REVIEW: Mesoporous Silica Nanoparticles as a Carrier Platform for Intracellular Delivery of Nucleic Acids
N. A. Keasberry1, C. W. Yapp2, and A. Idris2*
1University Brunei Darussalam, Faculty of Science, Brunei Darussalam2PAPRSB Institute of Health Sciences, University Brunei Darussalam, Brunei Darussalam; E-mail: adi.diris@gmail.com
* To whom correspondence should be addressed.
Received November 29, 2016; Revision received February 2, 2017
Virus-mediated gene delivery has been, to date, the most successful and efficient method for gene therapy. However, this method has been challenged because of serious safety concerns. Over the past decade, mesoporous silica nanoparticles (MSNs) have attracted much attention for intracellular delivery of nucleic acids. Delivery of cellular plasmid DNA (pDNA) is designed to replace the function of a defective gene and restore its normal function in the cell. Delivery of small interfering RNAs (siRNAs) can selectively knockdown genes by targeting specific mRNAs. The biocompatibility and unique structures of MSNs make these nanoparticles ideal candidates to act as biomolecule carriers. This concise review highlights current progress in the field of nucleic acid delivery using MSNs, specifically for delivery of pDNA, siRNA, and combinatorial delivery of nucleic acids and drugs. The review describes important design parameters presently being applied to MSNs to administer drugs and therapeutic nucleic acids.
KEY WORDS: silica nanoparticles, siRNA, pDNA, MSN, intracellular, gene therapyDOI: 10.1134/S0006297917060025
Inorganic nanoparticles such as iron oxide and silica have been extensively studied for a variety of biological applications. These include use as clinical agents in imaging based detection, diagnosis, and treatment of diseases. Silica nanoparticles have gained much attention in biomedical applications because it is relatively easy to tune their physicochemical properties for specific purposes. For example, size, shape, and porosity can be easily controlled, while surface modifications can be introduced to alter the chemical properties of silica nanoparticles [1]. Silica nanoparticles have high biological stability and low toxicity, and the exposed silanol groups on their surface enable versatile functionalization and modifications [2]. Silica nanoparticles are divided into two major categories, solid and mesoporous silica nanoparticles (MSNs). The different types of silica nanoparticles, for example solid, mesoporous, shaped, etched, and hollow, are synthesized via different methods [3, 4]. MSNs show potential as biomolecule delivery vehicles because of their unique mesoporous structure and physical properties such as high surface area, large pore volume, tunable pore diameter, and narrow size distribution [5]. Importantly, MSNs have low immunogenicity, minimizing unfavorable inflammatory reactions [6]. Several reports have described the synthesis of MSNs, including by, for example, liquid crystal templating, evaporation-induced self-assembly, and sol–gel [7-9]. The major advantage of using synthetic MSNs is that their surface can be chemically modified to enhance their biomolecule cargo-carrying and other functional capacities [10].
MSNs have been used in a wide range of applications, most notably for bioimaging and drug delivery [11]. Furthermore, the cellular mechanisms enabling drug delivery by MSNs have been well studied. They involve stimuli-responsive triggers such as pH [12], redox-activation or redox-modulated motifs [13-15], and supramolecular switches [16] to enable selective and sustained drug delivery into the cell. Most studies to date have utilized doxorubicin [17-20] rather than other drugs such as desipramine, camptothecin, itraconazole, and ibuprofen [21-24] as a model anti-cancer molecule for studying MSN-mediated drug delivery into cells. In addition, combinatorial delivery of multiple drugs using MSNs has been demonstrated [25, 26].
There is growing evidence that MSNs can be used as nucleic acid carriers for gene therapy. MSNs, unlike other nanoparticles, possess the ideal qualities to serve as nucleic acid carriers. Several reviews have discussed the use of MSNs as a drug delivery platform in extensive detail [5, 11, 27-29]. With the growing body of evidence addressing use of MSNs for nucleic acid delivery, a review of this topic deserves special attention. Therefore, our concise review discusses current advances in the use of MSNs for gene delivery.
MSNs: A POTENTIAL INTRACELLULAR NUCLEIC ACID DELIVERY
PLATFORM
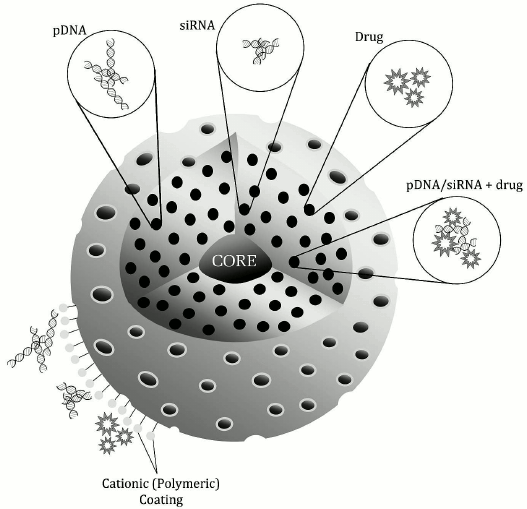
Gene delivery introduces foreign nucleic acids into target cells solely for therapy. Plasmid DNAs (pDNAs) bearing essential genes to correct the function of a target gene (e.g. an oncogenic gene) have been used for gene therapy to treat certain diseases. Small interfering RNAs (siRNAs) are attractive as anti-cancer therapies because they can alter expression of activated oncogenes, cell cycle regulators, or proapoptotic genes, all potentially critical for tumorigenesis or tumor survival. Several studies have described attempts to load nucleic acids into MSN preparations for delivery into cells (figure).
Schematic representation of the general features of MSNs for nucleic acid and drug delivery. The surface is typically covered with cationic polymers or other molecules containing positively charged terminal functional groups to complex the nucleic acid. The pores of MSNs can hold nucleic acids, drugs, or a combination of both
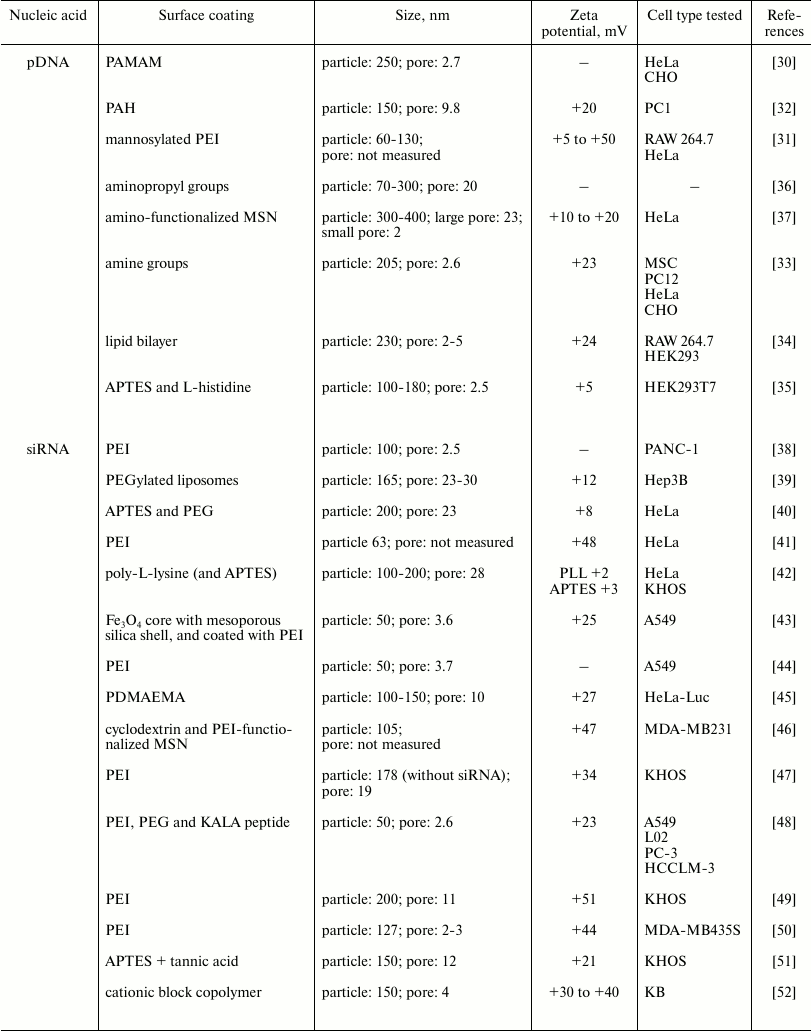
pDNA delivery. Intracellular delivery of pDNA enhances expression of therapeutic genes by using a strong constitutive promoter to replace the defective gene and restore normal function. Several studies have investigated the feasibility of using monodispersed MSNs loaded with pDNA for delivery into cells (Table 1). Early work by Radu et al. [30] showed that a polyamidoamine (PAMAM) dendrimer coating provided a positively charged surface for complexing to DNA, while leaving the pores free to encapsulate drugs and dyes. This study also showed that MSNs were not toxic and that they protected the DNA against enzymatic cleavage. Other polymers including polyethyleneimine (PEI) [31] and poly(allylamine hydrochloride) (PAH) [32] are also excellent coatings to promote complexation of DNA on the MSN surface. Both cationic polymeric coatings enhanced DNA loading onto MSNs and transfection efficiency. These MSNs have pore sizes of less than 10 nm, and the smaller pore sizes cannot usually accommodate larger DNA molecules. Therefore, cationic surfaces, which help bind the negatively charged DNA, are introduced through positively charged terminal groups of polymers coating the nanoparticle surface. Other ways to introduce a positive charge on the MSN surface are to conjugate a small molecule or lipid containing terminal amine groups. Kim et al. [33] used aminopropyl-triethoxysilane (APTES) to impart a positive charge through its amine group, while Dengler et al. [34] used positively charged lipid bilayers. The surface charges for each system were +23 and +24 mV, respectively, and both systems had similar particle and pore sizes (200-230 nm particles, 2-5 nm pores). Similarly, both studies showed promising cellular transfection in applications related to stem cell and spinal cord therapy. Alternatively, histidine was used to functionalize MSNs [35]. Even though the resulting particles showed lower uptake in vivo, there was good internalization in vitro compared with that of amino-functionalized MSNs. Some studies have shown that a larger pore size (~up to 20 nm) could also improve DNA loading capacity, independent of cationic polymer coating [36, 37]. When compared to MSNs with smaller pores, the larger pore MSNs were shown to completely incorporate plasmids, while smaller pores had pDNA piled onto the MSN surfaces [36]. Another study showed that loading of pDNA into pores was enhanced for a larger pore MSN compared to those with smaller pores, even at a lower concentration of the large pore MSN [37]. While no particular surface charge has been shown to be the ideal, there is a consensus that a positive charge (at least +5 to +30 mV zeta potential before DNA complexation) is necessary for ensuring efficient encapsulation and delivery of DNA, even when the pore size is small.
Table 1. Particle and pore sizes and surface
coatings in nucleic acid delivery systems with MSNs
siRNA delivery. siRNAs can selectively knock down target genes by targeting specific mRNAs. Many studies of siRNA delivery with MSNs used cationic polymeric coatings, such as PEI, similar to those used in DNA delivery studies (Table 1). Hom et al. [38] showed that siRNA was attached to the positively charged PEI external surface of MSNs via electrostatic interactions. This study also showed that the PEI coating promoted endosomal escape of the siRNA into the cytosol, necessary for much more effective target gene knockdown efficiency, in pancreatic cancer cells. Another group also used PEI-coated MSNs for siRNA delivery into breast cancer cells [50]. They observed reduced tumorigenic activity as a result of intracellular siRNA mediated knockdown of the target, TWIST 1, a transcription factor regulating angiogenesis. PEI can also be functionalized with other molecules prior to attachment to MSNs, including acetaldehyde-cysteine (AC) [47] or cyclodextrin [46]. AC-PEI coating enables controlled release of the siRNA inside cells via a GSH-triggered disulfide bond cleavage of the AC-PEI once inside the cytoplasm where GSH is present in much higher concentrations than outside the cell. This AC moiety provides a structural response to the intracellular microenvironment. In addition, it is autofluorescent, enabling MSNs to be tracked intracellularly [47]. The cyclodextrin moiety in cyclodextrin-PEI-coated MSNs reduced the charge-induced cytotoxicity of PEI for breast cancer cells [46]. Furthermore, cyclodextrin-PEI-coated MSNs showed good endosomal escape with good particle retention within the tumor in vivo [46]. In these studies, there appears to be no correlation between pore size and surface charge against efficacy of gene encapsulation and delivery, although in general these factors as well as pore morphology do play some role in adsorption and desorption of siRNA into the MSNs. In a recent study by Möller et al., even medium-sized pores (4 nm) with highly positively charged internal pore surfaces gave exceptionally high siRNA loadings [52].
Addition of a targeting peptide such as KALA, a cationic amphipathic cell-penetrating peptide, to the MSN surface produced enhanced endosomolytic function. Li et al. [43] and Chen et al. [48] synthesized MSNs coated with PEI and then subsequently conjugated to KALA peptides. Even with the small MSN pores (<5 nm, comparable to siRNA size), the highly positive surface charges (+25 mV [43] and +23.6 mV [48]) allowed for siRNA complexation and resulted in excellent delivery into target cells, with effective gene silencing and tumor inhibition.
Other surface coatings have also been explored as alternatives to PEI. Non-polymeric coatings such as APTES provide positively charged amine groups, and MSNs coated with APTES or polyethylene glycol (PEG) promoted efficient siRNA delivery into HeLa cells [40]. In these particles, most of the siRNA was encapsulated within the large MSN pores (23 nm), with PEG also added to minimize agglomeration. These MSNs, however, could be administered only intratumorally because of their tendencies to aggregate in vivo. siRNA is susceptible to degradation before reaching its intended target, and so should be protected. One way to do this has been by the addition of a tannic acid coating which covers the pores, but is pH responsive and only releases the siRNA when in the acidic conditions of the cytoplasm [51]. Successful delivery into the cytoplasm of KHOS cells in vitro was observed. Poly-2-dimethylaminoethyl methacrylate (PDMAEMA) is another polymer alternative that was demonstrated to enhance transfection efficiency. Though PDMAEMA is cytotoxic on its own, its toxicity is reduced when it is stably associated with MSNs [45]. However, the PDMAEMA-coated MSNs have poorer intracellular endosomal escape in HeLa cells compared with the commercial transfection agent Lipofectamine 2000. Another study compared DNA loading capacities of two functionalized sets of MSNs, one with low molecular weight poly-L-lysine (PLL) and the other with APTES [42]. Both had nearly equal DNA loading capacities even though the PLL had fewer amine groups, therefore a lower positive charge, than APTES (+1.90 mV for PLL compared to +3.23 mV for APTES). The DNA loading capacity of the PLL–MSNs was attributed to the existence of two types of interactions between PLL and DNA – electrostatic and specific binding of PLL to the adenine-thymine (A-T) sequence – compared with only the electrostatic interaction in APTES. Interactions between PLL and the cell surface were also favorable in HeLa and KHOS cells, leading to better cellular uptake and internalization and, hence, enhanced siRNA delivery. There is perhaps a relationship between larger pore size and the ability to utilize polymers that result in lower surface charge.
PEGylated liposomes have also been used to coat MSNs after encapsulating siRNA in the pores (23-30 nm), producing good transfection efficiency in Hep3B cells [39]. MSNs were synthesized with magnetic cores, forming core-shell or yolk-shell structures. The magnetic cores enabled controlled delivery of siRNAs to target sites guided by an external magnetic field. Several studies showed that, by synthesizing magnetic iron oxide cores with a mesoporous silica shell coated with PEI, successful intracellular siRNA delivery and efficient endosomal escape was achieved in HeLa [41], A549 [44], and KHOS [49] cells. All these examples demonstrate that positive charge is necessary for gene complexation, but more studies are required on optimal pore size.
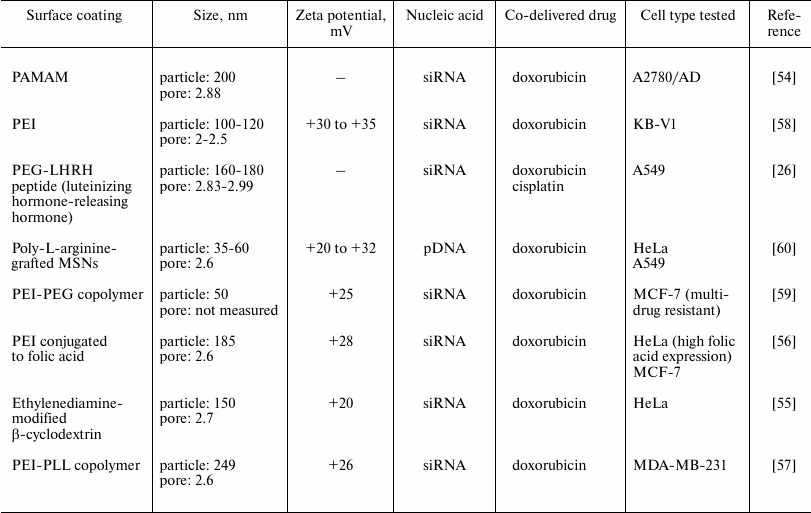
Co-delivery of nucleic acids and drugs. In recent years, there has been remarkable progress using MSNs as combinatorial delivery systems. With MSNs, nucleic acids have been delivered together with drugs to improve effectiveness of the drug being delivered, as well as to maximize delivery of the drug into the cell (Table 2). The traditional method of drug/gene delivery involved injection of a gene-carrying virus combined with administration of a therapeutic drug directly into the tumor. However, this usually resulted in low drug efficacy because of differences in the pharmacokinetics of the small molecule drug and the nucleic acid. MSNs are highly advantageous for such co-delivery systems because of their large surface area, well-established surface chemistry facilitating surface modifications, and porous structures enabling encapsulation of both drugs and nucleic acids [53]. An early example of co-delivery of a drug/nucleic acid combination was described by Chen et al. [54]. They evaluated delivery of doxorubicin, an apoptosis-inducing agent, along with a siRNA targeted at Bcl-2, a gene regulating apoptotic cell death, into A2780/AD ovarian cancer cells. Bcl-2 siRNA was coupled to PAMAM-coated MSNs via electrostatic interactions between the negatively charged RNA and the positively charged dendrimer, while doxorubicin was loaded into the pores of the MSN. When the co-delivery system was tested in cells, doxorubicin co-delivered with siRNA was more cytotoxic than that delivered without siRNA. Similar findings were observed in other cell types including HeLa [55, 56], A549 [26], and MDA-MB-231 [57] cells, also using Bcl-2-targeted siRNA and doxorubicin in combination, albeit with slight differences to polymer/coatings. Furthermore, in in vivo biodistribution studies, Taratula et al. [26] showed that the MSNs, when inhaled, were localized and retained in the lungs of mice. Other siRNA types were tested in combination with doxorubicin [58, 59]. P-glycoprotein (Pgp) siRNA with doxorubicin was utilized to overcome multidrug resistance in two types of cancer cells, KB-V1 [58] and MCF-7 [59]. In both studies, PEI was the cationic polymer coating selected for siRNA complexation, whereas the pores were coated with phosphonate to electrostatically bind doxorubicin within them. These MSNs produced efficient intracellular co-delivery of doxorubicin and siRNA. Like previous examples of systems delivering nucleic acids only, alternative coatings were investigated for combined delivery systems. Kar et al. [60] grafted poly-L-arginine to the surface of MSNs to complex pDNA, with doxorubicin in the pores. Not only does poly-L-arginine impart a positive charge to complex DNA, but also MSNs prepared in this manner showed excellent cell penetration in both HeLa and A549 cells. In general, these studies showed that the drugs are encapsulated in the pores (which were small, <5 nm), and the genetic material complexed to the surface of the particle through positively charged groups/polymers.
Table 2. Particle and pore sizes and surface
coatings in systems for co-delivery of drugs with nucleic acids using
MSNs
Current research advances make nucleic acid delivery into cells using MSNs a foreseeable possibility. Furthermore, combination therapies delivering both nucleic acids and drugs into target cells showed enhanced therapeutic effects, making this a strategy of increasing interest for cancer therapy. Most of the studies to date have used doxorubicin as the model drug agent and, overall, the efficiency of MSN-mediated intracellular delivery of drugs was very high. The clinical value of MSNs for delivering therapeutic nucleic acids in combination with different chemotherapeutic drugs warrants continued investigation. Results so far indicate that, indeed, combination delivery of nucleic acids and drugs using MSNs has advantages compared with delivering either agent alone for inhibiting tumor growth. For example, the co-delivery of chemotherapeutic drugs and therapeutic nucleic acids is a potential approach to overcome drug resistance in cancer therapy. With increasingly successful research employing anti-cancer agents, future studies should explore the possibility of using MSNs to treat other disorders besides cancer, such as autoimmune and metabolic diseases. Study design should focus on the rational selection of nucleic acid and drug ratios, including the interactions of the two agents within MSNs. These factors have often been overlooked. Clarifying this selection process would allow optimization of combinatorial formulations to achieve maximum therapeutic efficiencies. In conclusion, MSNs have the potential to be an ideal platform for the efficient, safe delivery of nucleic acids and drugs into patients.
REFERENCES
1.Slowing, I. I., Trewyn, B. G., Giri, S., and Lin,
V. S.-Y. (2007) Mesoporous silica nanoparticles for drug delivery and
biosensing applications, Adv. Funct. Mater., 17,
1225-1236.
2.Vogt, C., Toprak, M. S., Muhammed, M., Laurent, S.,
Bridot, J.-L., and Muller, R. N. (2009) High quality and tuneable
silica shell–magnetic core nanoparticles, J. Nanopart.
Res., 12, 1137-1147.
3.Tang, L., and Cheng, J. (2013) Nonporous silica
nanoparticles for nanomedicine application, Nano Today,
8, 290-312.
4.Liberman, A., Mendez, N., Trogler, W. C., and
Kummel, A. C. (2014) Synthesis and surface functionalization of silica
nanoparticles for nanomedicine, Surf. Sci. Rep., 69,
132-158.
5.Bharti, C., Nagaich, U., Pal, A. K., and Gulati, N.
(2015) Mesoporous silica nanoparticles in target drug delivery system:
a review, Int. J. Pharm. Invest., 5, 124-133.
6.Heidegger, S., Gossl, D., Schmidt, A., Niedermayer,
S., Argyo, C., Endres, S., and Bourquin, C. (2015) Immune response to
functionalized mesoporous silica nanoparticles for targeted drug
delivery, Nanoscale, 8, 938-948.
7.Tang, F., Li, L., and Chen, D. (2012) Mesoporous
silica nanoparticles: synthesis, biocompatibility and drug delivery,
Adv. Mater., 24, 1504-1534.
8.Wu, S.-H., Hung, Y., and Mou, C.-Y. (2011)
Mesoporous silica nanoparticles as nanocarriers, Chem. Commun.,
47, 9972-9985.
9.Wu, S.-H., Mou, C.-Y., and Lin, H.-P. (2013)
Synthesis of mesoporous silica nanoparticles, Chem. Soc. Rev.,
42, 3862-3875.
10.Slowing, I. I., Vivero-Escoto, J. L., Wu, C.-W.,
and Lin, V. S.-Y. (2008) Mesoporous silica nanoparticles as controlled
release drug delivery and gene transfection carriers, Adv. Drug
Deliv. Rev., 60, 1278-1288.
11.Wang, Y., Zhao, Q., Han, N., Bai, L., Li, J.,
Liu, J., and Wang, S. (2015) Mesoporous silica nanoparticles in drug
delivery and biomedical applications, Nanomed. Nanotechnol. Biol.
Med., 11, 313-327.
12.Li, Q.-L., Xu, S.-H., Zhou, H., Wang, X., Dong,
B., Gao, H., and Yang, Y.-W. (2015) pH and glutathione dual-responsive
dynamic cross-linked supramolecular network on mesoporous silica
nanoparticles for controlled anticancer drug release, ACS Appl.
Mater. Interfaces, 7, 28656-28664.
13.Sun, L., Liu, Y.-J., Yang, Z.-Z., and Qi, X.-R.
(2015) Tumor specific delivery with redox-triggered mesoporous silica
nanoparticles inducing neovascularization suppression and vascular
normalization, RSC Adv., 5, 55566-55578.
14.Zhao, N., Lin, X., Zhang, Q., Ji, Z., and Xu,
F.-J. (2015) Redox-triggered gatekeeper-enveloped starlike hollow
silica nanoparticles for intelligent delivery systems, Small,
11, 6467-6479.
15.Yoshitomi, T., and Nagasaki, Y. (2015)
Development of silica-containing redox nanoparticles for medical
applications, Biomater. Sci., 3, 810-815.
16.Song, N., and Yang, Y.-W. (2015) Molecular and
supramolecular switches on mesoporous silica nanoparticles, Chem.
Soc. Rev., 44, 3474-3504.
17.Shahabi, S., Doscher, S., Bollhorst, T.,
Treccani, L., Maas, M., Dringen, R., and Rezwan, K. (2015) Enhancing
cellular uptake and doxorubicin delivery of mesoporous silica
nanoparticles via surface functionalization: effects of serum, ACS
Appl. Mater. Interfaces, 7, 26880-26891.
18.Gabizon, A., Shmeeda, H., and Grenader, T. (2012)
Pharmacological basis of pegylated liposomal doxorubicin: impact on
cancer therapy, Eur. J. Pharm. Sci., 45, 388-398.
19.Giménez, C., De la Torre, C., Gorbe, M.,
Aznar, E., Sancenon, F., Murguía, J. R., and Amoros, P. (2015)
Gated mesoporous silica nanoparticles for the controlled delivery of
drugs in cancer cells, Langmuir, 31, 3753-3762.
20.Qu, Q., Ma, X., and Zhao, Y. (2015) Targeted
delivery of doxorubicin to mitochondria using mesoporous silica
nanoparticle nanocarriers, Nanoscale, 7, 16677-16686.
21.Chang, D., Gao, Y., Wang, L., Liu, G., Chen, Y.,
Wang, T., and Zeng, X. (2015) Polydopamine-based surface modification
of mesoporous silica nanoparticles as pH-sensitive drug delivery
vehicles for cancer therapy, J. Coll. Inter. Sci., 463,
279-287.
22.Lu, J., Liong, M., Zink, J. I., and Tamanoi, F.
(2007) Mesoporous silica nanoparticles as a delivery system for
hydrophobic anticancer drugs, Small, 3, 1341-1346.
23.Mellaerts, R., Jammaer, J. A. G., Van Speybroeck,
M., Chen, H., Van Humbeeck, J., Augustijns, P., and Martens, J. A.
(2008) Physical state of poorly water soluble therapeutic molecules
loaded into SBA-15 ordered mesoporous silica carriers: a case study
with itraconazole and ibuprofen, Langmuir, 24,
8651-8659.
24.Van Speybroeck, M., Barillaro, V., Thi, T. Do,
Mellaerts, R., Martens, J., Van Humbeeck, J., and Augustijns, P. (2009)
Ordered mesoporous silica material SBA-15: a broad-spectrum formulation
platform for poorly soluble drugs, J. Pharm. Sci., 98,
2648-2658.
25.Wang, H., Agarwal, P., Zhao, S., Yu, J., Lu, X.,
and He, X. (2015) A biomimetic hybrid nanoplatform for encapsulation
and precisely controlled delivery of therasnostic agents, Nat.
Commun., 6, 10081.
26.Taratula, O., Garbuzenko, O. B., Chen, A. M., and
Minko, T. (2011) Innovative strategy for treatment of lung cancer:
targeted nanotechnology-based inhalation co-delivery of anticancer
drugs and siRNA, J. Drug Target., 19, 900-914.
27.Mamaeva, V., Sahlgren, C., and Linden, M. (2013)
Mesoporous silica nanoparticles in medicine – recent advances,
Adv. Drug Deliver. Rev., 65, 689-702.
28.Li, Z., Barnes, J. C., Bosoy, A., Stoddart, J.
F., and Zink, J. I. (2012) Mesoporous silica nanoparticles in
biomedical applications, Chem. Soc. Rev., 41,
2590-2605.
29.Baeza, A., Colilla, M., and Vallet-Regí,
M. (2015) Advances in mesoporous silica nanoparticles for targeted
stimuli-responsive drug delivery, Expert Opin. Drug Deliv.,
12, 319-337.
30.Radu, D. R., Lai, C.-Y., Jeftinija, K., Rowe, E.
W., Jeftinija, S., and Lin, V. S.-Y. (2004) A polyamidoamine
dendrimer-capped mesoporous silica nanosphere-based gene transfection
reagent, J. Am. Chem. Soc., 126, 13216-13217.
31.Park, I. Y., Kim, I. Y., Yoo, M. K., Choi, Y. J.,
Cho, M.-H., and Cho, C. S. (2008) Mannosylated polyethylenimine coupled
mesoporous silica nanoparticles for receptor-mediated gene delivery,
Int. J. Pharm., 359, 280-287.
32.Qin, F., Zhou, Y., Shi, J., and Zhang, Y. (2009)
A DNA transporter based on mesoporous silica nanospheres mediated with
polycation poly(allylamine hydrochloride) coating on mesopore surface,
J. Biomed. Mater. Res. Part A, 90, 333-338.
33.Kim, T.-H., Kim, M., Eltohamy, M., Yun, Y.-R.,
Jang, J.-H., and Kim, H.-W. (2013) Efficacy of mesoporous silica
nanoparticles in delivering BMP-2 plasmid DNA for in vitro osteogenic
stimulation of mesenchymal stem cells, J. Biomed. Mater. Res. Part
A, 101, 1651-1660.
34.Dengler, E. C., Liu, J., Kerwin, A., Torres, S.,
Olcott, C. M., Bowman, B. N., and Milligan, E. D. (2013) Mesoporous
silica-supported lipid bilayers (protocells) for DNA cargo delivery to
the spinal cord, J. Control. Rel., 168, 209-224.
35.Brevet, D., Hocine, O., Delalande, A., Raehm, L.,
Charnay, C., Midoux, P., and Pichon, C. (2014) Improved gene transfer
with histidine-functionalized mesoporous silica nanoparticles, Int.
J. Pharm., 471, 197-205.
36.Gao, F., Botella, P., Corma, A., Blesa, J., and
Dong, L. (2009) Monodispersed mesoporous silica nanoparticles with very
large pores for enhanced adsorption and release of DNA, J. Phys.
Chem., 113, 1796-1804.
37.Kim, M.-H., Na, H.-K., Kim, Y.-K., Ryoo, S.-R.,
Cho, H. S., Lee, K. E., and Min, D.-H. (2011) Facile synthesis of
monodispersed mesoporous silica nanoparticles with ultralarge pores and
their application in gene delivery, ACS Nano, 5,
3568-3576.
38.Hom, C., Lu, J., Liong, M., Luo, H., Li, Z.,
Zink, J. I., and Tamanoi, F. (2010) Mesoporous silica nanoparticles
facilitate delivery of siRNA to shutdown signaling pathways in
mammalian cells, Small, 6, 1185-1190.
39.Ashley, C. E., Carnes, E. C., Epler, K. E.,
Padilla, D. P., Phillips, G. K., Castillo, R. E., and Brinker, C. J.
(2012) Delivery of small interfering RNA by peptide-targeted mesoporous
silica nanoparticle-supported lipid bilayers, ACS Nano,
6, 2174-2188.
40.Na, H.-K., Kim, M.-H., Park, K., Ryoo, S.-R.,
Lee, K. E., Jeon, H., and Min, D.-H. (2012) Efficient functional
delivery of siRNA using mesoporous silica nanoparticles with ultralarge
pores, Small, 8, 1752-1761.
41.Zhang, L., Wang, T., Li, L., Wang, C., Su, Z.,
and Li, J. (2012) Multifunctional fluorescent-magnetic
polyethyleneimine functionalized Fe3O4-mesoporous
silica yolk-shell nanocapsules for siRNA delivery, Chem.
Commun., 48, 8706-8708.
42.Hartono, S. B., Gu, W., Kleitz, F., Liu, J., He,
L., Middelberg, A. P. J., and Qiao, S. Z. (2012) Poly-L-lysine
functionalized large pore cubic mesostructured silica nanoparticles as
biocompatible carriers for gene delivery, ACS Nano, 6,
2104-2117.
43.Li, X., Chen, Y., Wang, M., Ma, Y., Xia, W., and
Gu, H. (2013) A mesoporous silica
nanoparticle–PEI–fusogenic peptide system for siRNA
delivery in cancer therapy, Biomaterials, 34,
1391-1401.
44.Wang, M., Li, X., Ma, Y., and Gu, H. (2013)
Endosomal escape kinetics of mesoporous silica-based system for
efficient siRNA delivery, Int. J. Pharm., 448, 51-57.
45.Lin, D., Cheng, Q., Jiang, Q., Huang, Y., Yang,
Z., Han, S.., and Dong, A. (2013) Intracellular cleavable
poly(2-dimethylaminoethyl methacrylate) functionalized mesoporous
silica nanoparticles for efficient siRNA delivery in vitro and
in vivo, Nanoscale, 5, 4291-4301.
46.Shen, J., Kim, H. C., Su, H., Wang, F., Wolfram,
J., Kirui, D., and Shen, H. (2014) Cyclodextrin and polyethylenimine
functionalized mesoporous silica nanoparticles for delivery of siRNA
cancer therapeutics, Theranostics, 4, 487-497.
47.Du, X., Xiong, L., Dai, S., Kleitz, F., and Qiao,
S. Z. (2014) Intracellular microenvironment-responsive dendrimer-like
mesoporous nanohybrids for traceable, effective, and safe gene
delivery, Adv. Funct. Mater., 24, 7627-7637.
48.Chen, Y., Gu, H., Zhang, D. S.-Z., Li, F., Liu,
T., and Xia, W. (2014) Highly effective inhibition of lung cancer
growth and metastasis by systemic delivery of siRNA via multimodal
mesoporous silica-based nanocarrier, Biomaterials, 35,
10058-10069.
49.Hartono, S. B., Yu, M., Gu, W., Yang, J.,
Strounina, E., Wang, X., and Yu, C. (2014) Synthesis of
multi-functional large pore mesoporous silica nanoparticles as gene
carriers, Nanotechnology, 25, 055701.
50.Finlay, J., Roberts, C. M., Dong, J., Zink, J.
I., Tamanoi, F., and Glackin, C. A. (2015) Mesoporous silica
nanoparticle delivery of chemically modified siRNA against TWIST1 leads
to reduced tumor burden, Nanomed. Nanotechnol., 11,
1657-1666.
51.Xiong, L., Bi, J., Tang, Y., and Qiao, S.-Z.
(2016) Magnetic core-shell silica nanoparticles with large radial
mesopores for siRNA delivery, Small, 12, 4735-4742.
52.Moller, K., Muller, K., Engelke, H., Brauchle,
C., Wagner, E., Bein, T., and Brauchle, C. (2016) Highly efficient
siRNA delivery from core-shell mesoporous silica nanoparticles with
multifunctional polymer caps, Nanoscale, 8,
4007-4019.
53.Bhattarai, S. R., Muthuswamy, E., Wani, A.,
Brichacek, M., Castañeda, A. L., Brock, S. L., and Oupicky, D.
(2010) Enhanced gene and siRNA delivery by polycation-modified
mesoporous silica nanoparticles loaded with chloroquine, Pharm.
Res., 27, 2556-2568.
54.Chen, A. M., Zhang, M., Wei, D., Stueber, D.,
Taratula, O., Minko, T., and He, H. (2009) Co-delivery of doxorubicin
and Bcl-2 siRNA by mesoporous silica nanoparticles enhances the
efficacy of chemotherapy in multidrug-resistant cancer cells,
Small, 5, 2673-2677.
55.Ma, X., Teh, C., Zhang, Q., Borah, P., Choong,
C., Korzh, V., and Zhao, Y. (2014) Redox-responsive mesoporous silica
nanoparticles: a physiologically sensitive codelivery vehicle for siRNA
and doxorubicin, Antioxid. Redox Signal., 21,
707-722.
56.Ma, X., Zhao, Y., Ng, K. W., and Zhao, Y. (2013)
Integrated hollow mesoporous silica nanoparticles for target drug/siRNA
co-delivery, Chemistry, 19, 15593-15603.
57.Zhou, X., Chen, L., Nie, W., Wang, W., Qin, M.,
Mo, X., and He, C. (2016) Dual-responsive mesoporous silica
nanoparticles mediated codelivery of doxorubicin and Bcl-2 siRNA for
targeted treatment of breast cancer, J. Phys. Chem. C,
120, 22375-22387.
58.Meng, H., Liong, M., Xia, T., Li, Z., Ji, Z.,
Zink, J. I., and Nel, A. E. (2010) Engineered design of mesoporous
silica nanoparticles to deliver doxorubicin and P-glycoprotein siRNA to
overcome drug resistance in a cancer cell line, ACS Nano,
4, 4539-4550.
59.Meng, H., Mai, W. X., Zhang, H., Xue, M., Xia,
T., Lin, S., and Nel, A. E. (2013) Codelivery of an optimal drug/siRNA
combination using mesoporous silica nanoparticles to overcome drug
resistance in breast cancer in vitro and in vivo,
ACS Nano, 7, 994-1005.
60.Kar, M., Tiwari, N., Tiwari, M., Lahiri, M., and
Gupta, S. S. (2013) Poly-L-arginine grafted silica mesoporous
nanoparticles for enhanced cellular uptake and their application in DNA
delivery and controlled drug release, Particle Particle Systems
Charact., 30, 166-179.