Mechanisms of Leptin and Ghrelin Action on Maturation and Functions of Dendritic Cells
E. G. Orlova1,a*, S. V. Shirshev1, and O. A. Loginova1
1Perm Federal Research Center, Institute of Ecology and Genetics of Microorganisms, Ural Branch of the Russian Academy of Sciences, 614081 Perm, Russia* To whom correspondence should be addressed.
Received July 3, 2018; Revised September 17, 2018; Accepted September 17, 2018
Molecular mechanisms of the immunomodulatory effects of leptin and ghrelin in concentrations typical for pregnancy on the maturation and functional activity of dendritic cells (DCs) generated from the peripheral blood monocytes of women are investigated. The presence of leptin during DC maturation did not affect the levels of CD83+CD1c+, CD86+CD1c+, and HLA-DR+CD1c+ DCs, but increased the amount and the activity of the enzyme indoleamine 2,3-dioxygenase (IDO). Cell culturing in the presence of ghrelin or combination of leptin and ghrelin reduced the percentage of CD86+CD1c+ DCs but did not affect the levels of CD83+CD1c+ and HLA-DR+CD1c+ DCs. In addition, ghrelin reduced the number of IDO molecules without affecting its activity. Simultaneous presence of leptin and ghrelin increased induced IDO activity without affecting the amount of the enzyme in DCs. The effects of leptin and ghrelin on the investigated functions of DCs in some cases correlated with high levels of cAMP. New mechanisms for leptin and ghrelin regulation of tolerogenic functions of DCs in pregnancy are proposed.
KEY WORDS: leptin, ghrelin, pregnancy, dendritic cells, IDO, cAMPDOI: 10.1134/S0006297919010012
Abbreviations: AC, adenylate cyclase; AMPK, AMP-activated protein kinase; CaM, calmodulin; cAMP, 3′,5′-cyclic adenosine monophosphate; CDs, clusters of differentiation, molecule expressed on the surface of immune system cells; CREB, cAMP response element-binding (CRE); DAG, diacylglycerol; DC, dendritic cell; Epac, exchange protein directly activated by cAMP; ERK, extracellular signal-regulated kinase; GHS-R, receptor of ghrelin; HLA-DR, human leukocyte antigen DR; IDO, indoleamine 2,3-dioxygenase; IL, interleukin; IP3, inositol 1,4,5-triphosphate; ITIM2, immunoreceptor tyrosine-based inhibitory motifs 2; JAK, Janus kinase; JNK, c-Jun N-terminal kinase; LepR, receptor of leptin; LPS, lipopolysaccharide; MAPK, mitogen-activated protein kinase; MyD88, myeloid differentiation primary response 88; NF-κB, nuclear factor kappa-light-chain-enhancer of activated B cells; p38MAPK, p38 mitogen-activated protein kinase; PDE3B, phosphodiesterase 3B; PI3K, phosphoinositide 3-kinase; PIP2, phosphatidylinositol 4,5-bisphosphate; PKA, protein kinase A; PKB(Akt), protein kinase B; PKC, protein kinase C; PLC, phospholipase C; SOCS3, suppressors of cytokine signaling 3; STAT3, signal transducer and activator of transcription-3; TLR-4, Toll-like receptor-4.
Dendritic cells (DCs) are the main antigen-presenting cells which
initiate and direct the immune response. The maturity and functional
activity of DCs determine the direction of naive CD4+ T helper (Th)
differentiation and the type of immune response, contributing both to
the activation of effector T cells against foreign agents and immune
response inhibition via potentiating the generation of inducible
regulatory T cells (iTreg) that determine the tolerance [1, 2].
Pregnancy is an exciting example of tolerance development by the mother’s immune system toward the semiallogenic fetus that occurs with the preservation of protective properties of mother’s organism as a whole [3]. During normal pregnancy, the proportion of immature DCs capable of inducing the state of immune tolerance increases in the peripheral blood [1, 3]. These cells are characterized by reduced expression of costimulatory/coactivator molecules (CD40, CD80, CD83, CD86), high levels of production of anti-inflammatory cytokines [interleukin (IL)-10 and transforming growth factor-β1 (TGF-β1)], and increased activity of the enzyme indolamine-2,3-dioxygenase (IDO) [1-3]. Tolerogenic (immature) DCs inhibit T cell response via promoting generation of iTreg cells and reducing the level of T helpers producing IL-17 (Th17), thereby causing anergy and apoptosis of cytotoxic T cells [3].
Unlike cytokines, IDO is a leading factor in the induction of peripheral tolerance during pregnancy [1-4]. The enzyme is expressed by syncytiotrophoblasts, decidual membrane cells, placental DCs, and macrophages [5]. IDO is an inducible enzyme that converts L-tryptophan into kynurenine, 3-hydroxykynurenine, and 3-hydroxyanthranilic acid [1-4]. This reaction leads to the deficiency of tryptophan, which is necessary for protein translation in activated T cells. The products of tryptophan degradation induce apoptosis of Th1 and cytotoxic T lymphocytes [1-6]. IDO inhibition in pregnant mice leads to the induction of anti-fetal immune response [7]. In addition, kynurenine can shift the Th17/Treg ratio toward Treg cells [8]. Therefore, high IDO activity provides one of the key mechanisms for the maternal-fetal tolerance formation [9].
Placental hormones can modulate the functional activity of leukocytes due to the expression of specific receptors on these cells [10-13]; hence, hormones can be considered as physiological inducers of tolerogenic DCs. Peptide hormones leptin and ghrelin are functional antagonists in the regulation of feeding behavior and glucose metabolism; they control energy homeostasis, as well as functioning of the immune and reproductive systems [10]. By acting on hypothalamus, leptin and ghrelin oppositely control hunger and appetite and regulate food consumption, adipose tissue metabolism, energy homeostasis, growth, and development [10]. Both hormones exhibit the immunoregulatory activity [13-16]. The levels of leptin and ghrelin in the peripheral blood significantly increase during pregnancy, because both hormones are actively produced by the placenta. They are necessary for successful fetus implantation and development [10]. Receptors for leptin (LepR) and ghrelin (GHS-R) have been found on DCs [12, 17, 18] and most cells of the immune system [17-22]. Leptin has the proinflammatory properties and promotes the dominance of the cell-mediated immune response [17, 19]. Ghrelin, on the contrary, displays the anti-inflammatory activity by blocking the leptin-induced proinflammatory response [12]. Both hormones can mutually regulate expression of receptors for each other on the cells [12, 20]. The combined action of leptin and ghrelin in a cell and at the organism level produces the cooperative effects that connect energy homeostasis and immune functions [10, 16, 20]. Leptin enhances maturation of DCs by promoting expression of CD40, production of IL-12, and ability to initiate Th1 response [17, 18, 21]. We have previously shown that leptin and ghrelin, in concentrations characteristic of pregnancy, oppositely modulate the synthesis of IL-12, IL-10, and TGF-β1 by DC and mediate DC differentiation of iTreg and Th17 cells [14-16], as well as regulate the IDO activity in the peripheral blood monocytes [22]. Elucidation of the hormonal mechanisms involved in the control of DC functions is important for understanding the patterns of immune response development and immune tolerance formation during pregnancy and can be the most efficient approach to the induction of antigen-specific tolerance to donor, recipient, or own antigens.
Analysis of published data shows that the effects of most placental hormones include inhibition of cytotoxic responses, formation of the Th2-type immune response, and generation of suppressor T cells in the utero-placental contact zone via stimulation of the cAMP-dependent signaling [23]. Our previous studies have shown that the cAMP signaling pathways are involved in the immunomodulatory effects of ghrelin and combination of ghrelin with leptin on lymphocytes [15]. According to other authors, cAMP acts as a secondary messenger in the leptin action on different types of cells [24, 25]. All of the above facts determine the need to evaluate the role of cAMP in the effects of leptin and ghrelin in the regulation of DC functions.
The aim of this study was to investigate the ability of leptin and ghrelin in concentrations typical for pregnancy to influence maturation of DCs, as well as the amount and activity of IDO, and to assess the role cAMP in these processes.
MATERIALS ANS METHODS
DCs were generated from the peripheral blood monocytes of healthy non-pregnant women of reproductive age (19-39 years) (n = 10). Blood samples were collected during the follicular phase of the menstrual cycle (1-7 days); peripheral blood mononuclear cells (PBMCs) were isolated by centrifugation in a Ficoll–verografin density gradient (1.077 g/cm3; Sigma, USA; Spofa, Czechia). DCs were generated by the standard method [26]. PBMCs ((1-5)·106 cells/ml) were resuspended in complete nutrition media (CNM; RPMI-1640 supplemented with 10% fetal calf serum, 1 mM HEPES, 2 mM L-glutamine, and 100 μg/ml streptomycin), plated in 24-well polystyrene plates (2 ml per well; Costar, USA), and cultured for 3 h at 37°C in 5% CO2 (3-h incubation time was sufficient for monocytes to adhere to the plastic surface). The non-adherent PBMCs were then washed off with cold RPMI-1640 and 2 ml of CNM with hormones was added to the wells. Leptin (Sigma, USA) was used at a concentration of 35 ng/ml, which is close to its concentration in the peripheral blood during the second and third trimesters of gestation [27]. Ghrelin (Sigma, Israel) was added at a concentration 1.25 ng/ml, which is comparable with its concentration in the peripheral blood during the first and second trimesters of gestation [28]. To investigate the combined effect of both hormones, they were added to the cells simultaneously (leptin, 35 ng/ml; ghrelin, 1.25 ng/ml). Physiologic saline used for dissolving the hormones was added to the control samples.
Formation of DCs was induced by adding granulocyte-macrophage colony-stimulating factor (GM-CSF) (100 ng/ml; R&D, USA) and IL-4 (20 ng/ml; BioLegend, USA) to the cells [26]. The cells were then incubated at 37°C in 5% CO2 for 5 days. After 3 days of culturing, a portion of the medium was replaced with the new one containing the same cytokines (IL-4 and GM-CSF) and lipopolysaccharide (LPS, 1 μg/ml, E. coli serotype 0111:B4; Sigma) to induce DC maturation [26]. After 5 days of culturing, the supernatants were removed, and the DC phenotype was assessed by flow cytometry using a FACSCalibur flow cytometer (Becton Dickinson, USA). Cells viability evaluated in the test with eosin after 120-h incubation with the hormones was 95-98%. The gating strategy included initial gating on the monocyte populations from the forward (FSC-H) and side scatter (SSC-H) plot, followed by exclusion of non-viable cells and then gating on viable cells and determination of specific DC markers in this region. Non-specific binding and fluorescence-negative window were determined using the corresponding isotype controls (mouse IgG1-FITC, IgG1-PE, IgG2b-PE, IgG2a-PE, and IgG1 kappa-FITC; eBio, USA). At least 105 cells were analyzed. Expression of the surface CD14, CD83, CD86, HLA-DR, and CD1c molecules reflecting the level of DC maturation was estimated in gate of monocyte cells using the corresponding monoclonal antibodies (anti-human CD14-FITC 61D3/mouse IgG1, anti-human CD83-PE HB15e/mouse IgG1, anti-human CD86-PE IT2.2/mouse IgG2b, anti-human HLA-DR-PE L243/mouse IgG2a, anti-human CD1c-FITC L161/mouse IgG1 kappa; eBio, USA).
To assess the effect of hormones on DC maturation, the number of CD83+, CD86+, HLA-DR+ in gate CD1c+ DC was studied [29].
The amount of IDO in lysates of DCs was determined by enzyme immunoassay (EIA) using commercial ELISA kit for IDO (Cloud-Clone Corp, USA) according to the manufacturer’s instructions.
To determine the IDO activity, L-tryptophan (100 μM; Sigma) was added to the samples. The samples were incubated for 4 h, and the supernatants were then collected and assayed for the concentration of kynurenine, the first stable product of tryptophan degradation [2]. LPS (100 ng/ml, E. coli serotype 0111:B4; Sigma) was used to induce IDO activity [30]. The level of kynurenine in the samples was evaluated spectrophotometrically by a standard method [2]. Spontaneous production of kynurenine in the studied samples was 1.63 ± 0.54 μM.
The levels of intracellular cAMP in the lysates of generated DCs were determined by EIA using a Direct cAMP ELISA kit (Enzo Life Sciences, USA) according to the manufacturer’s instructions.
Statistical analysis of the results was performed using the STATISTICA (v. 11.0) software package. The normality of data distribution was tested using the χ2-test. If the data followed the normal distribution, the statistical significance was determined using the Student’s paired t-test. The non-parametric Wilcoxon matched pairs test was used if the data were not normally distributed. The differences were considered significant at p < 0.05. For multiple comparisons, Bonferroni correction were used. Some of the results were processed using the Pearson’s (r) correlation analysis.
RESULTS AND DISCUSSION
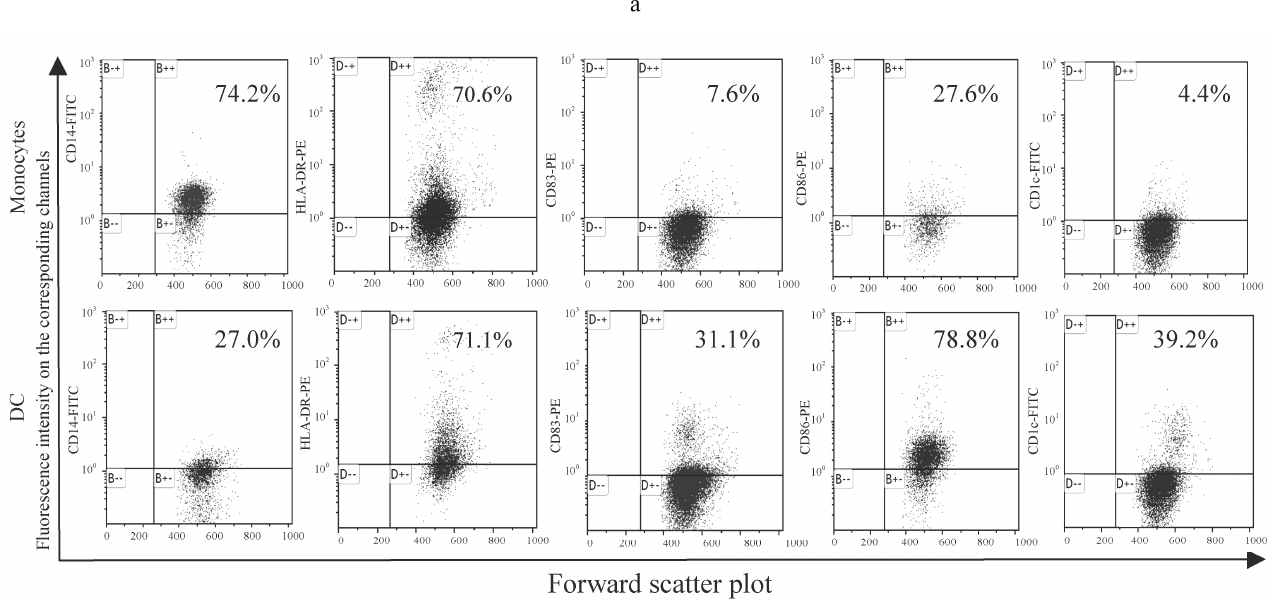
DC generation. Culturing monocytes in the presence of IL-4 and GM-CSF led to the formation of DCs with a typical phenotype characterized by the loss of CD14 (common monocyte marker) and increased expression of HLA-DR (major histocompatibility complex class II molecule), CD1c (antigen- presenting molecule), CD86 (costimulatory molecule), and CD83 (marker of mature DCs). Culturing monocytes in the absence of IL-4 and GM-CSF resulted in the increased expression of both CD14 and HLA-DR (Fig. 1). The levels of HLA-DR expression on the monocytes and maturing DCs were comparable, whereas the amounts of CD1c, CD83, and CD86 on maturing DCs were significantly lower. It should be noted that CD1c, also called blood dendritic cell antigen 1 (BDCA-1), is the main marker of classical (myeloid) DCs, including most DCs circulating in the peripheral blood and DCs generated from monocytes in vitro [29, 30]. Therefore, the used procedure successfully generated DCs from the peripheral blood monocytes.
Fig. 1. Expression of the membrane molecules CD14, HLA-DR, CD83, CD86, and CD1c on the monocytes and mature DCs obtained from monocytes by stimulation with IL-4 and GM-CSF followed by LPS activation. a) Histograms representing expression of CD14, HLA-DR, CD83, CD86, and CD1c on the monocytes and mature DCs. Ordinate axis, fluorescence intensity on the corresponding channels (FL); abscissa axis, forward scatter plot (FSC-H). Histograms from a typical experiment are presented. b) Expression of the membrane molecules CD14, HLA-DR, CD83, CD86, and CD1c on the monocytes and mature DCs (control). Abscissa axis, studied groups: M (monocytes), C (control) (mature DCs obtained from monocytes by stimulation with IL-4 and GM-CSF with subsequent activation by LPS); ordinate axis, percentage of cells carrying the corresponding marker; n = 10; the data are presented as mean ± standard error (M ± m); * statistically significant difference with the control (p < 0.05) according to the paired Student t-test.
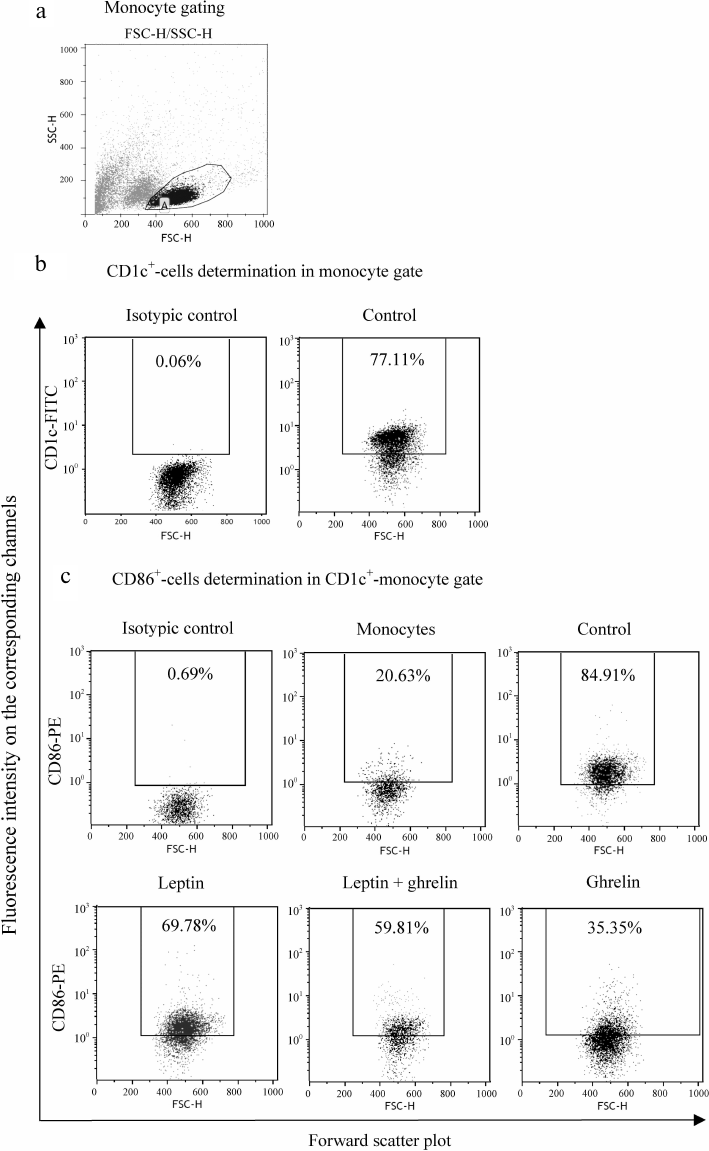
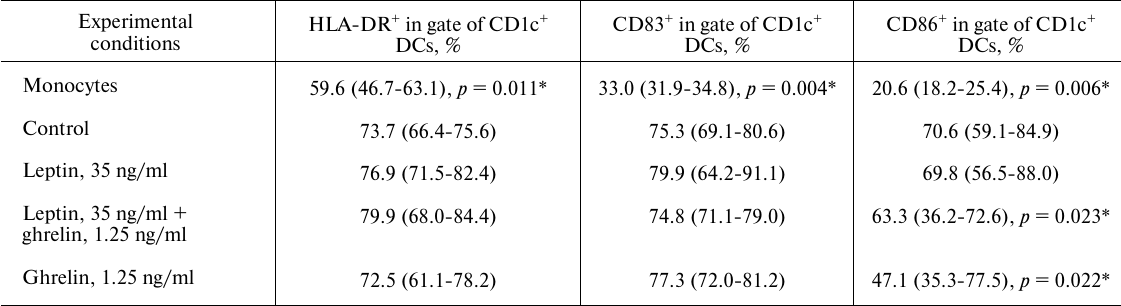
Effect of leptin on the maturation and functional activity of DCs. It is known that in homozygous ob/ob (leptin mutation) and db/db (leptin receptor mutation) mice, expression of the CD40, CD80, CD86, and HLA-DR maturation markers on DCs is reduced [31]. Injection of leptin in these animals restores the mature phenotype of DCs. Studies of the ability of leptin to generate human DCs in vitro demonstrated that the presence of this hormone in the concentrations characteristic of pregnancy has no statistically significant effect on the expression of HLA-DR, CD83, and CD86 on the membrane of cultured CD1c+ DCs (Fig. 2 and Table 1). Apparently, during physiological pregnancy, leptin does not realize its potential in contrast to the experimental models using mice with genetic defects. It is known that mature (myeloid) DCs take an active part in the immune response against antigenic structures upon transplantation [7]. The absence of the stimulating effect of leptin suggests that the hormone concentration during pregnancy is not sufficient to exhibit its action.
Fig. 2. Histograms characterizing the gating strategy (a, b) and CD86 expression on CD1c+ DCs under the influence of hormones (c). Ordinate axis, fluorescence intensity on the corresponding channels (FL); abscissa axis, forward scatter plot (FSC-H). Histograms from a typical experiment are presented. Designations: Control, mature DCs (obtained from monocytes via stimulation with IL-4 and GM-CSF followed by LPS activation); Leptin, mature DCs cultured in the presence of leptin, 35 ng/ml; Leptin + ghrelin, mature DCs cultured in the presence of leptin (35 ng/ml) and ghrelin (1.25 ng/ml); Ghrelin, mature DCs cultured in the presence of ghrelin, 1.25 ng/ml.
Table 1. Effects of leptin and ghrelin on
the expression of membrane molecules on CD1c+ DCs generated
in the presence of the hormones
Note: The data are presented as median with lower and upper quartiles
[IU (LQ; UQ)]. Here and in Table 2: Monocytes,
monocytes cultured in the absence of IL-4, GM-CSF, and LPS; Control,
mature DCs (obtained from the monocytes via stimulation with IL-4 and
GM-CSF followed by LPS activation); Leptin, 35 ng/ml, mature DCs
cultured in the presence of leptin, 35 ng/ml; Leptin, 35 ng/ml +
Ghrelin, 1.25 ng/ml, mature DCs cultured in the presence of leptin (35
ng/ml) and ghrelin (1.25 ng/ml); Ghrelin, 1,25 ng/ml, mature DCs
cultured in the presence of ghrelin, 1.25 ng/ml.
* Statistically significant differences with the control according to
the Wilcoxon matched pairs test for paired dependent samples; n
= 10.
The presence of leptin during DC maturation increased both the amount and the LPS-stimulated activity of IDO, thereby promoting L-tryptophan metabolism with the formation of kynurenine (Table 2). At the same time, no increase in the intracellular cAMP concentration was observed in vitro. Therefore, it can be concluded that during pregnancy, leptin does not affect the maturation of myeloid DCs, but rather enhances their tolerogenic potential through upregulation of IDO synthesis and, as a consequence, activation of L-tryptophan catabolism to kynurenine, which promotes anergy and death of effector T lymphocytes in the uteroplacental interface area. Previously, we have shown that leptin produces similar effect on the LPS-stimulated activity of IDO in the peripheral blood monocytes [22].
Table 2. Effects of leptin and ghrelin on
the amount and LPS-induced activity of IDO and intracellular cAMP
concentration in DCs generated in the presence of the hormones

Note: The results are presented as arithmetic mean ± standard
error (M ± SEM); * statistically significant differences with
the control according to the Student’s paired t-test;
n = 10.
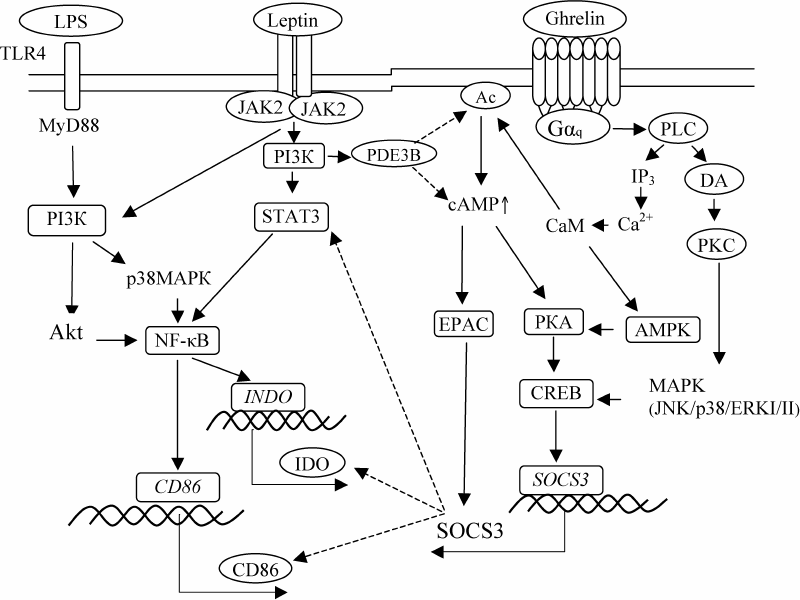
It should be noted that LPS and interferon-γ are the most important factors initiating DC maturation and IDO expression during pregnancy [32]. LPS is constantly present in the urinary tract as a product of bacterial contamination. LPS interaction with the Toll-like receptors 4 (TLR-4) on DCs leads to the activation of MyD88 (myeloid differentiation primary response 88 protein) and PI3K (phosphatidylinositide 3-kinase), as well as Akt (protein kinase B) and p38MAPK (mitogen-activated protein kinase) [30, 33]. Both Akt and p38MAPK induce activation of the nuclear transcription factor κB (NF-κB) [33], that directly binds to the INDO promoter and activates IDO expression [30]. Leptin interaction with LepR on DCs activates Jak2 kinases (Janus kinases) that phosphorylate IRS-2 protein (insulin receptor substrate 2) and initiate the PI3K/Akt signaling pathway [34, 35]. Phosphorylation of Akt kinase simultaneously activates MAPK and STAT3 (signal transduction protein and transcription activator 3), which in its dimeric form promotes NF-κB activation [21]. Therefore, leptin can enhance the IDO-inducing action of LPS through PI3K, MAPK, and STAT3, which explains our results.
Effect of ghrelin on the maturation and tolerogenic function of DCs. The presence of ghrelin in the concentration characteristic for pregnancy during DC maturation reduces CD86 expression on CD1c+ DCs but does not affect the amounts of CD83+CD1c+ and HLA-DR+CD1c+ DCs (Fig. 2 and Table 1). Taking into account that CD86 is a co-stimulating molecule necessary for the interaction with CD28 on T-lymphocytes and strengthening of the activating signal, it can be assumed that the action of ghrelin during pregnancy reduces T-lymphocyte activation or their anergy.
Unlike leptin, ghrelin did not affect the LPS-stimulated IDO activity during DC maturation but reduced the amount of the enzyme in these cells (Table 2). This effect was directly related to the increase in the intracellular cAMP concentration. Correlation analysis revealed the inverse relationship between the decrease in the IDO amount in the presence of ghrelin and increase in the cAMP concentration (r = –0.95; p < 0.05); i.e., ghrelin prevented an increase in the IDO amount proportionally to the increase in the intracellular cAMP level. It is known that ghrelin interaction with GHS-R, which belongs to the Gq-protein-coupled receptors (GqPCRs), leads to the activation of phospholipase C (PLC) and hydrolysis of phosphatidylinositol 4,5-bisphosphate (PIP2) to inositol 1,4,5-triphosphate (IP3) resulting in the increase in the Ca2+ and diacylglycerol (DAG) concentrations [36-38]. DAG activates PKC (protein kinase C), leading to the upregulation of the MAPK cascade activity (JNK/p38/ERKI/II) and phosphorylation of CREB (cAMP response element-binding protein) [36-39]. CREB induces transcription and synthesis of SOCS3 (suppressor of cytokine signal transduction 3) that binds to the ITIM2 (inhibiting phosphorylation motif of tyrosine 2) domain of IDO, thereby inducing proteasomal degradation of the IDO molecule [40, 41]. The increase in the cAMP intracellular level under the influence of ghrelin is probably due to the ability of Ca2+/CaM (calmodulin) to activate adenylate cyclase (AC) [42]. The increase in the cAMP concentration in the cells results in the activation of protein kinase A (PKA) and EPAC (exchange protein directly activated by cAMP), which also promotes SOCS3 synthesis [42]. In addition, CaM stimulates the activity of AMP-activated protein kinase (AMPK) [43] leading to the formation of active CREB. Hence, downregulation of the LPS-induced IDO expression under the influence of ghrelin in DCs can be explained by the cAMP/CREB/SOCS3-mediated proteasomal degradation of the enzyme [44]. Apparently, the SOCS3-dependent mechanism also determines the reduction of the CD86 expression on the CD1c+ DCs matured in the presence of ghrelin. According to the literature, SOCS3 can directly induce proteasomal degradation of CD86, as well as promote degradation of activated transcription factors responsible for CD86 expression [45, 46].
Combined effect of leptin and ghrelin on the maturation and functions of DCs. Blood cells simultaneously experience the influence of a large number of signaling molecules present in the peripheral blood; therefore, analysis of the combined effects of hormones, especially, a pair of hormones with antagonistic action, might help us to understand the physiological mechanisms of pregnancy. It is found that simultaneous presence of leptin and ghrelin during DC maturation in concentrations typical for pregnancy reduces expression of CD86 on CD1c+ DCs but does not affect the number of CD83+CD1c+ and HLA-DR+CD1c+ DCs (Fig. 2 and Table 1). According to the correlation analysis, the decrease in the amount of CD86+CD1c+ DCs in the presence of both hormones was caused by ghrelin (r = 0.75; p < 0.05). As mentioned above, ghrelin reduces CD86 expression on CD1c+ DCs via cAMP-dependent activation of CREB/SOCS3, which is also realized upon the combined action of the hormones [39, 40].
Analysis of the leptin and ghrelin combined effect on the activity and amount of IDO in DCs showed that the presence of hormones during DC maturation does not affect the amount of the enzyme by increases its LPS-stimulated activity in DCs, which is also accompanied by the increase in the kynurenine concentration (Table 2). According to the correlation analysis, the increase in the LPS-stimulated activity of IDO upon the combined action of the hormones was caused by leptin (r = 0.95; p < 0.05). This suggests that in the presence of ghrelin, leptin retains only its ability to activate IDO, since ghrelin prevents leptin-induced accumulation of new enzyme molecules due to their ghrelin/SOCS3-induced proteasomal degradation [39, 40]. This assumption is consistent with the published data that ghrelin is able to significantly inhibit leptin-induced phosphorylation of STAT3 via EPAC-dependent activation of SOCS3 [39, 40]. At the same time, the combined presence leptin and ghrelin during DC maturation did not noticeably affect the intracellular cAMP level (Table 2). It is known that leptin is able to reduce the cAMP level by activating the PI3K-catalyzed reaction. The product of this reaction, phosphatidylinositol 3,4,5-triphosphate (PIP3), stimulates Akt, which in turn, phosphorylates membrane-associated phosphodiesterase 3B (PDE3B) [47]. This effect of leptin explains the absence of the increase in the intracellular cAMP concentration upon the combined action of the hormones. Apparently, in the presence of both hormones, ghrelin predominantly acts through the PKC/MAPK-dependent CREB phosphorylation and SOCS3 activation.
Therefore, despite the fact that leptin and ghrelin exert the opposite modulating effects on the main functions of DCs, stimulation of the LPS-induced activity of IDO by leptin and downregulation of the co-stimulating CD86 molecule expression on CD1c+ DCs by ghrelin are preserved upon the combined action of these hormones. Figure 3 shows possible mechanisms of the hormonal control of the maturation and functional activity of DCs.
Fig. 3. Hypothetical scheme for regulation of CD86 expression, amount and LPS-stimulated IDO activity in DCs by the combination of leptin and ghrelin. Solid arrows, stimulating effect; dotted arrows, inhibitory effect.
The obtained results expand and complement our understanding of the mechanisms of leptin and ghrelin participation in the regulation of the functional activity of DCs in pregnancy and in the formation of immune tolerance in general. During physiological pregnancy, it is the combined effect of physiological concentrations of leptin and ghrelin in the peripheral blood that contributes to the activation of the IDO-dependent mechanism of peripheral tolerance induction with the involvement of DCs and predominant formation of iTreg cells. Understanding molecular mechanisms of the interaction between leptin and ghrelin is promising for pharmacological correction of DC differentiation and can also be used to develop efficient approaches to enhance the induction of specific tolerance to donor/recipient histocompatibility antigens or to own antigens.
Funding
This work was supported by the Russian Foundation for Basic Research (projects nos. 11-04-01109 and 13-04-00571).
Conflicts of Interest
The authors declare no conflicts of interest.
REFERENCES
1.Gregori, S. (2011) Dendritic cells in networks of
immunological tolerance, Tissue Antigens, 77, 89-99.
2.Braun, D., Longman, R. S., and Albert, M. L. (2005)
A two-step induction of indoleamine 2,3-dioxygenase (IDO) activity
during dendritic cell maturation, Blood, 106,
2375-2381.
3.Koldehoff, M., and Elmaagacli, A. H. (2013)
Thoughts on feto-maternal tolerance: is there a lesson to be learned
from allogeneic haematopoietic stem cell transplantation? Cell Biol.
Int., 37, 766-767.
4.Miwa, N., Hayakawa, S., Miyazaki, S., Myojo, S.,
Sasaki, Y., Sakai, M., Takikawa, O., and Saito, S. (2005) IDO
expression on decidual and peripheral blood dendritic cells and
monocytes/macrophages after treatment with CTLA-4 or interferon-gamma
increase in normal pregnancy but decrease in spontaneous abortion,
Mol. Hum. Reprod., 11, 865-870.
5.Kamimura, S., Eguchi, K., Yonezawa, M., and Sekiba,
K. (1991) Localization and developmental change of indoleamine
2,3-dioxygenase activity in the human placenta, Acta. Med.
Okayama., 45, 135-139.
6.Fallarino, F., Grohmann, U., Vacca, C., Bianchi,
R., Orabona, C., Fioretti, M. C., and Puccetti, P. (2002) T cell
apoptosis by tryptophan catabolism, Cell. Death Differ.,
9, 1069-1077.
7.Munn, D. H., Zhou, M., Attwood, J. T., Bondarev,
I., Conway, S. J., Marshall, B., Brown, C., and Mellor, A. L.
(1998) Prevention of allogeneic fetal rejection by tryptophan
catabolism, Science, 281, 1191-1193.
8.Baban, B., Chandler, P. R., Sharma, M. D., Pihkala,
J., Koni, P. A., Munn, D. H., and Mellor, A. L. (2009) IDO
activates regulatory T cells and blocks their conversion into Th17-like
T cells, J. Immunol., 183, 2475-2483.
9.Kudo, Y., Boyd, C. A., Sargent, I. L., and Redman,
C. W. (2001) Tryptophan degradation by human placental
indoleamine 2,3-dioxygenase regulates lymphocyte proliferation, J.
Physiol., 535, 207-215.
10.Tena-Sempere, M. (2013) Interaction between
energy homeostasis and reproduction: effects of leptin and ghrelin on
the reproductive axis, Horm. Metab. Res., 45,
919-927.
11.Fantuzzi, G., and Faggioni, R. (2000) Leptin in
the regulation of immunity, inflammation, and hematopoiesis, J.
Leukoc. Biol., 68, 437-446.
12.Dixit, V. D., Schaffer, E. M., Pyle, R. S.,
Collins, G. D., Sakthivel, S. K., Palaniappan, R., Lillard, J. W., and
Taub, D. D. (2004) Ghrelin inhibits leptin- and activation-induced
proinflammatory cytokine expression by human T cells, J. Clin.
Invest., 1, 57-66.
13.Shirshev, S. V. (2015) Molecular mechanisms of
hormonal and hormonal-cytokine control of immune tolerance in
pregnancy, Biochemistry (Moscow). Ser. A. Membr. Cell Biol.,
9, 21-40.
14.Orlova, E. G., Shirshev, S. V., and Loginova, O.
A. (2015) Leptin and ghrelin regulate dendritic cell maturation and
dendritic cell induction of regulatory T-cells, Dokl. Biol. Sci.,
462, 171-174.
15.Orlova, E. G., and Shirshev, S. V. (2017) Role of
PKA and PI3K in leptin and ghrelin regulation of adaptive
subpopulations of regulatory CD4+ T-lymphocyte formation,
Biochemistry (Moscow), 82, 1061-1072.
16.Orlova, E. G., and Shirshev, S. V. (2014) Role of
leptin and ghrelin in induction of differentiation of IL-17-producing
and T-regulatory cells, Bull. Exp. Biol. Med., 156,
819-822.
17.Mattioli, B., Straface, E., Quaranta, M. G.,
Giordani, L., and Viora, M. (2005) Leptin promotes differentiation and
survival of human dendritic cells and licenses them for Th1 priming,
Immunology, 174, 6820-6828.
18.Mattioli, B., Giordani, L., Quaranta, M. G., and
Viora, M. (2009) Leptin exerts an anti-apoptotic effect on human
dendritic cells via the PI3K-Akt signaling pathway, FEBS Lett.,
583, 1102-1106.
19.Faggioni, R., Fantuzzi, G., Fuller, J., Feingold,
K. R., and Grunfeld, C. (1998) IL-1-beta mediates leptin induction
during inflammation, Am. J. Physiol., 274, 204-208.
20.Komori, T., Doi, A., Furuta, H., Wakao, H.,
Nakao, N., Nakazato, M., Senba, E., and Morikawa, Y. (2010) Regulation
of ghrelin signaling by a leptin-induced gene, negative regulatory
element-binding protein, in the neurons, J. Biol. Chem.,
285, 37884-37894.
21.Lam, Q. L., Zheng, B. J., Jin, D. Y., and Lu, L.
(2007) Leptin induces CD40 expression through the activation of Akt in
murine dendritic cells, J. Biol. Chem., 282,
27587-27597.
22.Orlova, E. G., and Shirshev, S. V. (2011)
Regulation of leptin and ghrelin of IDO activity of monocytes, Vest.
Ural Med. Akad. Nauk, 38, 161-162.
23.Shirshev, S. V. (2010) AMP-dependent mechanisms
of endocrine control of immune system in pregnancy, Usp. Sovrem.
Biol., 130, 26-30.
24.Amarilyo, G., Iikuni, N., Liu, A., Matarese, G.,
and La Cava, A. (2014) Leptin enhances availability of apoptotic
cell-derived self-antigen in systemic lupus erythematosus, PLoS
One, 9, e112826.
25.Sahu, M., Anamthathmakula, P., and Sahu, A.
(2015) Phosphodiesterase-3B-cAMP pathway of leptin signalling in the
hypothalamus is impaired during the development of diet-induced obesity
in FVB/N mice, J. Neuroendocrinol., 27, 293-302.
26.Talayev, V. Y., Matveichev, A. V., Lomunova, M.
A., Talayeva, M. V., Tsaturov, M. E., Zaichenko, I. Y., and Babaykina,
O. N. (2010) The effect of human placenta cytotrophoblast cells on the
maturation and T cell stimulating ability of dendritic cells in
vitro, Clin. Exp. Immunol., 162, 91-99.
27.Hardie, L., and Trayhurn, P. (1997) Circulating
leptin in women: longitudinal study in menstrual cycle and during
pregnancy, Clin. Endocrinol., 47, 101-106.
28.Fuglsang, J., Skjaerbaek, C., Espelund, U.,
Frystyk, J., Fisker, S., Flyvbjerg, A., and Ovesen, P. (2005) Ghrelin
and its relationship to growth hormones during normal pregnancy,
Clin. Endocrinol., 62, 554-559.
29.Dzionek, A., Fuchs, A., Schmidt, P., Cremer, S.,
Zysk, M., Miltenyi, S., Buck, D. W., and Schmitz, J. J. (2000) BDCA-2,
BDCA-3, and BDCA-4: three markers for distinct subsets of dendritic
cells in human peripheral blood, Immunology, 165,
6037-6046.
30.Jung, I. D., Lee, C. M., Jeong, Y. I., Lee, J.
S., Han, J., and Park, Y. M. (2007) Differential regulation of
indoleamine 2,3-dioxygenase by lipopolysaccharide and interferon gamma
in murine bone marrow derived dendritic cells, FEBS Lett.,
581, 1449-1456.
31.Moraes-Vieira, P. M., Larocca, R. A., Bassi, E.
J., Peron, J. P., Andrade-Oliveira, V., Wasinski, F., Araujo, R.,
Thornley, T., Quintana, F. J., Basso, A. S., Strom, T. B., and Camara,
N. O. (2014) Leptin deficiency impairs maturation of dendritic cells
and enhances induction of regulatory T and Th17 cells, Eur. J.
Immunol., 44, 794-806.
32.Hwang, S. L., Chung, N. P., Chan, J. K., and Lin,
C. L. (2005) Indoleamine 2,3-dioxygenase (IDO) is essential for
dendritic cell activation and chemotactic responsiveness to chemokines,
Cell Res., 15, 167-175.
33.Fujigaki, H., Saito, K., Fujigaki, S., Takemura,
M., Sudo, K., Ishiguro, H., and Seishima, M. (2006) The signal
transducer and activator of transcription 1alpha and interferon
regulatory factor 1 are not essential for the induction of indoleamine
2,3-dioxygenase by lipopolysaccharide: involvement of p38
mitogen-activated protein kinase and nuclear factor-kappaB pathways,
and synergistic effect of several proinflammatory cytokines, J.
Biochem., 139, 655-662.
34.Borges, B. C., Garcia-Galiano, D., Rorato, R.,
Elias, L. L., and Elias, C. F. (2016) PI3K p110β subunit in leptin
receptor expressing cells is required for the acute hypophagia induced
by endotoxemia, Mol. Metab., 5, 379-391.
35.Niswender, K. D., Gallis, B., Blevins, J. E.,
Corson, M. A., Schwartz, M. W., and Baskin, D. G. (2003)
Immunocytochemical detection of phosphatidylinositol 3-kinase
activation by insulin and leptin, J. Histochem. Cytochem.,
3, 275-283.
36.Mrak, E., Casati, L., Pagani, F., Rubinacci, A.,
Zarattini, G., and Sibilia, V. (2015) Ghrelin increases beta-catenin
level through protein kinase A activation and regulates OPG expression
in rat primary osteoblasts, Int. J. Endocrinol., 2015,
547473.
37.Kola, B., Hubina, E., Tucci, S. A., Kirkham, T.
C., Garcia, E. A., Mitchell, S. E., Williams, L. M., Hawley, S. A.,
Hardie, D. G., Grossman, A. B., and Korbonits, M. (2005) Cannabinoids
and ghrelin have both central and peripheral metabolic and cardiac
effects via AMP-activated protein kinase, J. Biol. Chem.,
280, 25196-25201.
38.Schellekens, H., Dinan, T. G., and Cryan, J. F.
(2013) Taking two to tango: a role for ghrelin receptor
heterodimerization in stress and reward, Front. Neurosci.,
7, 148.
39.Fujitsuka, N., Asakawa, A., Morinaga, A.,
Amitani, M. S., Amitani, H., Katsuura, G., Sawada, Y., Sudo, Y.,
Uezono, Y., Mochiki, E., Sakata, I., Sakai, T., Hanazaki, K. H., Asaka,
M., and Inui, A. (2016) Increased ghrelin signaling prolongs survival
in mouse models of human aging through activation of sirtuin1, Mol.
Psychiatry, 21, 1613-1623.
40.Yu, J., Wang, Y., Yan, F., Zhang, P., Li, H.,
Zhao, H., Yan, C., Yan, F., and Ren, X. (2014) Noncanonical NF-κB
activation mediates STAT3-stimulated IDO upregulation in
myeloid-derived suppressor cells in breast cancer, J. Immunol.,
193, 2574-2586.
41.Heldsinger, A., Grabauskas, G., Wu, X., Zhou, S.,
Song, I., and Owyang, C. (2014) Ghrelin induces leptin resistance by
activation of suppressor of cytokine signaling 3 expression in male
rats: implications in satiety regulation, Endocrinology,
155, 3956-3969.
42.Ferguson, G. D., and Daniel, R. S. (2004) Why
calcium-stimulated adenylyl cyclases? Physiology, 19,
271-276.
43.Bayliss, J. A., Lemus, M. B., Stark, R., Santos,
V. V., Thompson, A., Rees, D. J., Galic, S., Elsworth, J. D., Kemp, B.
E., Davies, J. S., and Andrews, Z. B. (2016) Ghrelin-AMPK signaling
mediates the neuroprotective effects of calorie restriction in
Parkinson’s disease, J. Neurosci., 36,
3049-3063.
44.Orabona, C., Pallotta, M. T., Volpi, C.,
Fallarino, F., Vacca, C., Bianchi, R., Belladonna, M. L., Fioretti, M.
C., Grohmann, U., and Puccetti, P. (2008) SOCS3 drives proteasomal
degradation of indoleamine 2,3-dioxygenase (IDO) and antagonizes
IDO-dependent tolerogenesis, Proc. Natl. Acad. Sci. USA,
105, 20828-20833.
45.Baravalle, G., Park, H., McSweeney, M.,
Ohmura-Hoshino, M., Matsuki, Y., Ishido, S., and Shin, J. S. (2011)
Ubiquitination of CD86 is a key mechanism in regulating antigen
presentation by dendritic cells, J. Immunol., 187,
2966-2973.
46.Ardeshna, K. M., Pizzey, A. R., Devereux, S., and
Khwaja, A. (2000) The PI3 kinase, p38 SAP kinase, and NF-kappa B signal
transduction pathways are involved in the survival and maturation of
lipopolysaccharide-stimulated human monocyte-derived dendritic cells,
Blood, 96, 1039-1046.
47.Fruhbeck, G. (2006) Intracellular signalling
pathways activated by leptin, Biochem. J., 393, 7-20.