Prevalence of Autoantibodies against 3-DG-Glycated H2A Protein in Type 2 Diabetes
J. M. Ashraf1*, S. M. S. Abdullah1, S. Ahmad2, S. Fatma3, M. H. Baig4, J. Iqbal5, A. M. Madkhali1, and A. B. A. Jerah1
1Jazan University, Faculty of Applied Medical Sciences, Jazan, Saudi Arabia; E-mail: jmashraf@gmail.com2Integral University, Department of Biotechnology, Lucknow, India
3Banaras Hindu University, Faculty of Science, Varanasi, India
4Yeungnam University, Department of Medical Biotechnology, Gyeongsan, Republic of Korea
5Jazan University, Faculty of Medicine, Jazan, Saudi Arabia
* To whom correspondence should be addressed.
Received October 11, 2016; Revision received January 5, 2017
Advanced glycation end-products (AGEs) have been found to be critically involved in initiation or progression of diabetes secondary complications (nephropathy, retinopathy, neuropathy, and angiopathy). Various hyper-glycating carbonyl compounds such as 3-deoxyglucosone (3-DG) are produced in pathophysiological conditions that form AGEs in high quantity both in vivo and in vitro. In the first stage of this study, we glycated histone H2A protein by 3-DG, and the results showed the formation of various intermediates and AGEs as well as structural changes in the protein. In the second stage, we studied the immunogenicity of native and 3-DG-glycated H2A protein in female rabbits. The modified H2A was highly immunogenic, eliciting high titer immunogen-specific antibodies, while the unmodified form was almost nonimmunogenic. Antibodies against standard carboxymethyllysine (CML) and pentosidine were detected in the immunized female rabbits, which demonstrates the immunogenic nature of AGEs (CML and pentosidine) as well. The results show both structural perturbation and AGEs have the capacity of triggering the immune system due to the generation of neoepitopes that render the molecule immunogenic. This study shows the presence of autoantibodies against 3-DG-modified H2A, CML, and pentosidine in the sera of type 2 diabetes patients having secondary complications. Autoantibodies against damaged H2A and AGEs may be significant in the assessment of initiation/progression of secondary complications in type 2 diabetes mellitus patients or may be used as a marker for early detection of secondary complications in diabetes.
KEY WORDS: advanced glycation end-products, anti-AGE antibodies, ELISA, autoantibodies, immunogenicity, type 2 diabetesDOI: 10.1134/S0006297917050066
Abbreviations: AGEs, advanced glycation end-products; CML, carboxymethyllysine; 3-DG, 3-deoxyglucosone.
A typical nonenzymatic reaction of amino acids, proteins, or nucleic
acids with reducing sugars results in the initiation of formation of
Schiff base and/or Amadori products, which then undergo a sequence of
irreversible chemical reactions to form advanced glycation end-products
(AGEs) [1]. AGEs have been shown to adversely
affect the functional properties of proteins, lipids, and DNA [2, 3] and are considered directly
or indirectly involved in the development of diabetes and uremic
complications such as atherosclerosis, nephropathy, retinopathy, and
neuropathy [4-6].
It is reasonably well documented that consistent hyperglycemia has a major role in the pathogenesis of diabetes complications due to enhanced production of glycating intermediates (α-oxoaldehydes) and the continuous buildup of AGEs in body tissues [7, 8]. In the beginning, glucose was considered as the major source of AGEs; later, various sugars and non-sugar compounds were recognized and found to be involved in development of AGEs. It is very well established that under hyperglycemic conditions the level of α-oxoaldehydes (3-deoxyglucosone (3-DG), methylglyoxal and glyoxal) increases several-fold in diabetes patients, and higher content of α-oxoaldehydes-derived AGEs have been detected in tissue proteins [9-11]. Protein AGEs formed by 3-DG result in the inactivation of critical cellular proteins, which may lead to apoptosis or necrosis or cell growth arrest [12, 13].
The long-lived proteins including collagen, elastin, and lens crystalline and DNA are particularly susceptible to AGE modification [14, 15]. Histones (H2A, H2B, H3, and H4) are long-lived proteins that are essential components of eukaryotic chromatin structure [16]. Since individual histones (such as H2A) [17] and histones in nucleosomes [18] have been found to be glycated by reducing sugars in vitro and in vivo [19, 20], it is believed that this process affects chromatin structure and function [21].
The evidence that AGEs have antigenic properties led to the hypothesis that continuous formation of AGEs in a diabetic body can provoke autoimmune response [22]. Earlier study has shown the presence of protein AGEs and its immune complexes in several human tissues including kidney in diabetic nephropathic patients [23]. Study has found antigenicity of glycated poly-L-lysine in experimental animals, and autoantibodies were also detected against modified lysine polypeptide in diabetes patients [24]. Histone proteins are lysine-rich molecules, suggesting the importance of autoantibodies against lysine polypeptide in diabetes subjects. Furthermore, another study reported the detection of circulating autoantibodies against methylglyoxal-glycated histones in type 1 diabetes mellitus patients [25].
Increase in autoantibodies against AGEs may go in correspondence with the development of complications, and thus quantitative estimations of such autoantibodies may be important from the point of view of diabetes complications. Based on previous reports about detection of significant amounts of AGEs in different pathological conditions, the impact of nonenzymatic glycation in health and disease has become a focus of great significance.
In the present study, H2A histone protein was glycated with 3-DG. The extent of glycation, formation of intermediates and AGEs (carboxymethyllysine (CML), pentosidine), and structural change in glycated H2A histone were studied by various physicochemical techniques [26]. Furthermore, immunogenicity of native and glycated H2A was probed by inducing antibodies in experimental animals. Aliquots from the same (native and glycated H2A) samples were injected into experimental animals, and anti-glycated-H2A histone antibodies were characterized by immunoassay techniques. The presence of autoantibodies in type 2 diabetes patients was also probed against 3-DG-glycated H2A histone and AGEs (CML and pentosidine).
MATERIALS AND METHODS
Materials. Anti-human IgG, anti-rabbit IgG, 2,4-dinitrophenyl hydrazine, 9,10-phenanthrenequinone, dialysis tubing, alkaline phosphatase conjugates, p-nitrophenyl phosphate, sodium dodecyl sulfate (SDS), Tween-20, and Protein A-agarose (2.5 ml pre-pack column) were acquired from Sigma Chemical Company (USA). Triton X-100 was from Hi-Media (India). ELISA plates (96 wells) were purchased from NUNC (Denmark). All other reagents/chemicals were of the highest analytical grade available.
The same 3-DG-glycated H2A histone was used in this work as in our earlier study [26].
Immunization schedule. The immunization of random bred female New Zealand white rabbits with native and glycated H2A histone was performed as follows. Rabbits (n = 4; two each for native and glycated H2A) were immunized intramuscularly at multiple sites with 100 µg of antigen emulsified with an equal volume of Freund’s complete adjuvant. The animals were boosted with the same amount of antigen in Freund’s incomplete adjuvant at weekly intervals for 6 weeks. Antibody titer was evaluated at every alternate booster dose. Test bleeds, performed 7 days post boost gave significant antibody titer. The animals were bled and the serum (preimmune and immune) was heated at 56°C for 30 min to inactivate complement proteins and stored at –20°C with 0.1% sodium azide as preservative [27]. The Institutional Ethics Committee approved the study protocol.
Collection of sera. Fasting blood samples of type 2 diabetes patients were obtained from IIMS&R, Integral University. Consent was obtained from all subjects before the withdrawal of blood samples, and the protocol was approved by the Institutional Ethics Committee of IIMS&R. All procedures were in accordance with the Helsinki Declaration of 1975 as revised in 2008. All patients were evaluated for secondary complications (nephropathy, retinopathy, neuropathy, and angiopathy) of type 2 diabetes and were under anti-diabetic treatment. Age- and sex-matched normal human sera obtained from healthy subjects served as control. Samples were collected in glass test tubes and left to clot for 30 min at 37°C. Serum was separated by centrifugation at 2000g for 10 min. Serum samples were then heated at 56°C for 30 min to inactivate complement proteins and stored in aliquots at –20°C with 0.1% sodium azide as preservative. Some important clinical information on subjects included in the study is shown in Table 1.
Table 1. Important clinical characteristics
of normal and type 2 diabetic subjects
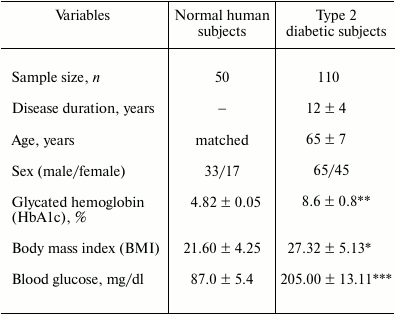
Note: Disease duration, age, HbA1c (%), BMI, and blood glucose levels
are the means and standard errors of three determinations. The value of
BMI is moderately higher in type 2 diabetic subjects (* p ≤
0.05 vs. normal human subjects); the p value in case of HbA1c
(%) is significantly higher (** p ≤ 0.012 vs. normal human
subjects) than that of the normal human subjects; and for the blood
glucose level it is highly significant (*** p ≤ 0.001 vs.
normal human subjects).
Protein A-agarose affinity chromatography. IgG from rabbit sera was purified by affinity chromatography on Protein A-agarose column following the manufacturer’s instruction and published literature [28]. In brief, serum (0.3 ml) diluted with an equal volume of PBS was applied on the column preequilibrated with the same buffer. The wash-through was recycled 2-3 times, and unbound material was removed by extensive washing with PBS. The bound IgG was eluted with 0.58% acetic acid in 0.85% sodium chloride and collected in a tube containing 1 ml of 1 M Tris-HCl (pH 8.5). Three-milliliter fractions were collected, and absorbance was read at 278 nm. The IgG concentration was determined considering that 1.4 OD278 corresponds to 1 mg/ml IgG. The isolated IgG was dialyzed against PBS and stored at –20°C with 0.1% sodium azide.
Enzyme linked immunosorbent assay (ELISA) for detection of antibodies and autoantibodies against respective antigens in immunized rabbits and diabetes patients, respectively. ELISA was performed on flat bottom polystyrene plates as described previously [29]. Polystyrene PolySorp immune plates with 96 wells were coated with 100 µl of native or glycated H2A or standard CML/pentosidine (10 µg/ml) in antigen coating buffer. The plates were first incubated for 2 h at 37°C and then overnight at 4°C. Each sample was assayed in duplicate, and half of the plate without antigen coating served as control. The wells of the test plates were washed three times with TBS-T (20 mM Tris, 2.68 mM KCl, 150 mM NaCl, pH 7.4, containing 0.05% Tween-20), and unoccupied sites were blocked with 150 µl of 2.5% fat-free skimmed milk in TBS (10 mM Tris, 150 mM NaCl, pH 7.4) for 4-6 h at 37°C. After incubation, the plates were washed 5-6 times with TBS-T. Test serum serially diluted in TBS-T was added to each well (100 µl/well) and incubated for 2 h at 37°C and then overnight at 4°C. After incubation, the plates were washed three times with TBS-T, and bound antibodies were assayed with anti-rabbit alkaline phosphatase conjugate in TBS. After incubation for 2 h, the plates were again washed three times with TBS-T and twice with distilled water and then coated with p-nitrophenyl phosphate (substrate for alkaline phosphatase). Absorbance (A) in each well was monitored at 410 nm on an automatic microplate reader. Each sample was run in duplicate. Results are expressed as mean of (Atest − Acontrol).
For detection of autoantibodies against glycated H2A or CML/pentosidine in diabetes patients’ sera, all the steps were same except in place of rabbit sera, human patients’ sera were used in 1 : 100 dilution in TBS buffer.
Competition ELISA for detection of specificity of antibodies and autoantibodies against respective antigens in immunized rabbits and diabetes patients, respectively. The antigenic specificity of the raised antibodies in immunized rabbits’ sera and of autoantibodies in diabetes patients’ sera was determined by competition ELISA [30]. The ELISA plates were coated with 100 μl of antigen (native or glycated H2A, 10 μg/ml) for 2 h at 37°C and overnight at 4°C. Varying amounts of inhibitors (0-20 μg/ml) were mixed with a constant amount of affinity purified IgG (40 µg/ml) and were allowed to interact for 2 h at room temperature and overnight at 4°C. The immune complex thus formed was coated in the wells instead of the serum. The remaining steps were the same as described for direct-binding ELISA. Percent inhibition was calculated using the formula:
Inhibition (%) = (1 – Ainhibited/Auninhibited) × 100.
Statistical analysis. Data are presented as mean ± standard deviation (SD). Statistical significance of data was determined by Student’s t-test, with p < 0.05 considered significant.
RESULTS AND DISCUSSION
Biomolecules in chronic and persistent diabetes patients are highly susceptible to be assaulted by various glycating agents like reducing sugars and carbonyl compounds, resulting in formation of AGEs through nonenzymatic glycation, which greatly changes protein structure and thus protein function [31-33]. Changes in protein structure by various modifications changes protein immunogenicity [34]. Several studies have shown that sera of diabetic individuals contain autoantibodies against various proteins AGE structures [35, 36] that can trigger autoimmune response. These antibodies have been established to be involved in the initiation and propagation of various pathological disorders like diabetes nephropathy and chronic renal failure, atherosclerotic lesions of arterial walls, and Alzheimer’s disease [25, 36, 37]. Considering the potential use of anti-AGE antibodies as a marker of AGE deposition in diabetes patients, we investigated the immunogenicity of 3-DG-glycated H2A and the presence of anti-AGE autoantibodies in type 2 diabetes sera and its likely role in diabetes autoimmunity causing secondary complications.
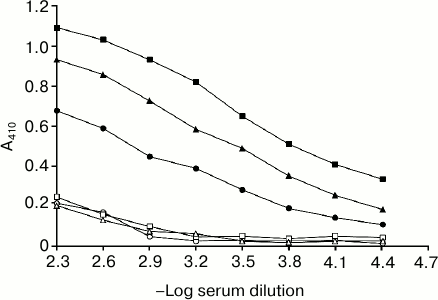
The first step of the study was to determine the effect of glycation on the immunogenicity of 3-DG-glycated H2A [26], which was evaluated by inducing antibodies in rabbits. Sera of animals immunized with native and glycated H2A were tested on PolySorp wells coated with the respective immunogens. Native H2A did not elicit an immune response and was found to be nonimmunogenic, but glycation by 3-DG made it highly immunogenic. Direct-binding ELISA showed that 3-DG-glycated H2A induced high titer antibodies (>1 : 12,800), while native H2A did not show significant titer under identical experimental conditions. Antibody titers against standard CML and pentosidine were also evaluated by direct-binding ELISA, showing high antibody titers around 1 : 12,800 in both cases (Fig. 1). Preimmune serum served as a negative control and did not show appreciable binding with the respective immunogens. The high titer antibody clearly indicates generation of neoepitopes in H2A by 3-DG, making it highly immunogenic. High antibody titers against standard CML and pentosidine suggest that CML and pentosidine are also highly immunogenic in nature as well as support the formation of AGEs (CML and pentosidine) during the glycation of H2A by 3-DG [26]. Structural changes in H2A caused by 3-DG glycation may also favor polymerization of native epitopes of H2A into potent immunogenic neoepitopes. Therefore, the result clearly shows that there is a direct correlation between glycation and immunogenicity.
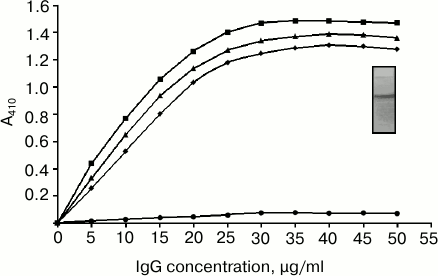
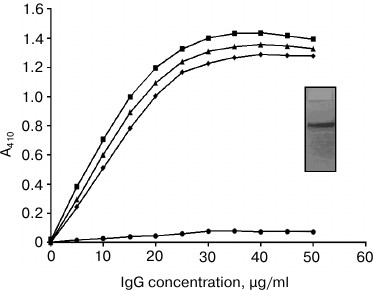
Antibodies were isolated from preimmune and immune (with native and glycated H2A as immunogens) rabbit antiserum by affinity chromatography on a Protein A-agarose column. The purity of isolated antibodies was evaluated by SDS-PAGE under nonreducing conditions. The purified IgG migrated as a single band upon electrophoresis on a 10% polyacrylamide gel. The appearance of a single band in SDS-PAGE confirms the isolated IgG is highly purified (Fig. 2; inset). Direct-binding ELISA of purified anti-glycated-H2A antibodies showed stronger reactivity toward its immunogen as well as CML and pentosidine compared to native H2A (Fig. 2). Preimmune antibodies showed negligible binding under identical conditions (data not shown).
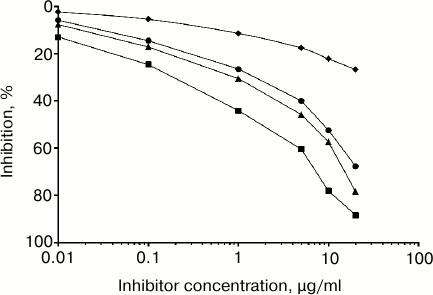
The antigenic specificity of the purified anti-glycated-H2A antibodies was characterized by competitive inhibition assay. A maximum of 88.4% inhibition of the anti-glycated-H2A antibodies was observed using the immunogen (i.e. glycated H2A) as inhibitor at 20 µg/ml (Fig. 3). The concentration of glycated H2A required for 50% inhibition was only 2.4 µg/ml. In the case of induced anti-native H2A antibodies, negligible inhibition was observed (data not shown). When 20 µg/ml CML and pentosidine was used as inhibitor, a maximum 78.5 and 67.2% inhibition was observed (Fig. 3). The concentrations of CML and pentosidine required to cause 50% inhibition were 3.6 and 4.2 µg/ml, respectively. Preimmune antibodies showed negligible inhibition with the studied inhibitors (glycated H2A, CML, and pentosidine) under identical experimental conditions. This demonstrates that the induced antibodies (anti-glycated H2A) are highly specific toward the glycated H2A, CML, and pentosidine epitopes.
Fig. 1. Level of antibodies against glycated H2A, standard CML, and pentosidine antigens. Direct-binding ELISA of glycated H2A, CML, and pentosidine with preimmune (open square) and immune (closed square), preimmune (open triangle) and immune (closed triangle), and preimmune (open circle) and immune (closed circle) sera, respectively. The microtiter plates were coated with the respective antigens (10 µg/ml).
Fig. 2. Direct-binding ELISA of affinity purified IgG (anti-glycated-H2A antibodies) with native H2A (closed circle), glycated H2A (closed square), CML (closed triangle), and pentosidine (closed rhombus). Microtiter plates were coated with the above-mentioned immunogens (100 µg/ml), and then increasing concentrations of purified IgG were applied (for details see “Materials and Methods”).
Fig. 3. Characterization of antigenic specificity of the purified anti-glycated-H2A antibodies by competitive inhibition assay. Inhibition of anti-glycated-H2A antibody binding by glycated H2A (closed square), standard CML (closed triangle), and pentosidine (closed circle) as well as inhibition of binding of preimmune antibodies (closed rhombus) by all three inhibitors (depicted as average of glycated H2A, standard CML, and pentosidine) was determined. The microtiter plate was coated with glycated H2A (10 µg/ml).
These inhibition results reiterate that the induced antibodies are immunogen-specific. Induced antibodies also showed cross-reactivity with CML and pentosidine, a characteristic feature of polyspecific antibodies. The distinguishing feature of experimentally induced antibodies against glycated H2A was the preferential recognition of CML and pentosidine. CML and pentosidine are important AGEs that are formed predominantly on lysine and arginine residues in vivo as well as in vitro, thus acting as a major antigen in the immune system [38]. Our findings are in accordance with previous studies that have shown accumulation of CML and pentosidine in tissues in various diseases, specifically in diabetic complications, which serves as an immunological epitope [39, 40].
Native H2A used as an inhibitor of anti-glycated-H2A antibodies showed negligible inhibition of the antibody activity. Through the highly specific or the modified epitopes, the antibodies also identify the basic architecture of H2A.
Binding of anti-glycated-H2A IgG antibodies with an array of glycated proteins (or amino acids) and their native analogs is presented in Table 2. It can be noted that glycated inhibitors are preferentially bound by the experimentally produced antibodies against glycated H2A compared to their native analogs. Involvement of lysine and arginine, besides other factors, in the immunogenicity of glycated H2A is evident from the ability of glycated lysine and glycated arginine in inhibiting the antibody binding to the extent of 66.8 and 57.3% (at concentration of 20 µg/ml), respectively. Furthermore, induced antibodies showed cross-reactivity with CML, pentosidine, and other glycated proteins or amino acids, a distinguishing feature of polyspecific antibodies. The remarkable feature of the experimentally induced antibodies against glycated H2A was the preferential recognition of glycated proteins or amino acids compared to their native conformers. This suggests that glycated epitopes of various proteins and amino acids share common antigenic properties. Hence, glycated lysine and arginine are considered as favorite substrates for anti-glycated-H2A antibodies, which denote the involvement of lysine and arginine in generation of neoepitopes on H2A protein upon glycation. Moreover, induced antibodies also showed strong binding affinity towards CML and pentosidine, which signify CML and pentosidine as highly immunogenic in nature.
Table 2. Antigen binding specificity of
anti-glycated-H2A IgG antibodies
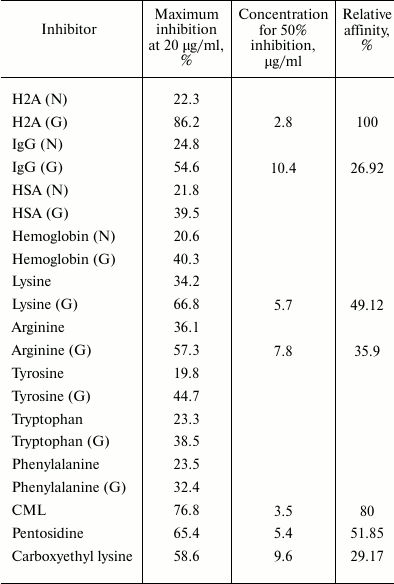
Note: N, native; G, glycated; HSA, human serum albumin. Antigenic
specificity of the induced anti-glycated-H2A IgG antibodies was
investigated by competitive inhibition ELISA. Microtiter plates were
coated with glycated H2A (10 µg/ml). The induced antibodies
exhibited wide range of heterogeneity in recognizing varied inhibitors.
A notable feature of experimentally produced antibodies against
glycated H2A was the preferential recognition of glycated proteins or
amino acids compared to their native conformers.
Based on the results, the immunological finding ascertains that glycation of H2A by 3-DG leads to structural alteration as well as development of AGEs that are responsible for the generation of neoepitopes on H2A, exposing it to be highly immunogenic and resulting in the assembly of antibodies. The antibodies produced, though highly specific for the immunogen, also exhibited polyspecific binding. In the case of humans, the polyspecific nature of antibodies may contribute to the etiology of many autoimmune diseases. AGE-H2A may be one of the decisive factors in antigen-driven induction of autoantibodies, which may serve as a marker in the early diagnosis/prognosis of diabetic nephropathy and therapies that prevent or control their progression, which might result in improved quality of life for patients.
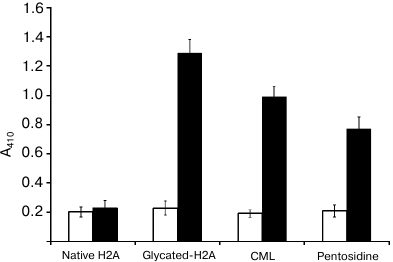
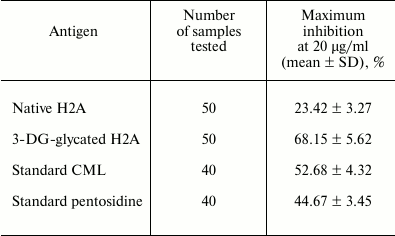
In further study, we aimed to detect circulating autoantibodies in type 2 diabetes sera against native and glycated H2A as well as against CML and pentosidine by direct-binding ELISA. The results are shown in Fig. 4. Glycated H2A displayed statistically significantly higher (p < 0.001) absorbance (1.288 ± 0.095) with type 2 diabetic sera compared to healthy subjects (0.228 ± 0.048). Moreover, CML and pentosidine also showed statistically significant (p < 0.001) absorbance of 0.989 ± 0.072 and 0.769 ± 0.083, respectively, compared to the healthy subjects (0.191 ± 0.025 and 0.209 ± 0.041). Whereas negligible binding of native H2A with healthy and type 2 diabetic subjects was observed, around 52% (57 of 110 samples), 43% (47 of 110 samples), and 38% (42 of 110 samples) of type 2 diabetic sera showed significantly enhanced binding with glycated H2A, CML, and pentosidine, respectively. Binding of autoantibodies in normal sera with coated antigens was similar. The purity of isolated antibodies was evaluated by SDS-PAGE under nonreducing conditions. The purified IgG migrated as a single band upon electrophoresis on a 10% polyacrylamide gel. The appearance of a single band indicates the purified IgG is highly pure (Fig. 5; inset). Furthermore, direct-binding ELISA of IgG purified from type 2 diabetic sera showed strong binding with all three antigens (glycated H2A, CML, pentosidine) (Fig. 5). Purified IgG from sera showing enhanced binding with antigens were further subjected to specificity analysis by inhibition ELISA (Table 3). The mean inhibition in the binding of IgG of type 2 diabetic sera with glycated H2A, CML, and pentosidine was 68.15 ± 5.62, 52.68 ± 4.32, and 44.67 ± 3.45%, respectively. Native H2A showed negligible inhibition of binding with the IgG (23.42 ± 3.27%). The results were significant at p < 0.001.
Fig. 4. Direct-binding ELISA of native H2A, glycated H2A, standard CML, and pentosidine with autoantibodies of normal human (open square) and diabetic (closed square) sera. Normal human serum served as control.
Fig. 5. Direct-binding ELISA of affinity purified IgG from diabetic sera with native H2A (closed circle), glycated H2A (closed square), CML (closed triangle), and pentosidine (closed rhombus). Microtiter plates were coated with respective immunogens (100 µg/ml) and then increasing concentrations of purified IgG were used and the direct-binding ELISA procedure was followed as described in “Materials and Methods”.
Table 3. Detection of autoantibodies against
various antigens in sera of type 2 diabetes mellitus patients
Earlier studies have demonstrated that increased serum levels of AGEs are pathological stimulus for generation of anti-AGE antibodies in patients with diabetes mellitus [41, 42] and are considered to play a contributory role in diabetic complications [43]. Our study substantiates previous studies that have reported the presence of anti-AGE antibodies in diabetic subjects having diabetic complications [41, 44, 45].
In conclusion, there is mounting evidence that suggests the presence of anti-AGE autoantibodies in diabetes and its complications. The character of these antibodies and which particular anti-AGE antibodies are relevant to the etiology of diabetic complications are yet to be established. The possibility of effective therapeutic intervention raised the importance of detecting anti-AGE antibodies, and progress in measuring anti-AGE antibodies using reliable methods will help determine the role they have in the pathogenesis of many diseases, especially diabetes and its complications. The binding pattern and the specificity that the anti-AGE antibody shows is rather high, suggesting its involvement in diabetic complications. Therefore, it suggests that these anti-AGE antibodies can be used as a biomarker for diagnosis and prognosis of the diabetes and its complications.
Acknowledgments
The authors thank to Jazan University (Saudi Arabia) and the Integral University (India) for providing instruments, facilities, and other items used for this study.
REFERENCES
1.Singh, R., Barden, A., Mori, T., and Beilin, L.
(2001) Advanced glycation end-products: a review, Diabetologia,
44, 129-146.
2.Njoroge, F. G., and Monnier, V. M. (1989) The
chemistry of the Maillard reaction under physiological conditions: a
review, Prog. Clin. Biol. Res., 304, 85-107.
3.Ahmad, S., Moinuddin, Dixit, K., Shahab, U., Alam,
K., and Ali, A. (2011) Genotoxicity and immunogenicity of DNA-advanced
glycation end products formed by methylglyoxal and lysine in presence
of Cu2+, Biochem. Biophys. Res. Commun.,
407, 568-574.
4.Brownlee, M. (1992) Glycation products and
pathogenesis of diabetic complications, Diabetes Care,
15, 1835-1843.
5.Bucala, R., Cerami, A., and Vlassara, H. (1995)
Advanced glycosylation end products in diabetic complications,
Diabetes Rev., 3, 258-268.
6.Chappey, O., Dosquet, C., Wautier, M. P., and
Wautier, J. L. (1997) Advanced glycation end-products, oxidant stress
and vascular lesions, Eur. J. Clin. Invest., 27,
97-108.
7.Uribarri, J., Cai, W., Peppa, M., Goodman, S.,
Ferucci, L., Striker, G., and Vlassara, H. (2007) Circulating
glycotoxins and dietary advanced glycation end products: two links to
inflammatory response, oxidative stress and aging, J. Gerontol. A
Biol. Sci. Med. Sci., 62, 427-433.
8.Park, L., Raman, K. G., Lee, K. J., Lu, Y., Ferran,
L. J., Jr., Chow, W. S., Stern, D., and Schmidt, A. M. (1998)
Suppression of accelerated diabetic atherosclerosis by the soluble
receptor for advanced glycation end products, Nat. Med.,
4, 1025-1031.
9.McLellan, A. C., Thornalley, P. J., Benn, J., and
Sonksen, P. H. (1994) Glyoxalase system in clinical diabetes mellitus
and correlation with diabetic complications, Clin. Sci.,
87, 21-29.
10.Oya, T., Hattori, N., Mizuno, Y., Miyata, S.,
Maeda, S., Osawa, T., and Uchida, K. (1999) Methylglyoxal modification
of protein. Chemical and immunochemical characterization of
methylglyoxal-arginine adducts, J. Biol. Chem., 274,
18492-18502.
11.Frye, E. B., Degenhardt, T. P., Thorpe, S. P.,
and Baynes, J. B. (1998) Role of the Maillard reaction in aging of
tissue proteins. Advanced glycation end product-dependent increase in
imidazolium cross-links in human lens proteins, J. Biol. Chem.,
273, 18714-18719.
12.Thornalley, P. J., Langborg, A., and Minhas, H.
S. (1999) Formation of glyoxal, methylglyoxal and 3-deoxyglucosone in
the glycation of proteins by glucose, Biochem. J., 344,
109-116.
13.Choudhary, D., Chandra, D., and Kale, R. K.
(1997) Influence of methylglyoxal on antioxidant enzymes and oxidative
damage, Toxicol. Lett., 93, 141-152.
14.Ashraf, J. M., Arif, B., Dixit, K., Moinuddin,
and Alam, K. (2012) Physicochemical analysis of structural changes in
DNA modified with glucose, Int. J. Biol. Macromol., 51,
604-611.
15.Festa, A., Schmolzer, B., Schernthaner, G., and
Menzel, E. J. (1998) Differential expression of receptors for advanced
glycation end-products on monocytes in patients with IDDM,
Diabetologia, 41, 674-680.
16.Wolffe, A. (1998) Chromatin. Structure and
Function, Academic Press, San Diego, p. 447.
17.Liebich, H. M., Gesele, E., Wirth, C., Woll, J.,
Jobst, K., and Lakatos, A. (1992) Non-enzymatic glycation of histones,
Biol. Mass Spectrom., 22, 121-123.
18.Gugliucci, A. (1994) Advanced glycation of rat
liver histone octamers: an in vitro study, Biochem. Biophys. Res.
Commun., 203, 588-593.
19.Cervantes-Laurean, D., Jacobson, E. L., and
Jacobson, M. K. (1996) Glycation and glycoxidation of histones by
ADP-ribose, J. Biol. Chem., 271, 10461-10469.
20.Gugliucci, A., and Bendayan, M. (1995) Histones
from diabetic rats contain increased levels of advanced glycation end
products, Biochem. Biophys. Res. Commun., 212, 56-62.
21.Bojanovic, J. J., Jevtovic, A. D., Pantic, V. S.,
Dugandzic, S. M., and Jovanovic, D. S. (1970) Thymus nucleoproteins.
Thymus histones in young and adult rats, Gerontologia (Basel),
16, 304-312.
22.Turk, Z., Ljubica, S., Turk, N., and Benko, B.
(2001) Detection of autoantibodies against advanced glycation end
products and AGE-immune complexes in serum of patients with diabetes
mellitus, Clin. Chim. Acta, 303, 105-115.
23.Makino, H., Shikata, K., Hironaka, K., Kushiro,
M., Yamasaki, Y., Sugimoto, H., Ota, Z., Araki, N., and Horiuchi, S.
(1995) Ultrastructure of nonenzymatically glycated mesangial matrix in
diabetic nephropathy, Kid. Int., 48, 517-526.
24.Ansari, N. A., Moinuddin, Alam, K., and Ali, A.
(2009) Preferential recognition of Amadori-rich lysine residues by
serum antibodies in diabetes mellitus: role of protein glycation in the
disease process, Hum. Immunol., 70, 417-424.
25.Ansari, N. A., and Dash, D. (2013) Biochemical
studies on methylglyoxal mediated glycated histones: implications for
presence of serum antibodies against the glycated histones in patients
with type 1 diabetes mellitus, ISRN Biochem., 198065.
26.Ashraf, J. M., Ahmad, S., Rabbani, G., Jan, A.
T., Lee, E. J., Khan, R. H., and Choi, I. (2014) Physicochemical
analysis of structural alteration and advanced glycation end products
generation during glycation of H2A histone by 3-deoxyglucosone,
IUBMB Life, 66, 686-693.
27.Ashraf, J. M., Arfat, M. Y., Arif, Z., Ahmad, J.,
Moinuddin, and Alam, K. (2015) A clinical correlation of anti-DNA-AGE
autoantibodies in type 2 diabetes mellitus with disease duration,
Cell Immunol., 293, 74-79.
28.Shahab, U., Moinuddin, Ahmad, S., Dixit, K., and
Ali, A. (2013) Genotoxic effect of N-hydroxy-4-acetylaminobiphenyl on
human DNA: implications in bladder cancer, PLoS One, 8,
e53205.
29.Moinuddin, Dixit, K., Ahmad, S., Shahab, U.,
Alam, K., and Ali, A. (2014) Human DNA damage by the synergistic action
of 4-aminobiphenyl and nitric oxide: an immunochemical study,
Environ. Toxicol., 29, 568-576.
30.Alam, K., Moinuddin, and Jabeen, S. (2007)
Immunogenicity of mitochondrial DNA modified by hydroxyl radical,
Cell Immunol., 247, 12-17.
31.Ashraf, J. M., Rabbani, G., Ahmad, S., Hasan, Q.,
Khan, R. H., Alam, K., and Choi, I. (2015) Glycation of H1 histone by
3-deoxyglucosone: effects on protein structure and generation of
different advanced glycation end products, PLoS One,
10, e0130630.
32.Ashraf, J. M., Ahmad, S., Rabbani, G., Hasan, Q.,
Jan, A. T., Lee, E. J., and Choi, I. (2015) 3-Deoxyglucosone: a
potential glycating agent accountable for structural alteration in H3
histone protein through generation of different AGEs, PLoS One,
10, e0116804.
33.Arfat, M. Y., Ashraf, J. M., Arif, Z., Moinuddin,
and Alam, K. (2014) Fine characterization of glucosylated human IgG by
biochemical and biophysical methods, Int. J. Biol.
Macromol., 69, 408-415.
34.Akhter, F., Khan, M. S., Singh, S., and Ahmad, S.
(2014) An immunohistochemical analysis to validate the rationale behind
the enhanced immunogenicity of D-ribosylated low-density lipoprotein,
PLoS One, 9, e113144.
35.Turk, Z., Ljubica, S., Turk, N., and Benko, B.
(2001) Detection of auto-antibodies against advanced glycation end
products and AGE-immune complexes in serum of patients with diabetes
mellitus, Clin. Chim. Acta, 303, 105-115.
36.Mustafa, I., Ahmad, S., Dixit, K., Moinuddin,
Ahmad, J., and Ali, A. (2012) Glycated human DNA is a preferred antigen
for anti-DNA antibodies in diabetes mellitus patients, Diabetes Res.
Clin. Prac., 95, 98-104.
37.Nagai, R., Fujiwara, Y., Mera, K., Yamagata, K.,
Sakashita, N., and Takeya, M. (2008) Immunochemical detection of N
epsilon-(carboxyethyl)lysine using a specific antibody, J. Immunol.
Methods, 332, 112-120.
38.Ikeda, K., Higashi, T., Sano, H., Jinnouchi, Y.,
Yoshida, M., Araki, T., Ueda, S., and Horiuchi, S. (1996)
Nε-(carboxymethyl)lysine protein adduct is a major
immunological epitope in proteins modified with advanced glycation end
products of the Maillard reaction, Biochemistry, 35,
8075-8083.
39.Reddy, S., Bichler, J., Wells-Knecht, K. J.,
Thorpe, S. R., and Baynes, J. W. (1995)
Nε-(carboxymethyl)lysine is a dominant advanced
glycation end product (AGE) antigen in tissue proteins,
Biochemistry, 34, 10872-10878.
40.Shibayama, R., Araki, N., Nagai, R., and
Horiuchi, S. (1999) Autoantibody against
Nε-carboxymethyllysine: an advanced glycation end
product of the Maillard reaction, Diabetes, 48,
1842-1829.
41.Ahmad, S., Moinuddin, Habib, S., Shahab, U.,
Alam, K., and Ali, A. (2014) Autoimmune response to AGE modified human
DNA: implications in type 1 diabetes mellitus, J. Clin. Trans
Endo, 1, 66-72.
42.Ahmad, S. (2014) Immunogenicity of DNA damage by
free radicals and carbonyls: a probable bio-marker for the auto-immune
diseases, J. Immun. Res., 1, 2.
43.Shahab, U., Tabrez, S., Khan, M. S., Akhter, F.,
Khan, M. S., Saeed, M., Ahmad, K., Srivastava, A. K., and Ahmad, S.
(2014) Immunogenicity of DNA-advanced glycation end product fashioned
through glyoxal and arginine in the presence of Fe3+: its
potential role in prompt recognition of diabetes mellitus
auto-antibodies, Chem. Biol. Int., 219,
229-240.
44.Ahmad, S., Moinuddin, Shahab, U., Khan, M. S.,
Habeeb, S., Alam, K., and Ali, A. (2014) Glyco-oxidative damage to
human DNA-neo-antigenic epitopes on DNA molecule could be a possible
reason for autoimmune response in type 1 diabetes, Glycobiology,
24, 281-291.
45.Akhter, F., Khan, M. S., Abdulrahman, A. A.,
Faisal, M., and Ahmad, S. (2016) Antigenic role of the adaptive immune
response to D-ribose glycated LDL in diabetes, atherosclerosis and
diabetic atherosclerosis patients, Life Sci., 151,
139-146.