2002) The GDNF
family: signaling, biological functions and therapeutic value, Nat.
Rev. Neurosci., 3, 383-394.
72.Sariola, H., and Saarma, M. (2003) Novel
functions and signaling pathways for GDNF, J. Cell Sci.,
116, 3855-3862.
73.Sun, X. L., Chen, B. Y., Duan, L., Xia, Y., Luo,
Z. J., Wang, J. J., Rao, Z. R., and Chen, L. W. (2014) The proform of
glia cell line-derived neurotrophic factor: a potentially biologically
active protein, Mol. Neurobiol., 49, 234-250.
74.Immonen, T., Alakuijala, A., Hytonen, M., Sainio,
K., Poteryaev, D., Saarma, M., Pasternack, M., and Sariola, H. (2008) A
proGDNF-related peptide BEP increases synaptic excitation in rat
hippocampus, Exp. Neurol., 210, 793-796.
75.Bradley, L. H., Fuqua, J., Richardson, A.,
Turchan-Cholewo, J., Ai, Y., Kelps, K. A., Glass, J. D., He, X., Zhang,
Z., Grondin, R., Littrell, O. M., Huettl, P., Pomerleau, F., Gash, D.
M., and Gerhardt, G. A. (2010) Dopamine neuron stimulating actions of a
GDNF propeptide, PLoS One, 5, doi:
10.1371/journal.pone.0009752.
76.Saavedra, A., Baltazar, G., and Duarte, E. P.
(2008) Driving GDNF expression: the green and the red traffic lights,
Prog. Neurobiol., 86, 186-215.
77.Ledda, F., Paratcha, G., Sandoval-Guzman, T., and
Ibanez, C. F. (2007) GDNF and GFRalpha1 promote formation of neuronal
synapses by ligand-induced cell adhesion, Nat. Neurosci.,
10, 293-300.
78.Naumenko, V. S., Kondaurova, E. M., Bazovkina, D.
V., Tsybko, A. S., Ilchibaeva, T. V., Khotskin, N. V., Semenova, A. A.,
and Popova, N. K. (2014) Effect of GDNF on depressive-like behavior,
spatial learning and key genes of the brain dopamine system in
genetically predisposed to behavioral disorders mouse strains,
Behav. Brain Res., 274, 1-9.
79.Igarashi, Y., Chiba, H., Utsumi, H., Miyajima,
H., Ishizaki, T., Gotoh, T., Kuwahara, K., Tobioka, H., Satoh, M.,
Mori, M., and Sawada, N. (2000) Expression of receptors for glial cell
line-derived neurotrophic factor (GDNF) and neurturin in the inner
blood-retinal barrier of rats, Cell Struct. Funct., 25,
237-241.
80.Nishikiori, N., Osanai, M., Chiba, H., Kojima,
T., Mitamura, Y., Ohguro, H., and Sawada, N. (2007) Glial cell-derived
cytokines attenuate the breakdown of vascular integrity in diabetic
retinopathy, Diabetes, 56, 1333-1340.
81.Shimizu, F., Sano, Y., Saito, K., Abe, M. A.,
Maeda, T., Haruki, H., and Kanda, T. (2012) Pericyte-derived glial cell
line-derived neurotrophic factor increase the expression of claudin-5
in the blood-brain barrier and the blood-nerve barrier, Neurochem.
Res., 37, 401-409.
82.Rocha, S. M., Cristovão, A. C., Campos, F.
L., Fonseca, C. P., and Baltazar, G. (2012) Astrocyte-derived GDNF is a
potent inhibitor of microglial activation, Neurobiol. Dis.,
47, 407-415.
83.Allen, S. J., Watson, J. J., Shoemark, D. K.,
Barua, N. U., and Patel, N. K. (2013) GDNF, NGF and BDNF as therapeutic
options for neurodegeneration, Pharmacol. Ther., 138,
155-175.
84.Lin, P. Y., and Tseng, P. T. (2015) Decreased
glial cell line-derived neurotrophic factor levels in patients with
depression: a meta-analytic study, J. Psychiatr. Res.,
63, 20-27.
85.Liu, Q., Zhu, H. Y., Li, B., Wang, Y. Q., Yu, J.,
and Wu, G. C. (2012) Chronic clomipramine treatment restores
hippocampal expression of glial cell line-derived neurotrophic factor
in a rat model of depression, J. Affect. Disord., 141,
367-372.
86.Uchida, S., Hara, K., Kobayashi, A., Otsuki, K.,
Yamagata, H., Hobara, T., Suzuki, T., Miyata, N., and Watanabe, Y.
(2011) Epigenetic status of GDNF in the ventral striatum determines
susceptibility and adaptation to daily stressful events, Neuron,
69, 359-372.
87.Verity, A. N., Wyatt, T. L., Lee, W., Hajos, B.,
Baecker, P. A., Eglen, R. M., and Johnson, R. M. (1999) Differential
regulation of glial cell line-derived neurotrophic factor (GDNF)
expression in human neuroblastoma and glioblastoma cell lines, J.
Neurosci. Res., 55, 187-197.
88.Nakashima, S., Matsuyama, Y., Yu, Y., Kiuchi, K.,
and Ishiguro, N. (2004) Suppression of GDNF production by MPSS
treatment following spinal cord injury in the rat, Neuroreport,
15, 2337-2340.
89.Henkel, A. W., Alali, H., Devassy, A., Alawadi,
M. M., and Redzic, Z. B. (2014) Antagonistic interactions between
dexamethasone and fluoxetine modulate morphodynamics and expression of
cytokines in astrocytes, Neuroscience, 280, 318-327.
90.McEwen, B. S., Bowles, N. P., Gray, J. D., Hill,
M. N., Hunter, R. G., Karatsoreos, I. N., and Nasca, C. (2015)
Mechanisms of stress in the brain, Nat. Neurosci., 18,
1353-1363.
91.Maheu, M., Lopez, J. P., Crapper, L., Davoli, M.
A., Turecki, G., and Mechawar, N. (2015) MicroRNA regulation of central
glial cell line-derived neurotrophic factor (GDNF) signaling in
depression, Transl. Psychiatry, 5, doi:
10.1038/tp.2015.11.
92.Ducray, A., Krebs, S. H., Schaller, B., Seiler,
R. W., Meyer, M., and Widmer, H. R. (2006) GDNF family ligands display
distinct action profiles on cultured GABAergic and serotonergic neurons
of rat ventral mesencephalon, Brain Res., 1069,
104-112.
93.Semenova, A. A., Bazovkina, D. B., Tsybko, A. S.,
Naumenko, V. S., and Popova, N. K. (2013) Effect of GDNF on the
behavior of ASC mice with high hereditary predisposition to catalepsy,
Zh. Vyssh. Nerv. Deyat. im. Pavlova, 63,
495-501.
94.Naumenko, V. S., Bazovkina, D. V., Semenova, A.
A., Tsybko, A. S., Il’chibaeva, T. V., Kondaurova, E. M., and
Popova, N. K. (2013) Effect of glial cell line-derived neurotrophic
factor on behavior and key members of the brain serotonin system in
mouse strains genetically predisposed to behavioral disorders, J.
Neurosci. Res., 91, 1628-1638.
95.Tsybko, A. S., Il’chibaeva, T. V.,
Kondaurova, E. M., Bazovkina, D. V., and Naumenko, V. S. (2014) The
effect of central administration of the neurotrophic factors BDNF and
GDNF on the functional activity and expression of the serotonin 5-HT2A
receptors in mice genetically predisposed to depressive-like behavior,
Mol. Biol. (Moscow), 48, 983-989.
96.Hisaoka, K., Nishida, A., Takebayashi, M., Koda,
T., Yamawaki, S., and Nakata, Y. (2004) Serotonin increases glial cell
line-derived neurotrophic factor release in rat C6 glioblastoma cells,
Brain Res., 1002, 167-170.
97.Tsuchioka, M., Takebayashi, M., Hisaoka, K.,
Maeda, N., and Nakata, Y. (2008) Serotonin (5-HT) induces glial cell
line-derived neurotrophic factor (GDNF) mRNA expression via the
transactivation of fibroblast growth factor receptor 2 (FGFR2) in rat
C6 glioma cells, J. Neurochem., 106, 244-257.
98.Menegola, E., Broccia, M. L., Di Renzo, F.,
Massa, V., and Giavini, E. (2004) Effects of excess and deprivation of
serotonin on in vitro neuronal differentiation, In vitro
Cell. Dev. Biol. Anim., 40, 52-56.
99.Hisaoka, K., Nishida, A., Koda, T., Miyata, M.,
Zensho, H., Morinobu, S., Ohta, M., and Yamawaki, S. (2001)
Antidepressant drug treatments induce glial cell line-derived
neurotrophic factor (GDNF) synthesis and release in rat C6 glioblastoma
cells, J. Neurochem., 79, 25-34.
100.Mercier, G., Lennon, A. M., Renouf, B.,
Dessouroux, A., Ramauge, M., Courtin, F., and Pierre, M. (2004) MAP
kinase activation by fluoxetine and its relation to gene expression in
cultured rat astrocytes, J. Mol. Neurosci., 24,
207-216.
101.Golan, M., Schreiber, G., and Avissar, S.
(2011) Antidepressants elevate GDNF expression and release from C6
glioma cells in a β-arrestin1-dependent, CREB interactive pathway,
Int. J. Neuropsychopharmacol., 14, 1289-1300.
102.Zhang, X., Zhang, Z., Xie, C., Xi, G., Zhou,
H., Zhang, Y., and Sha, W. (2008) Effect of treatment on serum glial
cell line-derived neurotrophic factor in depressed patients, Prog.
Neuropsychopharmacol. Biol. Psychiatry, 32, 886-890.
103.Naumenko, V. S., Popova, N. K., Lacivita, E.,
Leopoldo, M., and Ponimaskin, E. G. (2014) Interplay between serotonin
5-HT1A and 5-HT7 receptors in depressive disorders, CNS Neurosci.
Ther., 20, 582-590.
104.Montgomery, D. L. (1994) Astrocytes: form,
functions, and roles in disease, Vet. Pathol., 31,
145-167.