Effects of Mitochondrial Antioxidant SkQ1 on Biochemical and Behavioral Parameters in a Parkinsonism Model in Mice
V. V. Pavshintsev1,2*, L. S. Podshivalova2, O. Y. Frolova1, M. V. Belopolskaya1, O. A. Averina2, E. A. Kushnir1, N. V. Marmiy1, and M. L. Lovat1,2
1Institute of Mitoengineering, Lomonosov Moscow State University, 119234 Moscow, Russia; E-mail: vsevolodpav@mail.ru2Lomonosov Moscow State University, Faculty of Biology, 119234 Moscow, Russia
* To whom correspondence should be addressed.
Received July 28, 2017; Revision received August 23, 2017
According to one hypothesis, Parkinson’s disease pathogenesis is largely caused by dopamine catabolism that is catalyzed on mitochondrial membranes by monoamine oxidase. Reactive oxygen species are formed as a byproduct of these reactions, which can lead to mitochondrial damage followed by cell degeneration and death. In this study, we investigated the effects of administration of the mitochondrial antioxidant SkQ1 on biochemical, immunohistochemical, and behavioral parameters in a Parkinson-like condition caused by protoxin MPTP injections in C57BL/6 mice. SkQ1 administration increased dopamine quantity and decreased signs of sensory-motor deficiency as well as destruction of dopaminergic neurons in the substantia nigra and ventral tegmental area in mice with the Parkinson-like condition.
KEY WORDS: Parkinson’s disease, MPTP, mitochondrial antioxidants, SkQ1, sensorimotor deficiency, dopaminergic neuronsDOI: 10.1134/S0006297917120100
Abbreviations: BW, “Beam walking” test; CNS, central nervous system; DAPI, 4´,6-diamidino-2-phenylindole; DOPA, 3,4-dihydroxyphenylalanine; DOPAC, 3,4-dihydroxyphenylacetic acid; EPM, “Elevated plus maze” test; HEB, hematoencephalic barrier; HPLC, high performance liquid chromatography; MAO-B, monoamine oxidase-B; MPDP+, 1-methyl-4-phenyl-2,3-dihydropyridine-ion; MPP+, 1-methyl-4-phenyl-pyridinium; MPTP, 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine; OF, “Open field” test; PD, Parkinson’s disease; Porsolt, “Porsolt forced swim” test; ROS, reactive oxygen species; SD, sensorimotor deficiency; SkQ1 (PDTP), plastoquinonyl-decyltriphenylphosphonium; TH, tyrosine hydroxylase (marker of dopaminergic neurons); TM, “T-maze” test.
Parkinson’s disease (PD) is characterized by the degeneration of
dopaminergic neurons in the basal ganglia responsible for coordination
of complex motor acts [1]. Among the many factors
contributing to the development of this disease, one is of particular
interest – the presence of a direct link between oxidative stress
and the development of PD symptoms [2], also due to
reactive oxygen species (ROS)-mediated specific damage to neurons of
the substantia nigra [3]. Endogenous ROS, formed
during bioamine metabolism, can also contribute to PD pathogenesis. The
integral protein monoamine oxidase (MAO), located on the outer
mitochondrial membrane of most cells, and especially nerve cells, is
responsible for the basic monoamine catabolism in nerve endings; the
reduced enzyme is further oxidized by oxygen, and this process is
accompanied by the formation of hydrogen peroxide [4]. It is important to note that cytochrome oxidase
and most other oxidases form water in this reaction. In the presence of
Fe2+ and Cu2+, hydrogen peroxide induces the
formation of hydroxyl radical (OH•, redox
potential +1.3 V) capable of oxidizing almost any cell
structure, which leads to damage dopaminic neurons. This results in
deficiency of neuronal functions in the striatum and substantia nigra,
which leads to compensatory enhancement of dopamine synthesis in the
remaining dopaminergic neurons, contributing to the further development
of pathology [5]. Increased level of
8-oxo-2′-deoxyguanosine, a marker of oxidative DNA damage, in
patients with PD provides indirect confirmation of this hypothesis [6]. These pathological processes can also cause
changes in the structure of α-synuclein, leading to its
accumulation in neurons and aggregation with the formation of Lewy
bodies [7].
Administration of the protoxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) to C57BL/6 mice is a standard model of Parkinson’s disease in animals. This line of mice is the most common model used for PD research due to genetically less active system of antioxidant protection. Due to its lipophilicity, MPTP penetrates the hematoencephalic barrier (HEB) entering the brain, where it converts into 1-methyl-4-phenyl-2,3-dihydropyridinium ion (MPDP+) catalyzed by MAO-B in glial cells. MPDP+ is oxidized to the final product 1-methyl-4-phenyl-pyridinium (MPP+), which is actively transported into neurons by the dopamine transporter. Then it enters mitochondria following the electrochemical gradient, selectively inhibits complex I of the respiratory chain, and causes opening of pores in the mitochondrial membrane. Excessive ROS formation [8] initiates cell necrosis or apoptosis [5]. Experiments have shown that MPTP selectively simulates the key pathogenetic PD mechanism in animals – death of nigrostriatal dopaminergic neurons causing dopamine deficiency in the striatum, followed by the appearance of Parkinsonian symptoms in behavior [9].
Low molecular weight antioxidants are traditionally used to reduce the effects of oxidative damage of various etiologies to the organism [10]. Their common drawback is the impossibility of reaching the desired concentration in the affected area without systemic administration of high doses, at which antioxidants can acquire prooxidant properties [11]. This feature apparently determines the insufficient effectiveness of traditional antioxidants in the therapy of diseases associated with oxidative stress [10]. Moreover, since it is mitochondria that appear to be the main source of ROS causing oxidative damage to the organism in neurodegenerative disorders, it seems crucial to find the way to deliver significant amounts of antioxidant to this organelle [12].
Mitochondria-targeted antioxidants are part of a new group of drugs that have a unique ability to penetrate the lipid bilayer and accumulate in cells and their structures, especially in mitochondria, following the charge gradient. Oral administration of ubiquinonyl decyltriphenylphosphonium (MitoQ10) to mice in the Parkinson-like state caused by MPTP [13] reduced the progression of the disease symptoms. However, despite these results, the drawback of ubiquinone in the structure of MitoQ10 is its insufficient antioxidant activity, which necessitates the use of high therapeutic doses (4 mg/kg in the course of administration). The narrow window between the anti- and pro-oxidant effects of the drug seems to explain the failure in the attempted treatment of patients suffering from PD by prescribing them MitoQ10 for a year [14].
Plastoquinonyl decyltriphenylphosphonium (PDTP), or SkQ1, is another promising compound. Its contains decyltriphenylphosphonium cation and a more potent plant antioxidant, plastoquinol, which significantly increases its antioxidant properties [15]. This structure significantly reduces the effective dose of the drug, which reduces the likelihood of prooxidant effect. Previously, SkQ1 administration has been shown to slow the development of motor dysfunctions in models of several neurodegenerative pathologies: amyotrophic lateral sclerosis, Alzheimer’s disease, Huntington’s chorea, and damages caused by kidney and liver ischemia–reperfusion [16].
Administration of SkQ1 against the background of the development of the Parkinsonian-like state could reduce the increased ROS level in mitochondria of brain dopaminergic neurons, thus having a neuroprotective effect and reducing the rate of PD development.
MATERIALS AND METHODS
We used 60 SPF mice of the C57BL/6 line 8-week-old at the beginning of the experiment. The animals were kept in standard conditions in a barrier-type vivarium in rooms with air exchange rate of at least 15 volumes per hour, temperature 19-24°C (daily difference not more than 2°C), humidity 30-70%, and light mode 12/12. Each cage contained five animals with free access to water and food. The research protocol was approved by the Commission on Bioethics of the Institute of Mitoengineering of Moscow State University.
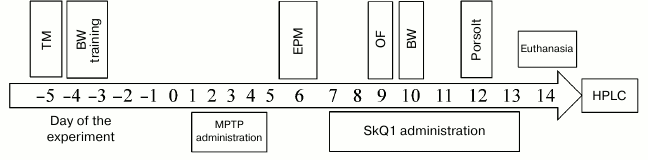
Scheme of experiments. Animals were divided into four groups (n = 15) according to parameters of their motor activity in the T-maze test: “intact control”, “MPTP” – intraperitoneal (i.p.) administration of protoxin MPTP during the first 4 days, 30 mg/kg daily, “MPTP + SkQ1” – i.p. administration of protoxin MPTP (4 days, 30 mg/kg daily), and 3 days later, SkQ1 i.p. administration, 1000 nmol/kg daily (days 7-13), control group “SkQ1” – i.p. administration of SkQ1, 1000 nmol/kg daily for a week. It is important to note that SkQ1 administration was started much later than that of MPTP, i.e. against the background of already formed Parkinson-like state, in contrast to the work of Ghosh et al., who injected MitoQ10 prior to MPTP, and then also after MPTP [13]. On the 6th-12th day after the first MPTP injection, we conducted behavior testing (“Beam walking” test (BW), “Elevated plus maze” test (EPM), “Open field” test (OF), and “Porsolt forced swim” test (Porsolt)). On day 14, the mice were euthanized for subsequent biochemical analysis (striatum bioamine level determination by HPLC (high performance liquid chromatography) and tyrosine hydroxylase (TH) count by Western blot), as well as immunohistological analysis of brain neurons (areas of substantia nigra and ventral tegmental area) to TH. The scheme of the experiment is shown in Fig. 1.
Fig. 1. Scheme of the experiment. TM, “T-maze”; BW, “Beam walking”; EPM, “Elevated plus maze”; OF, “Open field”; Porsolt, “Porsolt forced swim”.
Behavioral techniques. The “T-maze” consists of three closed arms located perpendicular to each other (starter and two lateral) (Scientific Production Association “Open Science”, Russia). The length of the arms was 30 cm, width 10 cm, height 20 cm. The mouse was placed in the starting arm. During 5 min, the NoldusEthovisionXT 8.5 program automatically recorded the following parameters: total run, travel time, time of immobility (at a speed of less than 0.2 cm/sec), average speed [17].
The “Elevated plus maze” test consists of two closed (15 cm height, 50 lx illuminance) and two open arms (illuminance 500 lx), located opposite each other (each arm 30 cm long) (SPA “Open Science”). The maze is raised 70 cm above the floor. The animal was placed in the center of the maze, head towards the open arm. During 5 min, the NoldusEthovisionXT 8.5 program automatically recorded the following parameters: total run, time of immobility, average speed, total time and number of entering the open arms [18].
The “Open Field” test is an arena with a diameter of 63 cm illuminated by bright light (450 lx) (SPA “Open Science”). The parameters recorded were: total run, time of immobility, average speed, number of acts of vertical motor activity and grooming [19].
In the “Beam walking” test, the animal should walk along a double bar (1 m) consisting of an upper beam tapering from 2 to 0.5 cm and a lower bar, 1 cm wider than the upper beam, and enter a dark shelter box (SPA “Open Science”). The test is used to evaluate the sensorimotor function of each limb [20]. It registers: setting a paw on the upper and lower bars, number of paw slips, and the total number of steps. The degree of severity of the sensorimotor deficit (SD) is calculated by the formula (in percentages):
mistakes + 0.5 × slips
SD = -- × 100%.
total number of steps
Biochemical tests. Western blot was performed on 16 striatum samples (four from each group, half of the striatum for the study). The tissue was weighed and homogenized in buffer (50 mM Tris-HCl, pH 7.5, 0.15 M NaCl, 1% NP40, 0.1% SDS) at a ratio of 1 ml buffer per 100 mg sample, and then incubated on ice for 30 min. The samples were then centrifuged at 12,000g for 15 min at 4°C. After centrifugation, the supernatant was separated, and the homogenate total protein was brought to 5 mg/ml using the Pierce™ BCA Protein Assay kit. Proteins in the samples were separated by Laemmli electrophoresis in 10% polyacrylamide gels. In addition to the samples, PageRuler™ Plus Prestained Protein Ladder markers of 10 to 250 kDa were applied on the gel. Protein fractions were transferred from the gel to PVDF membrane during 1 h at a current of 150 mA. The section corresponding to the molecular weight of the tested protein (~55 kDa) was cut out of the membrane based on the protein standards. After 1 h of blocking with a 5% solution of dried milk in 1× PBS-T, the membrane was washed 3 × 5 min with 1× PBS-T, then incubated in 1× PBS-T solution of primary antibodies (anti-TH, ab112; Abcam, USA; rabbit, 1 : 1000) overnight at 4°C. The membrane was again washed 3 × 5 min with 1× PBS-T, then incubated in solution of secondary antibodies conjugated with horseradish peroxidase (anti-rabbit HRP conj., 1 : 2000) in 1× PBS-T at room temperature for 1 h, after which the membrane was again washed 3 × 5 min with 1× PBS-T. The signal was visualized using the Super Signal West Pico Chemiluminescent Substrate system.
Bioamines were determined in the striatum by reversed-phase HPLC with amperometric detection with electrode oxidation potential +0.85 V. The composition of the mobile phase (solution A) was 70 mM sodium dihydrophosphate, 1.5 mM sodium octyl sulfonate, and 40 mM chloroacetic acid. The elution was carried out with a gradient of acetonitrile. The time of sample release from the column was 6-15 min with sensitivity range 0.4 ng/ml. We detected dopamine, 3,4-dihydroxyphenylalanine (DOPA), and 3,4-dihydroxyphenylacetic acid (DOPAC).
In immunohistochemical studies, mouse brain samples were fixed in a 4% solution of paraformaldehyde in 0.1 M phosphate buffer. Mid-brain cutting was performed according to the Allen brain atlas (http://atlas.brain-map.org). The samples were washed in water from the fixator, dehydrated in alcohols and dioxane (three shifts), impregnated with Histomix, and embedded in blocks. Five-micrometer-thick frontal serial sections were made from each block and mounted on glass. The histological sections were analyzed using the immunofluorescence method with primary antibodies to TH (ab112; Abcam) and goat anti-rabbit FITC, sc3839 (Santa Cruz, USA), as secondary antibodies. Incubation with primary antibodies was carried out overnight at 4°C and with secondary antibodies for 2 h at 37°C. The dilution of both primary and secondary antibodies was 1 : 50. Nuclei were stained using 4′,6-diamidino-2-phenylindole (DAPI) (sc24941; Santa Cruz). The preparations were studied using an immunofluorescence microscope (Micromed 3 LUM LED) equipped with a video capture system and corresponding software. Morphometric analysis was performed on photomicrographs made using the video eyepiece ToupCam 9.0 MP. The ratio of the stained material to the area of the studied regions and the number of nuclei were determined using a semiautomatic method on ciphered samples under operator control in NIH ImageJ 1.43u software. Percentage of area occupied by TH-immunoreactive material was calculated as a function of the total area of the studied structures in a series of sections (each 10th section of the structures studied separately from the right and left sides, at least 10 sections per mouse; lens magnification ×20).
The data were statistically processed using the STATISTICA 10 program with the one-way ANOVA method with a posteriori analysis using the Fisher criterion (LSD). The data were considered significant at p < 0.05. Data in the figures are presented as mean values and standard error of the mean; * p < 0.05 compared to the intact control group, # p < 0.05 compared to the MPTP group.
RESULTS
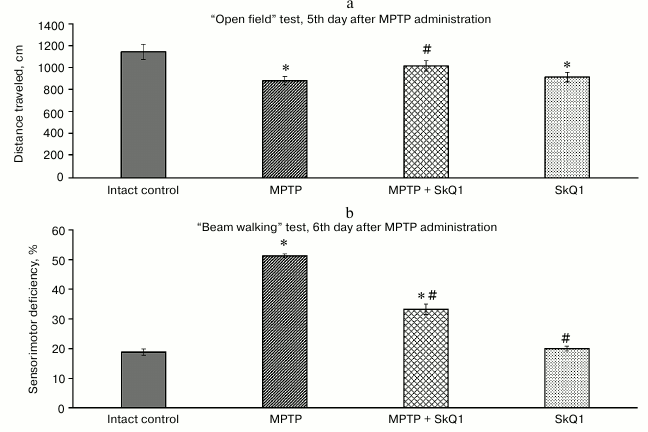
Behavioral tests. The “Elevated plus maze” test was conducted as a control of motor activity and anxiety on the 2nd day after the first (of four) MPTP administrations (but before SkQ1 administration). Two administrations of MPTP protoxin was sufficient for the development of motor dysfunctions in the mice. The average distance traveled decreased by 22.7% relative to the intact control group (1346 ± 48.3 and 1097 ± 56.1 cm, respectively). The MPTP-treated mice spent 10% more time in the closed arms than the intact animals (219 ± 6.2 and 199 ± 4.8 s, respectively). On the 5th and 6th day of MPTP administration, disturbances increased (Fig. 2): in the “Open field” test, there was a significant decrease in the average distance traveled relative to the intact group (881 ± 46.8, 1143 ± 69, respectively, p < 0.05), and in the “Beam walking” test there was an increase in the sensorimotor deficit (18.9 ± 1.09 and 51.36 ± 0.84%, p < 0.05). Administration of SkQ1, which started on the 5th day of intoxication, prevented the decrease in motor activity, the values being significantly different from the MPTP group (1016 ± 43.4 cm for the distance traveled, 33.3 ± 0.84% for the sensorimotor deficiency).
Fig. 2. “Open field” test (parameter – distance traveled, cm) (a) and “Beam walking” test (parameter – sensorimotor deficiency, average for four paws, %) (b); n = 60, 15 animals per group; * p < 0.05 compared to the intact control group, # p < 0.05 compared to the MPTP group.
In the “Porsolt forced swim” test (on the 8th day of the experiment, SkQ1 had been administered 5 times), none of the groups showed significant differences, which indicates that the tested compounds have no effect in this mode of administration on physical endurance and depressive-like state of animals. PD is known to damage primarily the initiation of arbitrary movements; locomotor functions caused by external influence suffer less.
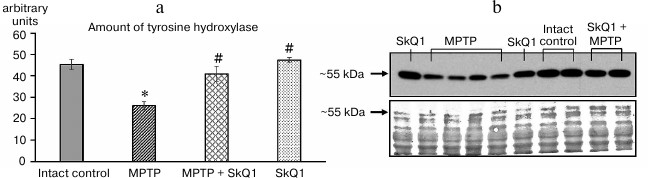
Results of biochemical tests. The results of TH estimation in striatum tissues by the Western blot method are shown in Fig. 3.
Fig. 3. a) Amount of TH (arbitrary units) in the striatum region (11th day after the first MPTP administration). b) Result of analysis of the amount of TH protein in samples by the Western blot method (top), control of loading of samples by total protein for normalization, stained with bicinchoninic acid (bottom); n = 60, 15 animals per group; * p < 0.05 compared to the intact control group, # p < 0.05 compared to the MPTP group.
Administration of MPTP reduced this parameter by 80% with respect to the intact control group (26.1 ± 3.51 and 45.3 ± 2.44 arbitrary units, respectively, p < 0.05), while SkQ1 administration prevented the decrease in this parameter, the obtained (40.9 ± 1.28 arbitrary units) having no statistically significant differences from the intact control group. SkQ1 administration (without MPTP) did not affect the amount of TH in the samples compared to the intact control.
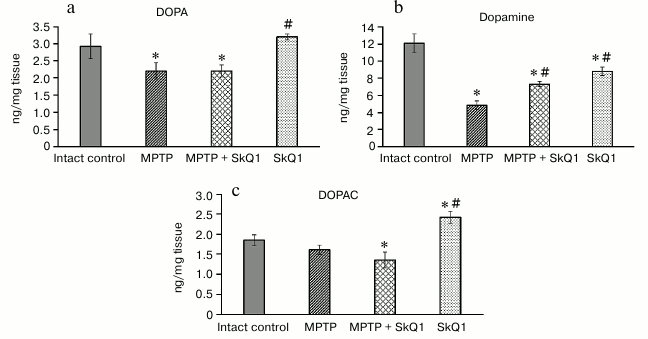
The biochemical analysis showed that a significant decrease in the amounts of DOPA (2.21 ± 0.176 and 2.93 ± 0.361 ng/mg, respectively) and dopamine (4.86 ± 0.297 and 12.13 ± 1.089 ng/mg, respectively) in the striatum occurred in the group receiving MPTP compared to intact control (Fig. 4). The content of dopamine metabolite DOPAC did not differ significantly. In the SkQ1 + MPTP group, there was higher dopamine concentration (7.34 ± 0.189 ng/mg, p < 0.05) than in the MPTP group. For mice receiving only SkQ1, there was a slight decrease in the amount of dopamine (8.84 ± 0.518 ng/mg) and an increase in DOPAC (2.43 ± 0.116 ng/mg) with respect to the intact control (p < 0.05).
Fig. 4. DOPA (a), dopamine (b) and DOPAC (c) content in striatum tissues (ng/mg tissue); n = 60, 15 animals per group; * p < 0.05 compared to the intact control group, # p < 0.05 compared to the MPTP group.
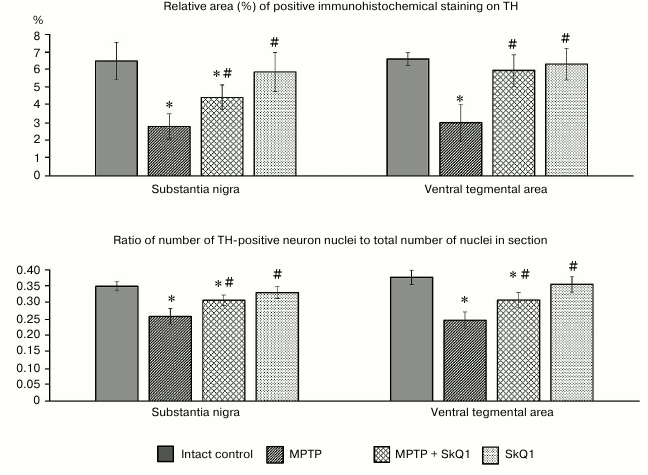
Administration of MPTP resulted in a significant (p < 0.05) reduction of the area occupied by TH-positive (dopaminergic) neurons compared to the intact control group, both in the substantia nigra region (2.79 ± 0.713 and 6.49 ± 1.046%, respectively) and in the ventral tegmental area (3.01 ± 1.028 and 6.61 ± 0.364%), the areas where PD development causes the greatest neuronal death (Fig. 5). At the same time, when calculating the ratio of cell nuclei containing TH-positive regions and the total number of nuclei in the section, we also observed a significant decrease in this parameter (0.26 ± 0.024 and 0.35 ± 0.014 for the substantia nigra, 0.24 ± 0.019 and 0.38 ± 0.022 for the ventral tegmental area). Combined SkQ1 + MPTP administration resulted in increase in the area of TH-containing material compared to that of the MPTP group, reaching significant differences in the substantia nigra (4.43 ± 0.708%). The ratio of nuclei of TH-positive cells was also significantly higher in both studied areas (0.31 ± 0.015 for substantia nigra, and 0.32 ± 0.022 for the ventral tegmental area), whereas the administration of only SkQ1 did not lead to changes in these parameters, which remained at the level of the intact control values.
Fig. 5. Relative area (%) of positive immunohistochemical staining on TH (top), ratio of the number of TH-positive neuron nuclei to the total number of nuclei in the section (bottom) in substantia nigra and ventral tegmental area of mice in different groups; * p < 0.05 compared to the intact control group, # p < 0.05 compared to the MPTP group.
DISCUSSION
When administered intraperitoneally, some MPTP enters brain neurons, where it is oxidized in dopaminergic neurons by MAO to the highly toxic form MPP+, which causes neurodegeneration by inhibiting respiratory complex I of mitochondria accompanied by formation of an excessive amount of ROS. The next day, MPP+ is partially metabolized and is excreted in the urine. Surviving dopaminergic neurons apparently increase dopamine metabolism, which contributes to further ROS generation due to the oxidation of dopamine catalyzed by MAO [21]. SkQ1 prevents ROS formation and neutralizes already existing ROS, which can explain its neuroprotective effect.
Administration of MPTP reduced TH level and led to a decrease in the amount of DOPA and dopamine in the striatum tissues. Similar results were obtained in immunohistochemical testing, where we observed a significant decrease in TH-positive areas and in the number of nuclei of TH-containing cells in both substantia nigra and ventral tegmental area. These data indicate not only a decrease in TH expression by cells, but also a direct reduction in the number of dopaminergic neurons in the studied regions. The results of behavioral studies showed that the protoxin administration reduced the level of motor activity and resulted in a sensorimotor deficiency, which is also a characteristic expression of symptoms of damage to dopaminergic neurons in this PD model [22, 23]. Our data confirm the effectiveness of the mode of MPTP administration used in this study and they are consistent with literature data on the degeneration of dopamine neurons of substantia nigra and ventral tegmental area [24], which leads to dopamine deficiency in the striatum, decrease in motor activity, and increase in sensorimotor deficiency characteristic of PD [25, 26].
The course of intraperitoneal SkQ1 administration (1000 nmol/kg) had no significant effect on the parameters of sensorimotor deficiency, slightly reducing only the overall motor activity. One-week-long administration of the antioxidant against the background of formed PD symptoms led to significant restoration of the behavioral parameters (motor activity, sensorimotor deficiency), as well as partial restoration of the dopamine levels against the background of DOPAC decrease, which may indicate a compensatory slowing of its catabolism.
Also, it has been shown that SkQ1 administration against the background of PD symptoms significantly compensated the decrease in TH amount, the area of TH-positive regions, and the number of dopaminergic neurons in substantia nigra and ventral tegmental area. These data may indicate both the involvement of the SkQ1 molecule in the normalization of TH expression (disturbed against the background of oxidative stress after MPTP administration) and the decrease in the severity of damage of these dopaminergic neurons. These results are consistent with the materials of the previously published work by Gnosh et al., who studied the properties of MitoQ10 (a compound similar in its mechanism of action to SkQ1) on a similar model of Parkinsonism.
Thus, SkQ1, being a mitochondria-targeted antioxidant, reduces oxidative stress and supports the reduction of the symptoms of the Parkinson-like state in the toxic model in C57BL/6 mice. The observed effect is most likely directly associated with the neuroprotective properties of the SkQ1 molecule, namely, with a decrease in the amount of ROS formed due to the effect of MPTP protoxin and uncompensated dopamine metabolism. The effectiveness of SkQ1 shown in this study, as well as on models of other neurodegenerative diseases (amyotrophic lateral sclerosis, Alzheimer’s disease, Huntington’s chorea), suggests the important role of ROS in the pathogenesis of many neurodegenerative diseases.
REFERENCES
1.Delaville, C., Deurwaerdere, P., and Benazzouz, A.
(2011) Noradrenaline and Parkinson’s disease, Front. Syst.
Neurosci., doi: 10.3389/fnsys.2011.00031.
2.Richardson, J. R., Ananya, R., Shalat, S. L.,
Buckley, B., Winnik, B., Gearing, M., Levey, A. I.,
O’Suilleabhain, P., and German, D. C. (2011)
Hexachlorocyclohexane levels in serum and risk of Parkinson’s
disease, Neurotoxicology, 32.
3.Langston, J. W. (2002) Parkinson’s disease:
current and future challenges, Neurotoxicology, 23,
443-450.
4.Edmondson, D. E., Binda, C., and Mattevi, A. (2007)
Structural insights into the mechanism of amine oxidation by monoamine
oxidases A and B, Arch. Biochem. Biophys., 464,
269-276.
5.Przedborski, S., Naini Ali, B., and Akram, M.
(2001) The Parkinson toxin 1-methyl-4-phenyl-1,2,3,6-tetrahydopyridine
(MPTP): a technical review of its utility and safety, J.
Neurochem., 76, 1265-1274.
6.Dorszewska, J., Florczak, J., Rozycka, A.,
Kempisty, B., Jaroszewska-Kolecka, J., Chojnacka, K., Trzeciak, W. H.,
and Kozubski, W. (2007) Oxidative DNA damage and level of thiols as
related to polymorphisms of MTHFR, MTR, MTHFD1 in Alzheimer’s and
Parkinson’s diseases, Acta Neurobiol. Exp., 67,
113-129.
7.Garrido, A., Aldecoa, I., Gelpi, E., and Tolosa, E.
(2017) Aggregation of α-synuclein in the gonadal tissue of 2
patients with Parkinson disease, JAMA Neurol., 74,
606-607.
8.Cassarino, D. S., Parks, J. K., Parker, W. D., and
Bennett, J. P. (1999) The parkinsonian neurotoxin MPP+ opens the
mitochondrial permeability transition pore and releases cytochrome
c in isolated mitochondria via an oxidative mechanism,
Biochim. Biophys. Acta, 1453, 49-62.
9.Ukraintseva, Y. S., Shchegolevskii, N. V.,
Korshunov, V. A., Kucheryanu, V. G., Ugryumov, M. V., and Bazya, A. S.
(2010) Modeling of the presymptomatic stage of parkinsonism in mice:
analysis of dopamine release in the striatum, Neurochem. J.,
4, 142-147.
10.Cohen-Kerem, R., and Koren, G. (2003)
Antioxidants and fetal protection against ethanol teratogenicity.
Review of the experimental data and implications to humans,
Neurotoxicol. Teratol., 25, 1-9.
11.Skulachev, V. P., Bogachev, A. V., and
Kasparinskiy, F. O. (2010) Membrane Bioenergetics [in Russian],
MSU, Moscow, pp. 269-321.
12.Skulachev, V. P. (2012) Mitochondria-targeted
antioxidants as promising drugs for treatment of age-related brain
diseases, Alzheimer’s Dis., 28, 283-289.
13.Ghosh, A., Chandran, K., Kalivendi, S. V.,
Joseph, J., Antholine, W. E., Hillard, C. J., Kanthasamy, A.,
Kanthasamy, A., and Kalyanaraman, B. (2010) Neuroprotection by a
mitochondria-targeted drug in a Parkinson’s disease model,
Free Radic. Biol. Med., 49, 1674-1684.
14.Snow, B. J., Rolfe, F. L., Lockhart, M. M.,
Frampton, C. M., O’Sullivan, J. D., Fung, V., Smith, R. A.,
Murphy, M. P., and Taylor, K. M. (2010) A double-blind,
placebo-controlled study to assess the mitochondria-targeted
antioxidant MitoQ as a disease-modifying therapy in Parkinson’s
disease, Mov. Disord., 25, 1670-1674.
15.Skulachev, V. P. (2009) An attempt to prevent
senescence: a mitochondrial approach, Biochim. Biophys. Acta,
1787, 437-461.
16.Lukashev, A. N., Skulachev, M. V., Ostapenko, V.,
Savchenko, A. Y., Pavshintsev, V. V., and Skulachev, V. P. (2014)
Advances in development of rechargeable mitochondrial antioxidants,
Prog. Mol. Biol. Transl. Sci., 127, 251-265.
17.Deacon, M. J., and Rawlins, P. N. (2006) T-maze
alternation in the rodent, Nat. Protocols, 1, 7-12.
18.Pellow, S., Chopin, P., File, S. E., and Briley,
M. (1985) Validation of open: closed arm entries in an elevated
plus-maze as a measure of anxiety in the rat, J. Neurosci.
Methods, 14, 149-167.
19.File, S. E., and Wardill, G. (1975) The
reliability of the hole-board apparatus, Psychopharmacologia,
44, 47-51.
20.Schallert, T., Cenci, M. A., and Whishaw, I. Q.
(2002) Animal models of neurological deficits: how relevant is the rat?
Nat. Rev. Neurosci., 3, 574-579.
21.Jackson-Lewis, V., and Przedborski, S. (2007)
Protocol for the MPTP mouse model of Parkinson’s disease, Nat.
Protocols, 2, 141-151.
22.Feng, G., Zhang, Z., Bao, Q., Zhang, Z., Zhou,
L., Jiang, J., and Li, S. (2014) Protective effect of chinonin in
MPTP-induced C57BL/6 mouse model of Parkinson’s disease, Biol.
Pharm. Bull., 37, 1301-1307.
23.Nagarajan, S., Chellappan, D. R., Chinnaswamy,
P., and Thulasingam, S. (2015) Ferulic acid pretreatment mitigates
MPTP-induced motor impairment and histopathological alterations in
C57BL/6 mice, Pharm. Biol., 53, 1591-1601.
24.Zhu, Y., Zhang, J., and Zeng, Y. (2012) Overview
of tyrosine hydroxylase in Parkinson’s disease, CNS Neurol.
Disord. Drug Targets, 11, 350-358.
25.Yang, W., Chen, Y. H., Liu, H., and Qu, H. D.
(2015) Neuroprotective effects of piperine on the
1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced Parkinson’s
disease mouse model, Int. J. Mol. Med., 36,
1369-1376.
26.Sagi, Y., Mandel, S., Amit, T., and Youdim, M. B.
(2007) Activation of tyrosine kinase receptor signaling pathway by
rasagiline facilitates neurorescue and restoration of nigrostriatal
dopamine neurons in post-MPTP-induced parkinsonism, Neurobiol.
Dis., 25, 35-44.