Such Various Stem Cells
M. A. Lagarkova1,a
1Federal Research and Clinical Center of Physical-Chemical Medicine, Federal Medical-Biological Agency, 119435 Moscow, Russia
Received December 19, 2018; Revised December 24, 2018; Accepted December 24, 2018
Perhaps there is no more intriguing topic in modern biology than stem cells. The growing interest in stem cells is dictated by the ability of stem cells to both self-renew and differentiate, at least into several type cells. If we learn to influence these properties or reproduce them in vitro, it will be possible to effectively use stem cells or their differentiated derivatives in medicine. Fundamental knowledge of mechanisms of the self-maintenance and differentiation of stem cells is important for understanding a variety of processes – from embryogenesis to aging and oncogenic transformation. The purpose of this issue is to introduce readers to different areas in research on mammalian stem cells, including human stem cells. In the issue both review articles and research papers are presented, and the authors hope that they will be of interest for biochemists, cell biologists, and specialists in the field of biomedicine.
KEY WORDS: stem cells, mesenchymal stem cells, hematopoietic stem cells, embryonic stem cells, induced pluripotent stem cells, differentiationDOI: 10.1134/S0006297919030015
Abbreviations: ESCs, embryonic stem cells; iPSCs, induced pluripotent stem cells; MSCs, mesenchymal stem cells.
It is a subject of publications and discussions – who suggested
for the first time that stem cells should exist [1].
In the known professional online blog devoted to stem cells, this question is also considered in a separate paper (https://ipscell.com/2012/04/who-really-discovered-stem-cells-the-history-you-need-to-know/). Most likely, the term “stem cell” (“Stammzelle”) was proposed in 1868 by Valentine Hacker in relation to the germline cells [1].
Despite the lively discussions about the priorities, the huge contribution of two Russian scientists to the study of stem cells is recognized as indisputable. Alexander Maximov in the beginning of the last century proposed the existence of a stem blood cell [2]. More than a half-century later, Alexander Friedenstein for the first time isolated from bone marrow cells capable of differentiation into adipose, bone, and cartilage tissues, and later these cells were termed mesenchymal stem cells (MSCs) [3].
Stem cells are different in both their origin and differentiation potential (figure). In the adult organism of mammals there are no “universal” stem cells that would be able to differentiate into all types of tissues. Thus, in the bone marrow there are two types of stem cells: hematopoietic stem cells capable of differentiation into all blood cells and mesenchymal stem cells, which can give rise to bone, cartilage, and adipose tissues. MSCs also constitute an important part of the hematopoietic stroma, but under no circumstances can differentiate into blood cells. Hematopoietic stem cells were the first stem cells whose existence was confirmed by functional tests [4], and the first stem cells that began to be applied in medicine.
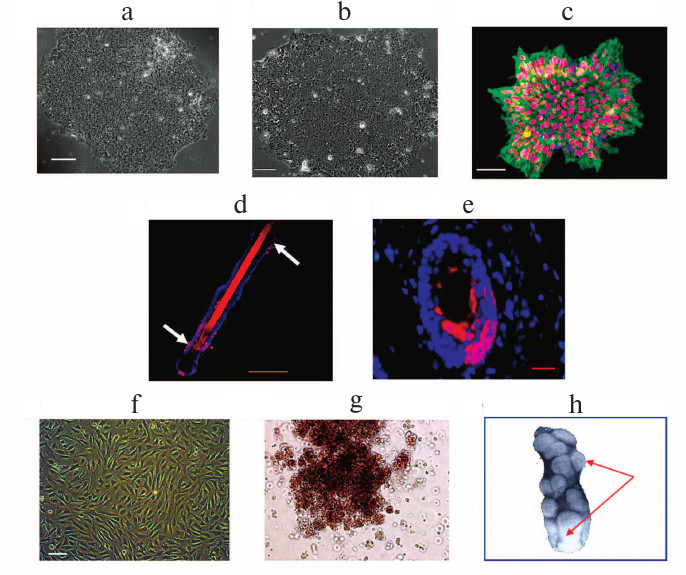
Such various stem cells. a) Colony of human embryonic stem cells (ESCs); b) colony of human induced pluripotent cells (iPSCs). Phase contrast. The scale ruler represents 100 μm. c) iPSC colony, immunofluorescent analysis for pluripotent cell markers: the transcription factor Nanog (red) and the surface antigen SSEA-4 (green). The nuclei are stained with DAPI (blue); d, e) localization of keratin 19-positive cells in a hair follicle (stained with red), immunofluorescent analysis: d) an entire follicle. White arrows indicate clusters of stem cells; e) the cross section. The nuclei are stained with DAPI. The scale ruler represents 500 μm; f) mesenchymal stem cells of human bone marrow in culture. Phase contrast. The scale ruler represents 100 μm; g) BFU-E/burst-producing erythroid unit (colony produced from a multipotent precursor cell in methylcellulose from CD34+-stem blood cells). Phase contrast; h) multipotent precursor blood cells after bone marrow transplantation produce colony-forming units (CFU-S) in the spleen of irradiated mice. CFU-S are indicated with arrows. Photos (a)-(c) and (g) were taken by the author. The photos (d) and (e) were kindly presented by E. S. Chermnykh and E. A. Vorotelyak. Photos (f) and (h) were kindly presented by N. I. Drize
Regional (adult) stem cells capable of differentiation into many type cells of definite tissues are present in the majority of tissues and organs, for instance, in skin, intestine, and brain. Stem cells of every specialization have their own niches, i.e., sets of microenvironment factors that provide both the rest state of the stem cell and its mobilization for division or differentiation if necessary. Traditionally, it was believed that the pool of stem cells within the same tissue should be homogenous: all stem cells should possess the equivalent potential of differentiation and self-renewal. However, recent data have shown that in many somatic cells the system of stem cells is surprisingly heterogenous and includes different types of stem cells [5].
In nature, cells capable of differentiation into all types of adult tissues exist only for a short time, during early embryogenesis until the gastrula stage, but they can be cultivated in vitro (embryonic stem cells) or obtained from differentiated cells by introducing four reprogramming transcription factors – Oct3/4, Sox2, Klf4, and c-Myc (induced pluripotent stem cells).
Cells of inner cell mass of the blastocyst cultured under laboratory conditions were termed embryonic stem cells (ESCs). Human and mouse ESCs are studied better than others [6, 7]. ESCs represent an artificially fixed transient state of the inner cell mass of the blastocyst. In the absence of differentiation signals and in the presence of factors supporting self-renewal, ESCs continue division without changing their morphological characteristics. By changing the cultivation conditions, it is possible to obtain directed differentiation of the ESCs into the cells that are derivatives of all three germ layers. Mouse ESCs became an indispensable tool for studying functions of genes, making it possible to obtain animals with genetic inactivation of a gene of interest. Human ESCs are a unique object for studying the early stages of embryogenesis. Moreover, there is a greatly increasing possibility of therapeutic application of technologies using differentiated derivatives of ESCs, for instance, the retinal pigment epithelium, neurons, and insulin-producing cells.
The technology of genetic reprogramming of somatic cells to the pluripotent state was developed in 2006 by the Japanese scientists Takahashi and Yamanaka [8]. This technology allows obtaining induced pluripotent stem cells (iPSCs)under laboratory conditions from somatic cells of adult organism, for instance, from skin fibroblasts. Similarly to ESCs, iPSCs can grow unlimited in culture and differentiate into every cell type. iPSCs have already become an important tool for modeling and studying human diseases, as well as for drug screening. Cells differentiated from iPSCs potentially can be used for replacement therapy in the treatment of diabetes, retinal degeneration, neurodegenerative pathologies, etc.
In this issue of the Biochemistry (Moscow), experimental works are presented with iPSCs used as objects under study: in some cases, they are used for studying functions of definite genes, in the other cases – for modeling diseases or as test-systems. One of the original works concerns an extremely important fundamental problem: what is the hierarchy of the bone marrow MSCs. Some reviews consider problems of origin of hematopoietic stem cells, grem cells, and of in vitro reproduction of their generation, as well as the role of the extracellular matrix in the self-maintenance and differentiation of stem cells.
Thus, the variety of stem cells and different aspects of their biology that are under study in modern biomedicine, as well as the wide spectrum of problems under consideration, prompted the compilers to entitle this special issue of the journal “Such Various Stem Cells”.
REFERENCES
1.Ramalho-Santos, M., and Willenbring, H. (2007) On
the origin of the term “stem cell”, Cell Stem Cell,
1, 35-38.
2.Maximow, A. A. (1909) Der Lymphozyt als gemeinsame
Stammzelle der verschiedenen Blutelemente in der embryonalen
Entwicklung und im postfetalen Leben der Saugetiere, Folia
Haematol., 8, 125-134.
3.Friedenstein, A. J., Chailakhjan, R. K., and
Lalykina, K. S. (1970) The development of fibroblast colonies in
monolayer cultures of guinea-pig bone marrow and spleen cells, Cell
Tissue Kinet., 3, 393-403.
4.Becker, A. J., McCulloch, E. A., and Till, J. E.
(1963) Cytological demonstration of the clonal nature of spleen
colonies derived from transplanted mouse marrow cells, Nature,
197, 452-454.
5.Goodell, M. A., Nguyen, H., and Shroyer, N. (2015)
Somatic stem cell heterogeneity: diversity in the blood, skin and
intestinal stem cell compartments, Nat. Rev. Mol. Cell Biol.,
16, 299-309.
6.Evans, M. J., and Kaufman, M. H. (1981)
Establishment in culture of pluripotential cells from mouse embryos,
Nature, 292, 154-156.
7.Thomson, J. A., Itskovitz-Eldor, J., Shapiro, S.
S., Waknitz, M. A., Swiergiel, J. J., Marshall, V. S., and Jones, J. M.
(1998) Embryonic stem cell lines derived from human blastocysts,
Science, 282, 1145-1147.
8.Takahashi, K., and Yamanaka, S. (2006) Induction of
pluripotent stem cells from mouse embryonic and adult fibroblast
cultures by defined factors, Cell, 126, 663-676.