Exogenous 3-Deoxyglucosone-Induced Carbonyl and Oxidative Stress Causes β-Cells Dysfunction by Impairing Gut Permeability in Rats
L. Zhou1#, X. D. Song1#, H. Xu1, G. Q. Liang1, F. Wang1, L. R. Zhang1, F. Huang1, J. Cai1, and G. R. Jiang1,a*
1Suzhou Academy of Wumen Chinese Medicine, Suzhou TCM Hospital Affiliated to Nanjing University of Chinese Medicine, Suzhou, Jiangsu, China# These authors contributed equally to this work.
* To whom correspondence should be addressed.
Received May 11, 2018; Revision received June 27, 2018
3-Deoxyglucosone (3DG) is a highly reactive dicarbonyl species, and its accumulation evokes carbonyl and oxidative stress. Our recent data reveal the role of 3DG as an independent factor for the development of prediabetes and suggest that intestine could be its novel target tissue. The present study investigated whether exogenous 3DG increases intestinal permeability by triggering carbonyl and oxidative stress, thus contributing to β-cell dysfunction. Rats were administered 3DG for two weeks by gastric gavage. Then levels of insulin, ROS, MDA, SOD, NLRP3, TNF-α and IL-1β in blood plasma as well as the ROS level and content of TNF-α and IL-1β in pancreas were assessed. Also, the expression of E-cadherin and ZO-1 as well as levels of 3DG, protein carbonylation, ROS, TNF-α and IL-1β in colon were determined. The 3DG-treated rats showed an elevation in systemic oxidative stress (ROS, MDA and SOD) and in inflammation (TNF-α and IL-1β), decreased plasma insulin level 15 min after the glucose load, and increased levels of TNF-α, IL-1β and ROS in pancreatic tissue. In colon tissues of the 3DG-treated rats, decreased E-cadherin expression and increased ROS production as well as an elevation of TNF-α and IL-1β levels were observed. Interestingly, elevation of colon protein carbonylation was observed in the 3DG-treated rats that displayed 3DG deposition in colon tissues. We revealed for the first time that 3DG deposition in colon triggers carbonyl and oxidative stress and, as a consequence, impairs gut permeability. The enhanced intestinal permeability caused by 3DG deposition in colon results in systemic and pancreatic oxidative stress and inflammatory process, contributing to the development of β-cell dysfunction.
KEY WORDS: 3-deoxyglucosone, prediabetes, intestinal permeability, carbonyl and oxidative stress, protein carbonylation, ROS, β-cell dysfunctionDOI: 10.1134/S0006297918110068
Abbreviations: AGEs, advanced glycation end products; 3DG, 3-deoxyglucosone; GSH, glutathione; GSH-PX, glutathione peroxidase; IL, interleukin; MDA, malondialdehyde; NLRP3, nucleotide-binding oligomerization domain-leucine-rich repeats containing pyrin domain 3; RCS, reactive carbonyl species; ROS, reactive oxygen species; SOD, superoxide dismutase.
Type 2 diabetes mellitus is a glucose metabolism disorder characterized
by hyperglycemia, which is typically accompanied by excessive
generation of reactive oxygen species (ROS) and reactive carbonyl
species (RCS) [1]. An imbalance between
intracellular ROS production and ROS detoxification by an antioxidant
defense can result in oxidative stress that triggers damages at
molecular and cellular levels [2]. Excessive
oxidative stress can reduce insulin secretion by β-cells as well
as impair insulin signaling pathway in target tissues such as muscles,
adipose tissue and liver [3, 4]. Additionally, RCS accumulation, i.e., carbonyl
stress, modifies proteins and eventually leads to formation of advanced
glycation end products (AGEs) [5, 6]. RCS-induced carbonyl stress in its turn can
increase RCS and ROS levels. Taken together, the carbonyl and oxidative
stress could affect conformation, activity and function of cellular
proteins, which play essential roles in the pathogenesis of diabetes
and its complications.
Studies have reported that obese and diabetic mice display enhanced intestinal permeability [7, 8]. The intestinal barrier is the major barrier for the maintenance of intestinal homeostasis and metabolism. Alterations in intestinal barrier integrity are closely associated with the onset of metabolic diseases. Intestinal oxidative stress can enhance intestinal permeability, and this is thought to be a crucial factor for the development of type 2 diabetes mellitus. The level of oxidative stress also may be the most important mediator triggering low-grade inflammation in obesity and diabetes as a result of increased intestinal permeability. Studies demonstrated that excessive ROS generation could damage cell membranes, leading to increased intestinal permeability to endotoxins and to local or systemic inflammatory reactions [9, 10]. Accumulating evidence has confirmed that some endogenous or exogenous events can enhance intestinal permeability by triggering ROS production such as lipopolysaccharide, Al and alcohol [11-13]. ROS overproduction also could be a result of RCS accumulation, suggesting that RCS-induced carbonyl stress is capable of enhancing intestinal permeability, at least in part, by increasing oxidative stress. Thus, it is likely that the enhancement of intestinal permeability by RCS accumulation can be mediated not only via carbonyl stress through formation of protein carbonyls but also via oxidative stress through enhanced ROS formation.
3-Deoxyglucosone (3DG), one of the RCS, is easily formed from D-glucose and is an important precursor for AGEs. In living organisms, it is formed during the Maillard reaction and glucose degradation. 3DG can also be formed in food during caramelization processes and Maillard reaction, and the occurrence of 3DG in a great variety of commonly consumed foods has been published. High 3DG contents were measured in fruit juices (up to 410 mg/liter), balsamic vinegar (up to 2622 mg/liter), honey (10 mmol/kg), and bakery products such as cookies (up to 385 mg/kg) [14]. Based on the 3DG content in commonly consumed foods, the dietary intake of 3DG was estimated to range from 20 to 160 mg/day [14]. Our recent studies in clinical and animal research have postulated that 3DG is an independent factor for the development of prediabetes. The 3DG-induced β-cells dysfunction is a main mechanism linking 3DG with glucose homeostatic dysregulation. An excess of 3DG and other RCS such as glyoxal and methylglyoxal causes carbonyl overload and stress in such conditions as diabetes [15, 16]. Furthermore, 3DG can modify amino acid residues in proteins, which could lead to both ROS and AGE formation. Indeed, there is a substantial evidence of the 3DG-induced oxidative stress in various cell types [17]. 3DG was also reported to rapidly induce the ROS generation in vitro [18]. It is likely that the effects of 3DG on β-cells dysfunction are exerted not only via carbonyl stress through formation of protein carbonyls but also via oxidative stress through enhanced ROS formation. The earlier study has indicated that the absorption rate of 3DG from foodstuffs is very slow after single oral administration of 3DG [19]. We reported previously that 2-week 3DG administration by gastric gavage resulted in deposition of 3DG in intestinal tissue, and the highest content of 3DG was found in a colon. Thus, the intestine could be its novel target tissue [20, 21]. Given the demonstrated role of alterations in intestinal permeability in the development of β-cells dysfunction, the present study investigated whether exogenous 3DG increases intestinal permeability by triggering the production of carbonyl and oxidative stress, linking exogenous 3DG with pancreatic β-cell dysfunction.
The aim of the present study was to examine the impact of 2-week administration of 3DG by gastric gavage on changes in systemic and pancreatic oxidative stress and in inflammatory process in Sprague–Dawley (SD) rats in addition to the pancreatic β-cell dysfunction. Specifically, we aimed to evaluate the time course of the alterations that occur during pancreatic β-cell dysfunction development: protein carbonylation, oxidative stress, and inflammation in the colon as well as intestinal permeability.
MATERIALS AND METHODS
Synthesis of 3DG. According to the method of Kato et al. [19], 3DG was synthesized from glucose. The product was identified by IR, 1H-NMR, 13C-NMR and MS [22].
Animals. Eleven-week-old SD rats were purchased from Matt Albert Technology Co. Ltd (China) and housed in a controlled environment. All experimental procedures with animals were conducted in compliance with the Guide for the Care and Use of Laboratory Animals [23]. The study was approved by the local Ethic Committee of Suzhou Hospital of Traditional Chinese Medicine.
The rats had free access to a standard rodent chow diet (Shuangshi Laboratory Animal Feed Science Co. Ltd, China) and water. The diet contained water (≤10%), crude proteins (≥20.5%), crude fat (≥4%), crude fiber (≤5%), crude ash (≤8%) and mixture of vitamins and micronutrients. The rats were randomly divided into three groups, and each group consisted of six rats. The rats of the first, second and third group were administered a vehicle (0.9% NaCl, control), 5 mg/kg 3DG and 50 mg/kg 3DG, respectively, perorally by gastric gavage daily for two weeks. Body weight was measured daily. The rats were fasted overnight before the experiments.
Immunohistochemistry. After two-week administration of 3DG, the rats were euthanized, and 2-cm segments were prepared from the colon of the rats. Serial cryosections (5 μm) of each segment were then immunostained using anti-E-cadherin antibody and anti-ZO-1 antibody (both from Abcam, USA) according to the method of Hwang et al. [24].
Biochemical analyses. The oral glucose tolerance test was conducted by a traditional method [25]. Glucose levels in the blood taken from the tail vein were determined with a glucose meter ACCU-CHEK (Roche, USA). Additional blood samples were collected from the carotid artery 15 min after the glucose load to measure insulin levels using the Rat Insulin ELISA kit (Linco Research, Inc., USA) according to the manufacturer’s instructions. Interleukin (IL)-1β and tumor necrosis factor (TNF)-α in plasma and in pancreatic and colon tissues were determined using the ELISA kits (Genzyme, USA). Plasma levels of nucleotide-binding oligomerization domain-leucine-rich repeats containing pyrin domain 3 (NLRP3) and ROS were determined using the ELISA kits (AMDL Inc., USA). Plasma levels of malondialdehyde (MDA) and superoxide dismutase (SOD) and plasma activities of glutathione (GSH) and glutathione peroxidase (GSH-PX) were determined using the commercially available kits (Jiancheng Institute of Biotechnology, China). Plasma levels of GSH and GSH-PX were measured using GSH ELISA Kit (Abbexa Ltd, UK) and GSH-PX ELISA Kit (MyBioSource, USA), respectively.
Protein carbonylation. Amount of protein carbonyls (in nmol carbonyls per mg total protein) was determined in colon tissue of rats from the all three groups using a commercial kit (Sigma-Aldrich, USA) and following the manufacturer’s instructions. To determine protein concentration, a bicinchoninic acid protein assay kit (Sigma-Aldrich) was used following the manufacturer’s instructions.
Statistical analysis. Experimental results are expressed as mean ± SD. Statistical significance of differences was analyzed by the one-way analysis of variance. All p ≤ 0.05 were considered statistically significant.
RESULTS
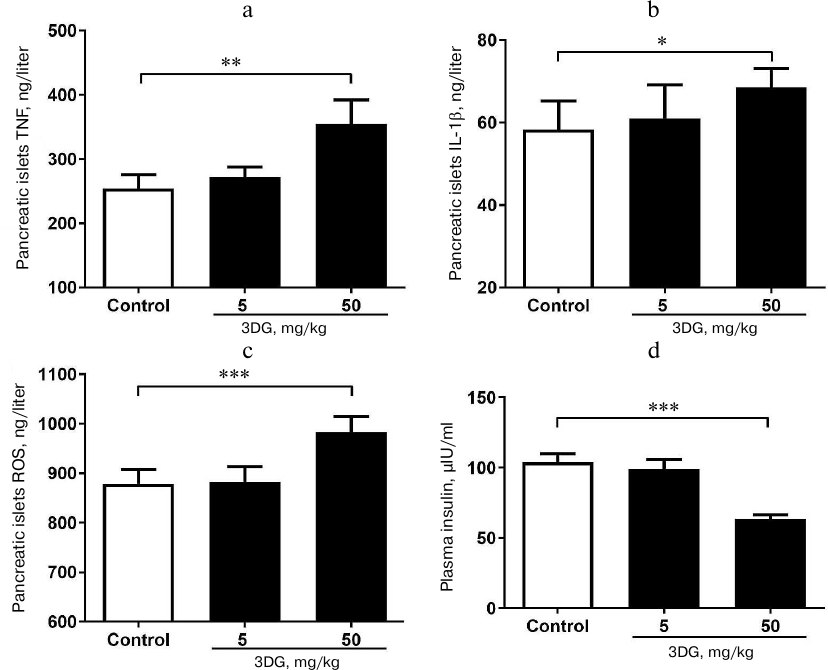
Increased levels of TNF-α, IL-1β and ROS in pancreatic tissue were observed in rats with 3DG-induced pancreatic β-cell dysfunction. As previously reported [26, 27], the increase in inflammatory cytokines and oxidative stress markers is involved in pancreatic islet cell dysfunction. We showed that both the TNF-α and IL-1β levels in pancreatic islets were significantly increased in the 50 mg/kg 3DG-treated group compared to the control group (Figs. 1a and 1b). We also examined the ROS level in pancreatic islets and obtained the consistent result that 50 mg/kg 3DG-treatment induced increase in ROS level in pancreatic islets (Fig. 1c). To evaluate the function of pancreatic islet cells in 3DG-treated rats, we examined the plasma insulin. After 15-min oral glucose load, the plasma insulin level was significantly decreased in 50 mg/kg 3DG-treated rats compared to the control group rats (Fig. 1d).
Fig. 1. Increased levels of TNF-α (a), IL-1β (b) and ROS (c) in pancreatic tissue of rats with 3DG-induced pancreatic β-cell dysfunction. Pancreatic islets cytokines and ROS were measured in rats fasted overnight, after 2-week 3DG treatment. Plasma insulin levels (d) were measured 15 min after the oral glucose load (2.5 g/kg) in 3DG- (5 or 50 mg/kg) or vehicle-treated rats (2-week treatment); *p < 0.05, **p < 0.01, p < 0.001 (n = 6).
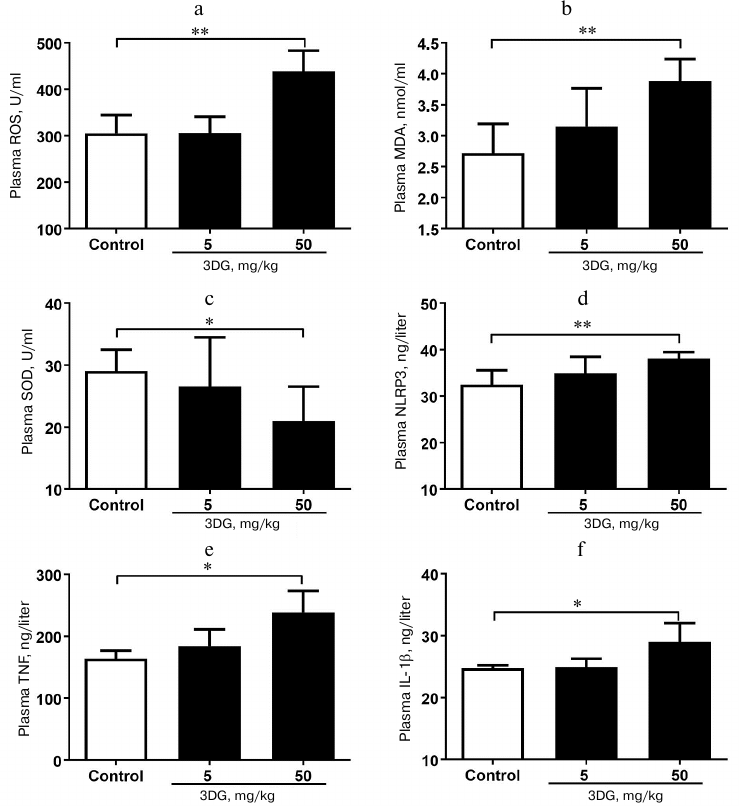
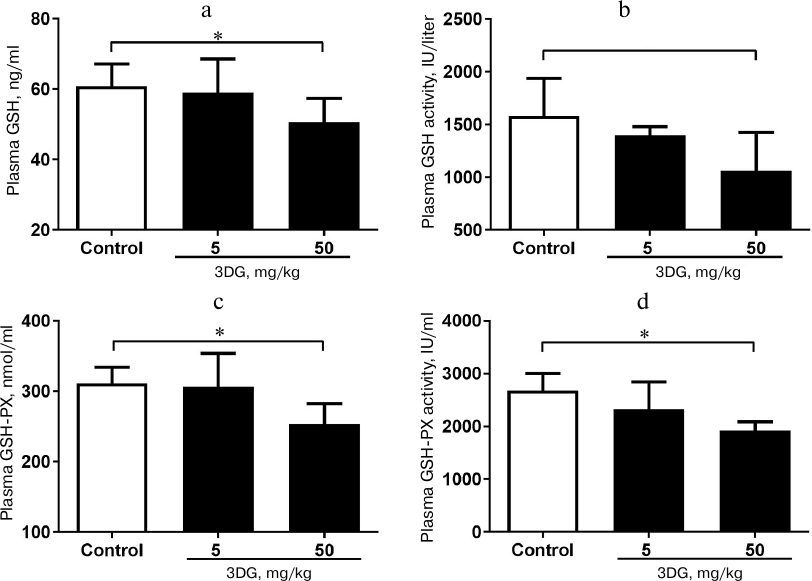
Systemic oxidative stress and inflammation increased in rats after 3DG administration for two weeks. Since the oxidative stress was previously reported to activate the inflammasome-associated NLRP3, a protein, triggering inflammatory reaction with subsequent release of inflammatory cytokines [28], we further assessed the oxidative stress in plasma in response to 3DG treatment. As shown in Figs. 2a and 2b, plasma ROS and MDA levels were significantly increased in the 50 mg/kg 3DG-treated group compared to the control, while plasma SOD level was significantly decreased in the 50 mg/kg 3DG-treated group compared to the control (Fig. 2c). In addition, both plasma level and activity of GSH and GSH-PX were significantly decreased in the 50 mg/kg 3DG-treated group compared to the control (Fig. 3, a-d). We also examined plasma NLRP3 level and found it to be increased in the 50 mg/kg 3DG-treated group (Fig. 2d). This was accompanied by higher plasma IL-1β and TNF-α levels in the 50 mg/kg 3DG-treated group compared to the control (Figs. 2e and 2f).
Fig. 2. Systemic oxidative stress and inflammation increased in rats after two weeks of 3DG administration. ROS (a), MDA (b), SOD (c), NLRP3 (d), TNF-α (e) and IL-1β (f) plasma levels were measured in rats fasted overnight, after 2-week 3DG treatment; *p < 0.05, **p < 0.01 (n = 6).
Fig. 3. Systemic redox homeostasis disorder in rats after two weeks of 3DG administration. GSH (a) and GSH-PX (c) plasma levels as well as GSH (b) and GSH-PX (d) plasma activity were measured in rats fasted overnight, after 2-week 3DG treatment.
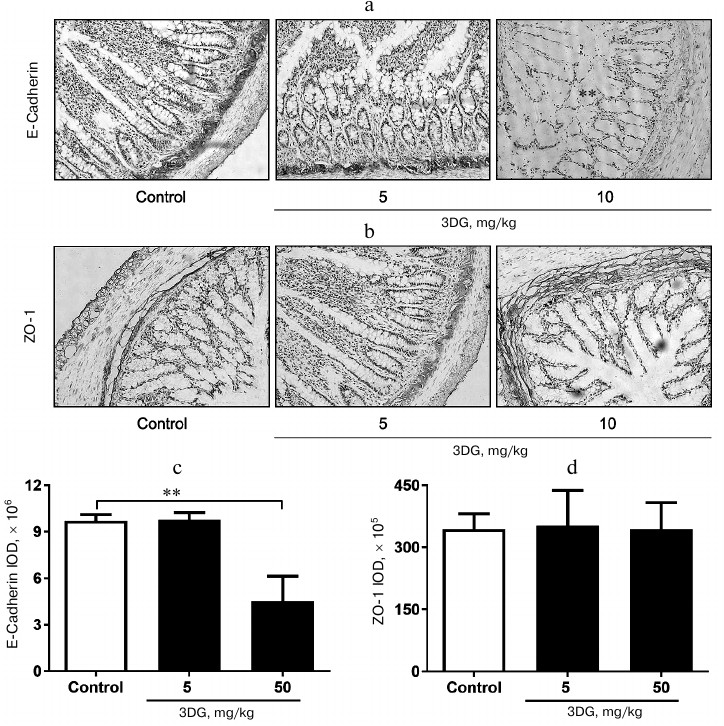
The expression of adherens-junction protein E-cadherin decreased in colon tissue of rats after two weeks of 3DG administration. To investigate consequences of 3DG administration on an intestinal barrier, we further evaluated expression of E-cadherin and ZO-1 in colon after 2-week 3DG treatment. As shown in Fig. 4a, the expression of E-cadherin was extremely decreased in the 50 mg/kg 3DG-treated group compared to the control. The integrated optical density (IOD) of E-cadherin in the 50 mg/kg 3DG-treated group was lower than that in the control (Fig. 4c). Nevertheless, ZO-1 expression was not significantly altered by 3DG treatment (Fig. 4b). The IOD of ZO-1 remained unchanged by 3DG treatment (no statistically significant differences compared to the control) (Fig. 4d).
Fig. 4. Immunohistochemical analysis of E-cadherin (a) and ZO-1 (b) protein expression in colon tissue of 3DG- or vehicle-treated SD rats (2-week treatment). The expression of adherens-junction protein E-cadherin decreased in colon tissue of rats after two weeks of 3DG administration. IOD values of E-cadherin (c) and ZO-1 (d) were measured using Image J 6.0 program; ** p < 0.01 (n = 6).
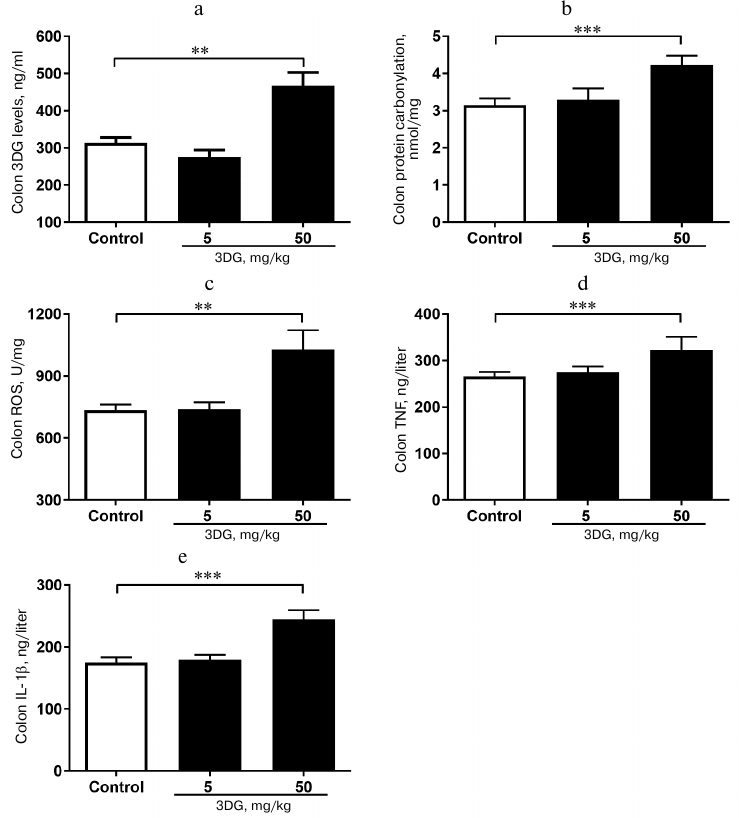
The elevation in 3DG content, protein carbonylation as well as ROS, TNF-α and IL-1β levels was observed in colon tissue of rats after two weeks of 3DG administration. Since bioavailability of 3DG is poor, we assessed whether 3DG is capable of accumulating in intestinal tissue. Our previous study [19] showed the increased 3DG content in intestinal tissue of rats after two weeks of 3DG administration. In accord with this, the present study demonstrated that the colon 3DG level was extremely increased in the 50 mg/kg 3DG-treated group compared to the control (Fig. 5a). To investigate the consequences of 3DG accumulation in intestinal tissue, we further evaluated the levels of protein carbonylation and ROS in colon. As shown in Fig. 5b, the protein carbonylation in colon was significantly increased in the 50 mg/kg 3DG-treated group compared to the control. There was an increase in the ROS level in colon of the 50 mg/kg 3DG-treated group compared to the control (Fig. 5c). In addition, both the colon TNF-α and IL-1β levels were significantly increased in the 50 mg/kg 3DG-treated group compared to the control (Figs. 5d and 5e).
Fig. 5. The elevation in 3DG content, protein carbonylation as well as ROS, TNF-α and IL-1β levels was observed in colon tissue of rats after two weeks of 3DG administration. 3DG (a), protein carbonylation (b), ROS (c), TNF-α (d) and IL-1β (e) colon levels were measured in rats fasted overnight, after 2-week 3DG treatment; **p < 0.01, p < 0.001 (n = 6).
DISCUSSION
Our previous study has demonstrated 3DG accumulation in intestinal tissue of rats after 2-week 3DG administration by gastric gavage. Consistently, the present study showed that 3DG content in colon tissue of rats after two weeks of intragastric 3DG administration was significantly increased. As expected, 2-week administration of 3DG induced an increase in ROS production and protein carbonylation in colon tissue of rats that was accompanied by an enhanced intestinal permeability characterized by decreased E-cadherin expression. The TNF-α and IL-1β levels were also elevated in colon tissue of 3DG-treated rats. Thus, the 3DG-treated rats have an elevation in systemic oxidative stress and inflammatory cytokines expression. Furthermore, the increased ROS production and inflammatory cytokines expression were also observed in pancreatic tissue of 3DG-treated rats that displayed obviously pancreatic β-cell dysfunction.
Increasing evidence suggests that the assembly of tight junctions between epithelial cells requires the prior formation of adherens junctions, and thus alteration of the cadherin-dependent adherens junctions modulates tight junction formation and epithelial paracellular barrier function [29-31]. Besides, recent studies have revealed that the adherens junction was more vulnerable than tight junction [32, 33]. Our results showed the decreased expression of adherens-junction protein E-cadherin in colon tissue of 3DG-treated rats that was accompanied by colon 3DG deposition. Although assessment of protein expression by immunohistochemical analysis revealed no significant changes in the manifestation of ZO-1, we speculate that changes in the arrangement of the tight junctions might explain the increased permeability caused by 3DG. Several reports have demonstrated that a broad array of agents can cause or directly contribute to alterations in intestinal permeability by several mechanisms, i.e., by increasing intestinal oxidative stress resulting in elevated ROS production, by pathogenic bacteria, and by regulating lipopolysaccharide level [34-36]. ROS damage cell membranes via peroxidation, formation of MDA and open connections between enterocytes [9, 10, 25]. Our present results showed the increased ROS level in the colon of 3DG-treated rats, suggesting that 3DG deposition in colon can induce damage of cell membranes and open connections between enterocytes through ROS that can modify structure and function of proteins. Beyond this, a class of small molecule compounds, which can directly disturb gut barrier function by regulating proteins of intercellular junctions, was reported [34, 37]. 3DG was shown to directly modify amino acid residues in proteins, suggesting that 3DG deposition in colon may affect junction proteins’ conformation, activity, and function via carbonyl stress [38]. Additionally, oxidative stress is closely linked with intestinal inflammation that participates in triggering alterations of both junction proteins [39, 40]. Our data show the increased TNF-α and IL-1β levels in colon tissue of 3DG-treated rats that contributes to the enhanced intestinal permeability. RCS-induced carbonyl stress also can increase ROS levels [38, 39]. Taking together, our results demonstrate that 3DG-treated rats display increased intestinal permeability; this phenomenon is linked to carbonyl and oxidative stress. As intestine permeability increases, intestinal inflammatory factors can get released into the circulation. Conversely, the increased systemic inflammation observed in 3DG-treated rats can evidence in favor of the enhanced intestinal permeability due to less absorption of 3DG. Numerous studies have shown that metabolic syndrome is closely coupled with alterations in intestinal permeability [34, 36]. We observed pancreatic β-cell dysfunction in the 3DG-treated rats that agrees with the literature. Among the mechanisms involved in this phenomenon, the extension of systemic and pancreatic inflammation and oxidative stress observed in 3DG-administered rats were proposed, since the effects of oxidative stress and inflammation in pancreatic β-cells are well known.
The term “carbonyl stress” describes an imbalance between the formation and removal of RCS [43]. 3DG, one of the RCS, was observed to be deposited in colon tissue of rats after 2-week exogenous 3DG administration, suggesting that carbonyl stress in the intestine was induced by exogenous 3DG in our experiments. The reaction of RCS with amino acids could lead both to ROS and AGE formation [44]. 3DG was reported to directly and rapidly induce ROS generation in vitro [18]. This could be due to the 3DG ability to modify and inactivate ROS-scavenging enzymes (superoxide dismutases, glutathione peroxidases, and glutathione transferases) by cross-linking protein molecules and modification of their biological properties [45]. Thus, it is likely that the 3DG-induced carbonyl stress can increase ROS levels. Indeed, increased ROS levels in intestinal tissue were observed in 3DG-treated rats with induced carbonyl stress. It is known that ROS are not only participants of signaling pathways but also lead to carbonylation of particular amino acid side chains by direct metal-catalyzed oxidation [1]. Thus, an excessive ROS level can further aggravate carbonyl stress. Thus, 3DG deposition in colon induced the carbonyl stress, and the 3DG-induced carbonyl stress could increase ROS level, aggravating the carbonyl stress.
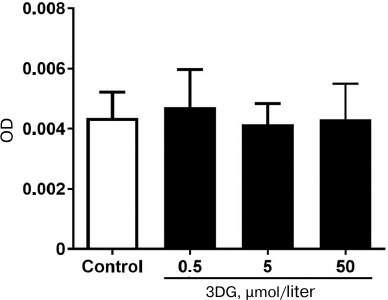
Carbonylation of proteins is a permanent process that may alter function or structure of proteins and has a broad spectrum of downstream results. It has been shown that alteration of proteins structure and function plays a central role in the development of chronic complications in diabetes [1, 46]. In the present study we found that 3DG accumulation in intestinal tissue increased protein carbonylation that is one of the main markers of carbonyl and oxidative stress. Carbonyl groups are indirectly introduced into proteins via non-oxidative covalent binding of RCS to side chains of the nucleophilic amino acids such as arginine, lysine, and cysteine. These RCS are derived from hydroxyl radical-mediated oxidation of lipids (e.g., 4-hydroxy-trans-2,3-nonenal (4-HNE), MDA, acrolein) and from autoxidation of carbohydrates (e.g., glyoxal, methylglyoxal, 3DG) [47]. 3DG accumulated in intestinal tissue is indirectly introduced into proteins. ROS selectively occur at the surface of proteins within “hot spots” of carbonylation enriched with the susceptible amino acids arginine, lysine, proline or threonine, and this process apparently strengthens itself since carbonylation increases the reactivity of neighboring carbonylatable sites [48]. Thus, the excessive level of ROS enhanced introduction of carbonyl groups into proteins. Meanwhile, protein carbonylation assay was used to detect protein carbonylation at various 3DG concentrations. The optical density of protein samples with various concentrations of 3DG did not statistically differ compared to the control. This result implied that free 3DG did not affect the protein carbonylation assay (Fig. 6). Thus, protein carbonylation induced by 3DG could be mediated by the carbonyl stress and ROS formation.
Fig. 6. Free 3DG (0.5, 5 and 50 μΜ) does not affect protein carbonylation.
Taken together, 3DG induced ROS production. It not just modified or inactivated the ROS-scavenging enzymes but also promoted protein carbonylation. Then, the ROS further promoted protein carbonylation. The reactions of α-dicarbonyl compounds with amino acids lead to both ROS and AGE formation, which could contribute to the development and progression of diabetic complications.
In summary, we report for the first time that 3DG deposition in colon triggers carbonyl and oxidative stress and consequently impairs gut permeability. The enhanced intestinal permeability by 3DG deposition in colon causes systemic and pancreatic oxidative stress and inflammation, contributing to the development of pancreatic β-cell dysfunction. These results also suggest that intestine can be the novel target tissue for 3DG, which links exogenous 3DG intake with systemic pathological changes. These results provide an insight into a potential pathway linking intake of some non-digestible dietary ingredients with development of pancreatic β-cell dysfunction. Our results also suggest that suppression of 3DG-induced carbonyl stress could be a novel therapeutic strategy to potentially mitigate adverse effects of 3DG, a drug that is used for treatment and prevention of prediabetes.
Funding
This work was supported by the research funds from Suzhou Youth Science and Education Project (KJXW2017043), Youth project of Suzhou TCM Hospital (YQN2015003), Jiangsu Province’s Key Provincial Talents Program (QNRC2016259) and Suzhou Science and Technology Department (SYSD2017191).
Author Contributions
All authors contributed to the study concept and design and the interpretation of the data. L. Zhou, X. D. Song, and G. R. Jiang wrote the first draft of the manuscript. All authors edited the manuscript and approved the final version. G. R. Jiang is responsible for the integrity of the work as a whole.
Conflict of Interests
The authors declare no conflicts of interests.
Ethical Approval
All experimental protocols were approved by the local Ethics Committee of Suzhou Hospital of Traditional Chinese Medicine. All experimental procedures with animals were conducted in compliance with the Guide for the Care and Use of Laboratory Animals.
REFERENCES
1.Hecker, M., and Wagner, A. H. (2018) Role of
protein carbonylation in diabetes, J. Inherit. Metab. Dis.,
41, 29-38.
2.Wang, C. H., Wu, S. B., Wu, Y. T., and Wei, Y. H.
(2013) Oxidative stress response elicited by mitochondrial dysfunction:
implication in the pathophysiology of aging, Exp. Biol. Med.
(Maywood), 238, 450-460.
3.Gerber, P. A., and Rutter, G. A. (2017) The role of
oxidative stress and hypoxia in pancreatic beta-cell dysfunction in
diabetes mellitus, Antioxid. Redox Signal., 26,
501-518.
4.Keane, K. N., Cruzat, V. F., Carlessi, R., de
Bittencourt, P. I., and Newsholme, P. (2015) Molecular events linking
oxidative stress and inflammation to insulin resistance and β-cell
dysfunction, Oxid. Med. Cell. Longev., 2015, 181643.
5.Miyata, T., van Ypersele de Strihou, C., Kurokawa,
K., and Baynes, J. W. (1999) Alterations in nonenzymatic biochemistry
in uremia: origin and significance of “carbonyl stress” in
long-term uremic complications, Kidney Int., 55,
389-399.
6.Aldini, G., Dalle-Donne, I., Facino, R. M.,
Milzani, A., and Carini, M. (2007) Intervention strategies to inhibit
protein carbonylation by lipoxidation-derived reactive carbonyls,
Med. Res. Rev., 27, 817-868.
7.Brun, P., Castagliuolo, I., Di, L. V., Buda, A.,
Pinzani, M., Palu, G., and Martines, D. (2007) Increased intestinal
permeability in obese mice: new evidence in the pathogenesis of
nonalcoholic steatohepatitis, Am. J. Physiol. Gastrointest. Liver
Physiol., 292, G518-525.
8.Cani, P. D., Bibiloni, R., Knauf, C., Waget, A.,
Neyrinck, A. M., Delzenne, N. M., and Burcelin, R. (2008) Changes in
gut microbiota control metabolic endotoxemia-induced inflammation in
high-fat diet-induced obesity and diabetes in mice, Diabetes,
57, 1470-1481.
9.Muccioli, G. G., Naslain, D., Backhed, F.,
Reigstad, C. S., Lambert, D. M., Delzenne, N. M., and Cani, P. D.
(2010) The endocannabinoid system links gut microbiota to adipogenesis,
Mol. Syst. Biol., 6, 392.
10.Hall, D. M., Buettner, G. R., Oberley, L. W., Xu,
L., Matthes, R. D., and Gisolfi, C. V. (2001) Mechanisms of circulatory
and intestinal barrier dysfunction during whole body hyperthermia,
Am. J. Physiol. Heart Circ. Physiol., 280, H509-521.
11.Dong, M., Hu, N., Hua, Y., Xu, X., Kandadi, M.
R., Guo, R., Jiang, S., Nair, S., Hu, D., and Ren, J. (2013) Chronic
Akt activation attenuated lipopolysaccharide-induced cardiac
dysfunction via Akt/GSK3β-dependent inhibition of apoptosis and ER
stress, Biochim. Biophys. Acta, 1832, 848-863.
12.Yu, L., Zhai, Q., Tian, F., Liu, X., Wang, G.,
Zhao, J., Zhang, H., Narbad, A., and Chen, W. (2016) Potential of
Lactobacillus plantarum CCFM639 in protecting against aluminum
toxicity mediated by intestinal barrier function and oxidative stress,
Nutrients, 8, E783.
13.Davis, B. T., Voigt, R. M., Shaikh, M., Forsyth,
C. B., and Keshavarzian, A. (2017) CREB protein mediates
alcohol-induced circadian disruption and intestinal permeability,
Alcohol Clin. Exp. Res., 41, 2007-2014.
14.Degen, J., Hellwig, M., and Henle, T. (2012)
1,2-Dicarbonyl compounds in commonly consumed foods, J. Agric. Food
Chem., 60, 7071-7079.
15.Lin, J. A., Wu, C. H., Lu, C. C., Hsia, S. M.,
and Yen, G. C. (2016) Glycative stress from advanced glycation end
products (AGEs) and dicarbonyls: an emerging biological factor in
cancer onset and progression, Mol. Nutr. Food Res., 60,
1850-1864.
16.Turk, Z. (2010) Glycotoxins, carbonyl stress and
relevance to diabetes and its complications, Physiol. Res.,
59, 147-156.
17.Okado, A., Kawasaki, Y., Hasuike, Y., Takahashi,
M., Teshima, T., Fujii, J., and Taniguchi, N. (1996) Induction of
apoptotic cell death by methylglyoxal and 3-deoxyglucosone in
macrophage-derived cell lines, Biochem. Biophys. Res. Commun.,
225, 219-224.
18.Che, W., Asahi, M., Takahashi, M., Kaneto, H.,
Okado, A., Higashiyama, S., and Taniguchi, N. (1997) Selective
induction of heparin-binding epidermal growth factor-like growth factor
by methylglyoxal and 3-deoxyglucosone in rat aortic smooth muscle
cells. The involvement of reactive oxygen species formation and a
possible implication for atherogenesis in diabetes, J. Biol.
Chem., 272, 18453-18459.
19.Kato, H., van Chuyen, N., Shinoda, T., Sekiya,
F., and Hayase, F. (1990) Metabolism of 3-deoxyglucosone, an
intermediate compound in the Maillard reaction, administered orally or
intravenously to rats, Biochim. Biophys. Acta, 1035,
71-76.
20.Zhang, L., Song, X., Zhou, L., Liang, G., Xu, H.,
Wang, F., Huang, F., and Jiang, G. (2016) Accumulation of intestinal
tissue 3-deoxyglucosone attenuated GLP-1 secretion and its
insulinotropic effect in rats, Diabetol. Metab. Syndr.,
8, 78.
21.Wang, F., Zhou, L., Song, X., Liang, G., Xu, H.,
Zhang, L., and Jiang, G. (2017) Acute reduction of incretin effect and
glucose intolerance in rats by single intragastric administration of
3-deoxyglucosone, Exp. Clin. Endocrinol. Diabetes, 125,
4-11.
22.Jiang, G. L., Zhang, L. V., Wang, F., Xu, H., and
Fei, D. (2011) Synthesis and structure analysis of the 3-deoxyglucosone
(3-DG), J. Soochow Univ., 27, 60-68.
23.Guide for the Care and Use of Laboratory
Animals (2011) Eighth edition, The National Academies Press,
Washington, DC.
24.Hwang, I., An, B. S., Yang, H., Kang, H. S.,
Jung, E. M., and Jeung, E. B. (2013) Tissue-specific expression of
occludin, zona occludens-1, and junction adhesion molecule A in the
duodenum, ileum, colon, kidney, liver, lung, brain, and skeletal muscle
of C57BL mice, J. Physiol. Pharmacol., 64, 11-18.
25.Wang, Q., and Brubaker, P. L. (2002)
Glucagon-like peptide-1 treatment delays the onset of diabetes in
8-week-old db/db mice, Diabetologia, 45, 1263-1273.
26.Eizirik, D. L., Colli, M. L., and Ortis, F.
(2009) The role of inflammation in insulitis and beta-cell loss in type
1 diabetes, Nat. Rev. Endocrinol., 5, 219-226.
27.Gerber,P. A., and Rutter,G. A. (2017) The role of
oxidative stress and hypoxia in pancreatic beta-cell dysfunction in
diabetes mellitus, Antioxid. Redox Signal., 26,
501-518.
28.Lugrin, J., Rosenblatt-Velin, N., Parapanov, R.,
and Liaudet, L. (2014) The role of oxidative stress during inflammatory
processes, Biol.Chem., 395, 203-30.
29.Man, Y., Hart, V. J., Ring, C. J., Sanjar, S.,
and West, M. R. (2000) Loss of epithelial integrity resulting from
E-cadherin dysfunction predisposes airway epithelial cells to
adenoviral infection, Am. J. Respir. Cell Mol. Biol., 23,
610-617.
30.Siliciano, J. D., and Goodenough, D. A. (1988)
Localization of the tight junction protein, ZO-1, is modulated by
extracellular calcium and cell–cell contact in Madin–Darby
canine kidney epithelial cells, J. Cell Biol., 107,
2389-2399.
31.Troxell, M. L., Gopalakrishnan, S., McCormack,
J., Poteat, B. A., Pennington, J., Garringer, S. M., Schneeberger, E.
E., Nelson, W. J., and Marrs, J. A. (2000) Inhibiting cadherin function
by dominant mutant E-cadherin expression increases the extent of tight
junction assembly, J. Cell. Sci., 113 (Pt. 6),
985-996.
32.Gassler, N., Rohr, C., Schneider, A., Kartenbeck,
J., Bach, A., Obermuller, N., Otto, H. F., and Autschbach, F. (2001)
Inflammatory bowel disease is associated with changes of enterocytic
junctions, Am. J. Physiol. Gastrointest. Liver Physiol.,
281, G216-228.
33.Hanby, A. M., Chinery, R., Poulsom, R., Playford,
R. J., and Pignatelli, M. (1996) Downregulation of E-cadherin in the
reparative epithelium of the human gastrointestinal tract, Am. J.
Pathol., 148, 723-729.
34.Bischoff, S. C., Barbara, G., Buurman, W.,
Ockhuizen, T., Schulzke, J. D., Serino, M., Tilg, H., Watson, A., and
Wells, J. M. (2014) Intestinal permeability – a new target for
disease prevention and therapy, BMC Gastroenterol., 14,
189.
35.Bein, A., Zilbershtein, A., Golosovsky, M.,
Davidov, D., and Schwartz, B. (2017) LPS induces hyper-permeability of
intestinal epithelial cells, J. Cell. Physiol., 232,
381-390.
36.Tran, C. D., Grice, D. M., Wade, B., Kerr, C. A.,
Bauer, D. C., Li, D., and Hannan, G. N. (2015) Gut permeability, its
interaction with gut microflora and effects on metabolic health are
mediated by the lymphatics system, liver and bile acid, Future
Microbiol., 10, 1339-1353.
37.Sequeira, I. R., Kruger, M. C., Hurst, R. D., and
Lentle, R. G. (2015) Ascorbic acid may exacerbate aspirin-induced
increase in intestinal permeability, Basic Clin. Pharmacol.
Toxicol., 117, 195-203.
38.Niwa, T. (1999) 3-Deoxyglucosone: metabolism,
analysis, biological activity, and clinical implication, J.
Chromatogr. B Biomed. Sci. Appl., 731, 23-36.
39.Vargas-Robles, H., Citalan Madrid, A. F.,
Garcia-Ponce, A., Silva-Olivares, A., Shibayama, M., Betanzos, A., Del
Vaalle Mondragon, L., Nava, P., and Schnoor, M. (2016) Experimental
colitis is attenuated by cardioprotective diet supplementation that
reduces oxidative stress, inflammation, and mucosal damage, Oxid.
Med. Cell. Longev., 2016, 8473242.
40.Paszti-Gere, E., Csibrik-Nemeth, E., Szeker, K.,
Csizinszky, R., Jakab, C., and Galfi, P. (2012) Acute oxidative stress
affects IL-8 and TNF-α expression in IPEC-J2 porcine epithelial
cells, Inflammation, 35, 994-1004.
41.Roberts, M. J., Wondrak, G. T., Laurean, D. C.,
Jacobson, M. K., and Jacobson, E. L. (2003) DNA damage by carbonyl
stress in human skin cells, Mutat. Res., 522, 45-56.
42.Kang, J. H. (2003) Oxidative damage of DNA
induced by methylglyoxal in vitro, Toxicol. Lett.,
145, 181-187.
43.Rahbar, S., and Figarola, J. L. (2003) Novel
inhibitors of advanced glycation endproducts, Arch. Biochem.
Biophys., 419, 63-79.
44.Araki, A. (1997) Oxidative stress and diabetes
mellitus: a possible role of alpha-dicarbonyl compounds in free radical
formation, Nihon Ronen Igakkai Zasshi, 34, 716-720.
45.Niwa, T., Takeda, N., Yoshizumi, H., Tatematsu,
A., Ohara, M., Tomiyama, S., and Niimura, K. (1993) Presence of
3-deoxyglucosone, a potent protein crosslinking intermediate of
Maillard reaction, in diabetic serum, Biochem. Biophys. Res.
Commun., 196, 837-843.
46.Almogbel, E., and Rasheed, N. (2017) Protein
mediated oxidative stress in patients with diabetes and its associated
neuropathy: correlation with protein carbonylation and disease activity
markers, J. Clin. Diagn. Res., 11, BC21-21BC25.
47.Adams, S., Green, P., Claxton, R., Simcox, S.,
Williams, M. V., Walsh, K., and Leeuwenburgh, C. (2001) Reactive
carbonyl formation by oxidative and non-oxidative pathways, Front.
Biosci., 6, A17-24.
48.Maisonneuve, E., Ducret, A., Khoueiry, P.,
Lignon, S., Longhi, S., Talla, E., and Dukan, S. (2009) Rules governing
selective protein carbonylation, PLoS ONE, 4, e7269.