The Repertoire of Human Antiglycan Antibodies and Its Dynamics in the First Year of Life
N. R. Khasbiullina1,2,a,b*, N. V. Shilova2, M. J. Navakouski2, A. Yu. Nokel2, O. Blixt3, L. O. Kononov1, Yu. A. Knirel1, and N. V. Bovin2
1Zelinsky Institute of Organic Chemistry, Russian Academy of Sciences, 119991 Moscow, Russia2Shemyakin–Ovchinnikov Institute of Bioorganic Chemistry, Russian Academy of Sciences, 117997 Moscow, Russia
3University of Copenhagen, T4221871 Frederiksberg, Denmark
* To whom correspondence should be addressed.
Received November 23, 2018; Revised March 18, 2019; Accepted March 19, 2019
The repertoire of antiglycan antibodies of peripheral blood was studied using a microarray containing 487 glycan antigens: fragments of mammalian glycans (N- and O-chains of glycoproteins, as well as glycolipids) and also bacterial polysaccharides. The sera samples correspond to the third, sixth, and twelfth months of life. The infants were divided into four groups according to their nutrition type: breast milk, standard formula, and partially or extensively hydrolyzed formula. During the first year of life, the total amount of IgG decreased; presumably, the lifetime of maternal IgG in the newborns’ bloodstream is much greater than is generally assumed. At the same time, the IgM content was low during the first six months and increased significantly by the twelfth month. The antiglycan IgM repertoire of one-year-old infants was still different from that of their mothers, as well as from the repertoire of unrelated donors, in particular, by the absence of antibodies against the Galβ1-3GlcNAc (LeC) disaccharide, which is found in almost all healthy humans. It is noteworthy that the level of IgM of breast-fed infants was significantly lower than that of formula-fed by the twelfth month.
KEY WORDS: natural antibodies, glycans, polysaccharides, bacteria, glycochip, array, infant nutrition, innate immunityDOI: 10.1134/S0006297919060038
Abbreviations: BM, breast milk; EHF, extensively hydrolyzed formula (milk); nAbs, natural antibodies; PHF, partially hydrolyzed formula (milk); SF, standard formula (milk).
It is generally accepted that foreign antigens cause the formation of
antibodies that are known as adaptive, and their levels and affinities
are directly related to the presence of these antigens. At the same
time, extensive experimental material has accumulated about antibodies
whose level and affinity for the antigen are practically constant
throughout life [1, 2]. These
immunoglobulins, in contrast to the adaptive ones, are known as natural
antibodies (nAbs). nAbs, being part of innate immunity, mediate various
functions such as protection from pathogens, clearance of metabolites,
surveillance of transformed cells, and regulation [3-7]. All classes of biomolecules
can be found among the antigens recognized by nAbs; this is explained
by the importance of the corresponding protective processes [3, 4]. In the context of nAbs
origin, early age is of particular interest, since the immune system of
a child has a special status: despite the very early onset of the
development of immunity in ontogenesis, it is functionally immature;
the child receives immunoglobulins G (IgG) from the mother’s
blood system, and there are practically no immunoglobulins M (IgM)
in the child’s blood [8-12].
It is generally believed that nAbs are primed largely under the influence of the intestinal microflora; therefore, the study of the relationship between the developing immunity, nutrition, and the microbial community is of great interest [13-15]. However, model organisms, in particular mice, with their specific microbiota composition, are not adequate models for studying human nAbs [16, 17]. Here we investigated the dynamics of antibody repertoires in children aged 3, 6, and 12 months, taking into account the effect of their nutrition. Pathogenic microorganisms possess a myriad of antigens, the structure of their glycoconjugates differing dramatically from those of mammals; hence, it is not surprising that nAbs against carbohydrate epitopes constitute an essential, if not dominant, part of their repertoire [18-20]. This allows us to make general immunological conclusions based on the study of antiglycan nAbs. At the same time, it is risky to make generalizations based on the study of only a few antibodies. Therefore, we profiled the nAbs using a representative array of 487 antigens.
MATERIALS AND METHODS
Microarrays were prepared from 341 different synthetic amine-functionalized glycans and 146 bacterial O-polysaccharides, using N-hydroxysuccinimide-derivatized glass slides (slide H; Schott-Nexterion, Germany), as described elsewhere [21]. The glycan library included blood group antigens and some of the most frequently occurring terminal oligosaccharides, as well as core motifs of mammalian N- and O-linked glycoproteins and glycolipids, tumor-associated carbohydrate antigens, and bacterial polysaccharides mainly as a carbohydrate part of lipopolysaccharides. Synthetic glycan structures (>95% purity, generally synthetized in the Laboratory of Carbohydrates, Institute of Bioorganic Chemistry, Russian Academy of Sciences) are structurally the same as natural ones. Structures, NMR data of polysaccharides, and related references can be found in http://csdb.glycoscience.ru/bacterial (Zelinsky Institute of Organic Chemistry, Russia). The glycans at concentrations of 50 µM and 10 µg/ml (for oligo- and polysaccharides, respectively) were printed in 6-12 replicates. A complete list of printed glycans can be found in Table S1 of the Supplement to this report on the journal website (http://protein.bio.msu.ru/biokhimiya) and Springer site (Link.springer.com). Two chips from each batch were analyzed using standard Complex Immunoglobulin Preparation (CIP; Immunogem, Russia) at a concentration of 1 mg/ml with biotinylated goat-anti-human Abs (Thermo Fisher Scientific, USA) at concentration of 10 µg/ml followed by streptavidin–Alexa Fluor 555 Conjugate (Thermo Fisher Scientific) at concentration of 1 μg/ml. Batches of printed microarrays with intra- and inter-chip correlation more than 0.9 were used. After printing, glycochips were blocked for 90 min at 25°C with blocking buffer (100 mM boric acid, 25 mM ethanolamine, 0.2% (v/v) Tween 20, pH 8.5) (Sigma-Aldrich, USA), which was then washed out with milli-Q grade water (Mediana-Filtr, Russia) and dried by the air using a Galaxy Mini-Array centrifuge (VWR International, South Korea). Blocked microarrays were stored at –20°C for further analysis. Before serum analysis, microarrays were incubated in an incubation chamber (Simport, Canada) for 15 min at 25°C with PBS (Sigma-Aldrich) plus 0.1% (v/v) Tween 20 (buffer 1), and the buffer was then carefully removed from the microchip surface using Whatman filter paper (Sigma-Aldrich). Human sera were diluted 1 : 15 in PBS plus 1% (w/v) BSA (Sigma-Aldrich) and 0.1% (v/v) Tween 20 (buffer 2). Diluted serum was spread over the slide surface and incubated with agitation (32-36 rpm) at 37°C for 90 min. After the incubation and a round of washing steps with buffer 1 and buffer 3 (PBS with 0.01% v/v Tween 20), the microarrays were incubated for 1 h at 37°C (32-36 rpm) with a mixture of goat-anti-human IgG conjugated to Alexa Fluor 555 and goat-anti-human IgM conjugated to Alexa Fluor 647 (Thermo Fisher Scientific) at a concentration of 8 μg/ml (each) in buffer 2. After another round of washing (buffer 1, buffer 3, and finally milli-Q grade water), the microarrays were dried by airflow using the Galaxy Mini-Array centrifuge. The microarrays were scanned using a ScanArray Gx scanner (PerkinElmer, USA) using excitation wavelength 543 and 635 nm. The resulting data were processed using ScanArray Express 4.0 software and the fixed 70 µm-diameter circle method as well as Microsoft Excel software. From 6 to 12 spot replicates represent each oligosaccharide or polysaccharide on the array, and data are reported as median RFU (relative fluorescent units) of replicates. Median deviation was measured as interquartile range. A signal as RFU exceeding the fluorescence intensity of the background value by a factor of five was considered as significant.
The blood of healthy adult donors, children aged 3, 6, and 12 months, as well as their mothers, was obtained by a standard fence from the ulnar vein. When selecting donors for the study, pathology of gestation, congenital diseases, and infections were excluded.
RESULTS
Antiglycan IgG and IgM during the first year of life. We used a glycan microarray to study the repertoires of children’s antibodies at ages of 3, 6, and 12 months and those of their mothers immediately after delivery. The children were divided into groups according to the type of nutrition: breast milk (BM), standard formula milk (SF), as well as two specialized types: extensively hydrolyzed (EHF) and partially hydrolyzed (PHF) formulas.
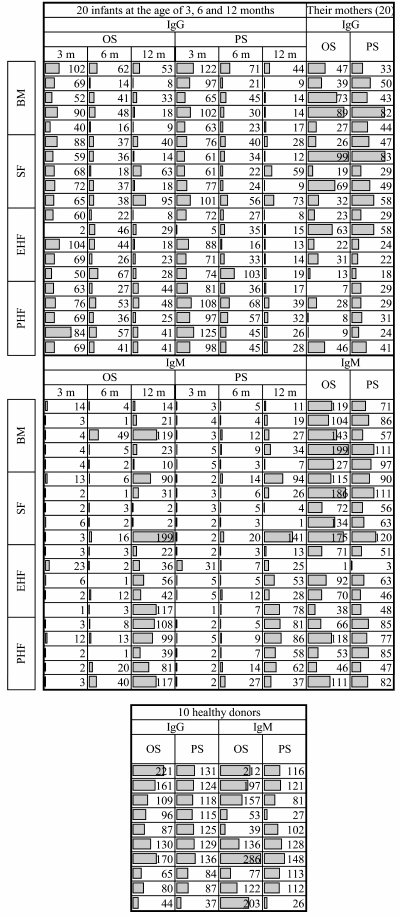
During the first year of life, IgG diversity decreases. The opposite tendency is characteristic for IgM: at the age of 3 and 6 months, antiglycan IgM is practically undetectable, but at the age of 12 months the repertoire of antibodies of this class is diverse and comparable to adults. Figure 1 shows the time course of antiglycan IgG and IgM diversity (the specificity will be considered below) in the sera of children during the first year of life. Each bar represents the total number of antigens to which serum antibodies are bound, and also for comparison, the corresponding data for mothers and unrelated healthy adult donors are presented. The latter group was necessary because pregnant and lactating women are characterized by physiological immunodeficiency [22, 23].
Fig. 1. Dynamics of antiglycan IgG and IgM in the sera of 3-, 6-, and 12-month-old infants with different types of nutrition, and in the sera of their mothers, and in 10 healthy unrelated adult donors. Each number represents the absolute number of glycans (oligo- and polysaccharides) that bind antibodies in the sera samples. OS, oligosaccharides; PS, polysaccharides; 3m, 6m, and 12m, age of children (months) at the time of blood sampling; BM, breast milk; SF, standard formula; PHF and EHF, partially and extensively hydrolyzed formula, respectively.
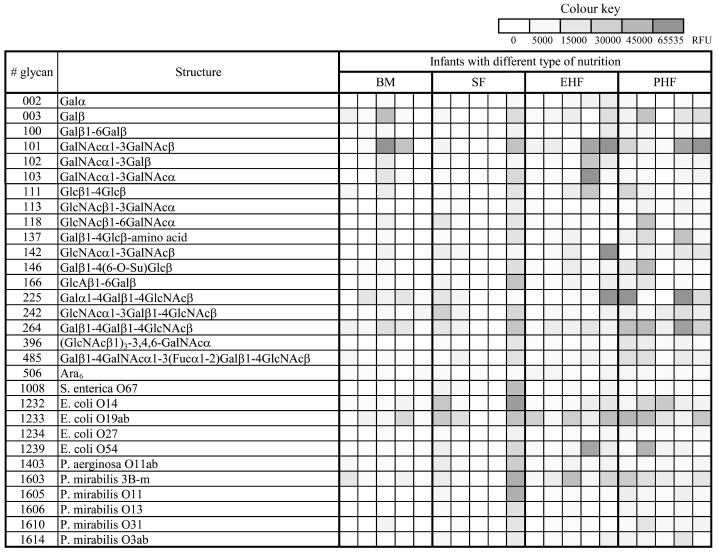
IgM repertoires in groups of infants who received different types of nutrition. Tendencies towards a decrease in the diversity of antiglycan IgG and the appearance of antiglycan IgM by only 12 months are characteristic of all children whose blood serums were studied in this work, regardless of what type of nutrition they received during the first year of life. At the age of 3 and 6 months, IgM is practically undetectable in children, and at 12 months, the repertoire of antiglycan antibodies of this class is comparable in diversity to an adult (Fig. 1). It should be noted that among bacterial polysaccharides to which there are antibodies, neither structural nor generic similarity is found; apparently, the appearance of antibodies to them occurs individually. In Fig. 2, a list of glycans is presented in the form of a color map demonstrating the maximum IgM binding signals in the blood sera of one-year-old children.
Fig. 2. Glycans showed maximal fluorescence signals of binding with IgM in sera of 12-month-old infants who received different type of nutrition. The data are represented as a thermal map where a darker shade of red color corresponds to a higher fluorescent signal. Designations as in Fig. 1 legend.
Comparison of the repertoires of IgM of infants receiving different types of nutrition and those of adult donors showed that infants fed with PHF exhibited the greatest variety of glycan-binding immunoglobulins, which is typical of adults. The table shows such a comparison for oligosaccharides and Table S2 (Supplement) for polysaccharides. Supplementary Table S3 contains all fluorescence signals for IgM capable of binding glycans in the serum of infants, their mothers, and unrelated adults.
Comparison of IgM repertoires in 12-month-old children and unrelated
adult donors
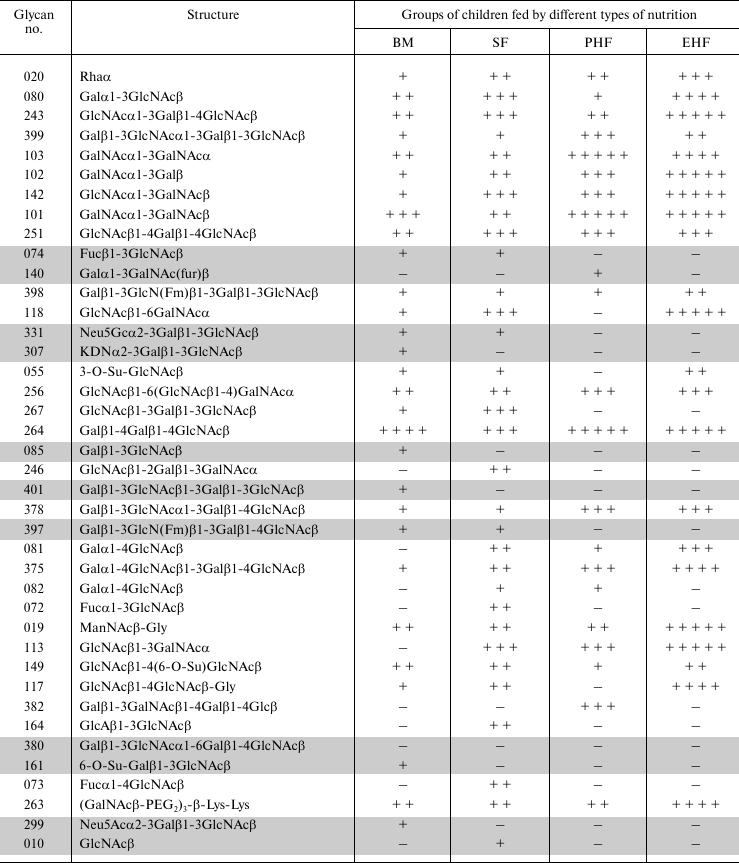
Note: The table represents 40 oligosaccharides whose signals for binding
to antibodies have the maximum intensity in adult donors (glycans are
sorted in descending order of the signal; for 10 donors, the median of
the signal values is measured). The right side of the
table presents the signals of binding of antibodies to these glycans in
the serum of 12-month-old children who received different types of
nutrition. The presence of a meaningful binding signal for each child
is indicated as “+”. The binding of antibodies to glycans,
highlighted in gray in the table, is practically absent in children.
It should be noted that almost all glycans (highlighted in the table) against which adults, but not 12-month-old children, have antibodies are structurally related to the LeC disaccharide (Galβ1-3GlcNAcβ). Earlier, we have described nAbs with this specificity as anti-LeC antibodies [24, 25].
IgG repertoires in the groups of infants who received different types of nutrition. The data are shown in Table S4 (Supplement). There are two reasons why we omit detailed discussion of IgG: (i) the nAbs mainly belong to IgM; (ii) it is impossible to discriminate between infant and maternal IgG, because IgG can cross the placental barrier during intrauterine development [26].
DISCUSSION
By the time of birth, the B-cell system of immunity is morphologically formed. In contrast, functional maturity develops for a rather long time after birth, almost until adolescence [9, 27]. However, the most significant events from the point of view of the formation of the immune system occur during the first year of life [13-15]. The origin of nAbs has been studied in model systems, but the results of these studies should be interpreted with caution, since the repertoire of animal (including mouse) nAbs differs significantly from the human one [16, 17]. In addition, other animals and humans substantially differ in the permeability of the intestinal epithelium for immunoglobulins; for example, maternal IgG of rodents, cattle, and cats enter the bloodstream of the offspring through milk. In humans, the process of IgG transfer from mother to child ends at the time of their physical separation, i.e., at the moment of birth [28].
The first months of a child’s life are characterized by the so-called physiological (in other words, natural and necessary) immunosuppression, with reduced numbers of neutrophils and proinflammatory cytokines [29, 30]. At first glance, a baby may seem defenseless, but this immunosuppression proves necessary, since it provides the possibility of forming tolerance to antigens of regular microbiota and food; it also ensures synchronous development of the components of the immune system in the course of interaction of the infant with new environmental antigens [31-33]. While humoral immunity is gradually forming, the role of protection is performed by acute phase proteins, which are capable of providing short-term nonspecific protection in the cases of injuries and infections, as well as components of the mother’s milk, such as antimicrobial peptides. The recognition of antigens by antigen-presenting cells (APCs) also has age-related characteristics. Specifically, the production of TNFα (tumor necrosis factor-alpha), IFNγ (interferon-gamma), and IL-12 (interleukin-12) is decreased in newborns, while IL-10 (interleukin-10) and IL-6 (interleukin-6) are secreted at the rate typical of normal adults [34, 35].
Glycans of microorganisms are among the antigens that are the most important for the early development of the immune system, because, in contrast to peptide antigens, they can be recognized, first, without the formation of an MHC (main histocompatibility complex) complex and second, directly by B cells, which in this case act as APCs due to the characteristic structure of the B cell receptor (BCR). At the same time, the amount of the antigen itself can influence the immune response of B cells: a small amount of glycan cannot activate the B cell, because the BCR molecule has to simultaneously bind many copies of this antigen to transmit the signal into the cell [36-39].
Antiglycan antibodies are a convenient object for studying the general principles of the formation of nAbs. The repertoire of antiglycan Abs is wide [19, 40], and a considerable part of it (almost all non-allo-antibodies) is the same in all individuals; in addition, there is a convenient tool for their identification, namely, our glycochip [41-43]. We studied the sera of healthy infants as well as their mothers and ten unrelated healthy adults. This third group was necessary for estimating the average repertoire in the population, because of the specifics of the antibody level and composition in pregnant and lactating women [22, 23].
The diversity of antiglycan IgG in the period from 3 to 12 months of age decreased (Fig. 1); this was observed for both antibodies against mammalian glycans and those against bacterial polysaccharides. The maximum diversity of IgG observed at the age of 3 months corresponded to the maternal repertoire; during the next several months, until the age of one year, their diversity was decreasing; this agreed with the literature data [44, 45]. However, there is one important contradiction. It is known [46] that the half-life of the IgG molecule does not exceed 25 days, which means that either infants' antibodies at the age of 3 months are not of maternal origin or their circulation time in children’s blood is much longer than it is believed to be. We assume that IgG transferred into the fetus can have an extended circulating time compared with conventional IgG. Indeed, according to some studies, antibodies of pregnant women have a number of specific characteristics, in particular, an altered glycosylation [47, 48], which may increase the circulation time of the immunoglobulin. By the way, the longer circulation provides protection for the period necessary for the appearance of IgM. It is also likely that the late IgM production and prolonged maternal IgG existence both maintain the formation of oral tolerance against self-antigens and commensal bacteria.
The time course of IgM changes in the period from 3 to 12 months of age was completely different than in the case of IgG; at ages of 3 and 6 months, the IgM range was still narrow (Fig. 1) compared with adults. By the age of 12 months their repertoire was extended. Since IgM in normal pregnancy cannot cross the placenta, the production of IgM in infants has not yet reach a sufficient level; therefore, according to the published data, the proportion of antibodies of this class in newborns does not exceed 6-10% of the adult level [44]. It should be noted that, in the group of breastfed (BM) children, the antibody repertoire was different from the three other groups; the repertoire of their antiglycan IgM was the least similar to the “adult” one. At first glance, this finding is unexpected, because the BM infant remains in close contact with the mother; i.e., they are under much more favorable conditions than the children from the other three groups. A possible explanation is that breast milk contains antimicrobial peptides, proteins (including immunoglobulins), and glycans (free glycans and glycoconjugates) [35, 49], which fulfill the function of antibodies, if only partly. In other words, a wide variety of antibodies under these conditions is not necessary. Then, the accelerated formation of nAbs under the conditions of artificial nutrition can be interpreted as untimely and, hence, less demanded. However, it should be noted that this difference in the repertoire is not extremely pronounced.
The maximum variety of IgM was observed in children fed with PHF, which is a mixture of partially hydrolyzed milk proteins, mostly peptides of higher sizes, which are a reservoir of antigenic determinants. These peptides may serve as mimotopes [50-54] in the process of priming of antiglycan B1 cells.
We should especially note the absence of antibodies against the LeC epitope in all of the 20 children at the age of 12 months; conversely, in practically all tested adults (~150), anti-LeC belongs to antibodies of top rank [55]. These antibodies seem to play a role in the surveillance of the appearance of tumor cells [56-58]. Why they are completely absent at early age, when they appear, and what triggers their priming, remains to be seen. At the same time, most other top-rank antiglycan nAbs, such as those against Rha, Galα1-4(3)GlcNAc, and GlcNAcα1-3Galβ1-4GlcNAcβ, are already present in children at 12 months of age (table). Moreover, the titers and the occurrence of other antibodies against tumor-associated glycans, including GalNAcα1-3GalNAcβ (Fs-2, carcinomas [59], no. 101 in Fig. 2) and Galβ1-4Galβ1-4GlcNAcβ (melanoma, no. 264 [60]), in children are higher than those of other antibodies.
In summary, the natural antibodies of children only approach the repertoire for adults by 12 months of age. It is interesting that the appearance of some antibodies, such as anti-LeC, is delayed to an older age in all studied children. This can be explained by the fact that LeC is not merely a tumor-associated, but also onco-embryonic antigen; this will need to be explored experimentally. In addition, judging by our results, the lifetime of the maternal IgG in the infant’s body seems to be substantially longer than commonly believed.
Funding. The work of N. Khasbiullina, N. Shilova, and N. Bovin was supported by grant no. 14-14-00131 of the Russian Science Foundation. The work of Yu. Knirel on isolation and characterization of bacterial polysaccharides was supported by grant no. 14-14-01042 of the Russian Science Foundation.
Acknowledgements. We are thankful to our colleague from the University of Frontera (MD, PhD Jaime Inostroza) who provided sera samples.
Conflict of interest. The authors declare no conflict of interest in financial or in any other area.
Ethical approval. The blood of healthy adult donors, children aged 3, 6, and 12 months, as well as their mothers, was obtained by a standard fence from the ulnar vein with the informed consent of the responsible persons and/or their representatives in accordance with the principles described in the Helsinki Declaration and confirmed by the decision of the Ethical Committee of the University of Frontera (Temuco, Chile).
REFERENCES
1.Mouthon, L., Lacroix-Desmazes, S., Nobrega, A.,
Barreau, C., Coutinho, A., and Kazatchkine, M. D. (1996) The
self-reactive antibody repertoire of normal human serum IgM is acquired
in early childhood and remains conserved throughout life, Scand. J.
Immunol., 44, 243-251, doi:
10.1046/j.1365-3083.1996.d01-306.x.
2.Holodick, N. E., Rodriguez-Zhrubenko, N., and
Hernandez, A. M. (2017) Defining natural antibodies, Front.
Immunol., 8, 872, doi: 10.3389/fimmu.2017.00872.
3.Boyden, S. V. (1966) Natural antibodies and the
immune response, Adv. Immunol., 5, 1-28.
4.Lutz, H. U. (2007) Homeostatic roles of naturally
occurring antibodies: an overview, J. Autoimmun., 29,
287-294, doi: 10.1016/j.jaut.2007.07.007.
5.Shoenfeld, Y., and Toubi, E. (2005) Protective
autoantibodies: role in homeostasis, clinical importance, and
therapeutic potential, Arthritis Rheum., 52,
2599-2606, doi: 10.1002/art.21252.
6.Vas, J., Gronwall, C., and Silverman, G. J. (2013)
Fundamental roles of the innate-like repertoire of natural antibodies
in immune homeostasis, Front. Immunol., 4, 4, doi:
10.3389/fimmu.2013.00004.
7.Boes, M. (2000) Role of natural and immune IgM
antibodies in immune response, Mol. Immunol., 37,
1141-1149, doi: 10.1016/S0161-5890(01)00025-6.
8.Swiatczak, B., and Cohen, I. R. (2015) Gut feelings
of safety: tolerance to the microbiota mediated by innate immune
receptors, Microbiol. Immunol., 59, 573-585, doi:
10.1111/1348-0421.12318.
9.Dowling, D. J., and Levy, O. (2014) Ontogeny of
early life immunity, Trends Immunol., 35, 299-310, doi:
10.1016/j.it.2014.04.007.
10.Basha, S., Surendran, N., and Pichichero, M.
(2014) Immune response in neonates, Expert Rev. Clin. Immunol.,
10, 1171-1184, doi: 10.1586/1744666X.2014.942288.
11.Ben-Hur, H., Gurevich, P., Elhayany, A.,
Avinoach, I., Achneider, D. F., and Zusman, U. (2005) Transport of
maternal immunoglobulins through the human placental barrier in normal
pregnancy and during inflammation, Int. J. Mol. Med.,
16, 401-407, doi: org/10.3892/ijmm.16.3.401.
12.Van de Perre, P. (2003) Transfer of antibody via
mother’s milk, Vaccine, 21, 3374-3376, doi:
10.1016/S0264-410X(03)00336-0.
13.Singh, R. K., Chang, H. W., Yan, D., Lee, K. M.,
Ucmak, D., Wong, K., Abrouk, M., Farahnik, B., Nakamura, M., Zhu, T.
H., Bhutani, T., and Liao, W. (2017) Influence of diet on the gut
microbiome and implications for human health, J. Transl. Med.,
15, 73, doi: 10.1186/s12967-017-1175-y.
14.Arrieta, M. C., Stiemsma, L. T., Amenyogbe, N.,
Brown, E. M., and Finlay, B. (2014) The intestinal microbiome in early
life: health and disease, Front. Immunol., 5, 427, doi:
10.3389/fimmu.2014.00427.
15.Francino, M. P. (2014) Early development of the
gut microbiota and immune health, Pathogens, 3,
769-790, doi: 10.3390/pathogens3030769.
16.Bello-Gil, D., Khasbiullina, N. R., Shilova, N.
V., Bovin, N. V., and Manez, R. (2017) Repertoire of BALB/c mice
natural anti-carbohydrate antibodies: mice vs. humans
difference, and otherness of individual animals, Front.
Immunol., 8, 1449, doi: 10.3389/fimmu.2017.01449.
17.Vandamme, T. F. (2014) Use of rodents as models
of human diseases, J. Pharm. Bioallied Sci., 6, 2-9, doi:
10.4103/0975-7406.124301.
18.Khasbiullina, N. R., and Bovin, N. V. (2015)
Hypotheses of the origin of natural antibodies: a
glycobiologist’s opinion, Biochemistry (Moscow),
80, 820-835, doi: 10.1134/S0006297915070032.
19.Bovin, N. V. (2017) Natural antibodies to
glycans, Biochemistry (Moscow), 78, 786-797, doi:
10.1134/S0006297913070109.
20.Dotan, N., Altstock, R. T., Schwarz, M., and
Dukler, A. (2006) Anti-glycan antibodies as biomarkers for diagnosis
and prognosis, Lupus, 15, 442-450, doi:
10.1191/0961203306lu2331oa.
21.Blixt, O., Head, S., Mondala, T., Scanlan, C.,
Huflejt, M. E., Alvarez, R., Bryan, M. C., Fazio, F., Calarese, D.,
Stevens, J., Razi, N., Stevens, D. J., Skehel, J. J., van Die, I.,
Burton, D. R., Wilson, I. A., Cummings, R., Bovin, N., Wong, C. H., and
Paulson, J. C. (2004) Printed covalent glycan array for ligand
profiling of diverse glycan binding proteins, Proc. Natl. Acad. Sci.
USA, 101, 17033-17038, doi:
10.1073/pnas.0407902101.
22.Robinson, D. P., and Klein, S. L. (2012)
Pregnancy and pregnancy-associated hormones alter immune responses and
disease pathogenesis, Horm. Behav., 62, 263-271, doi:
10.1016/j.yhbeh.2012.02.023.
23.Nair, R. R., Verma, P., and Singh, K. (2017)
Immune-endocrine crosstalk during pregnancy, Gen. Comp.
Endocrinol., 242, 18-23, doi:
10.1016/j.ygcen.2016.03.003.
24.Chugh, M., Piskarev, V., Galanina, O.,
Khasbiullina, N., Kadam, P., Shilova, N., Pazynina, N., Dobrochaeva,
K., Bhanushali, P., Kozlov, N., Tupitsyn, N., and Bovin, N. (2017)
Glycoprotein CA19.9 specific monoclonal antibodies recognize sialic
acid independent glycotope, Tumor Biol., 39, doi:
10.1177/1010428317725434.
25.Obukhova, P., Piskarev, V., Severov, V.,
Pazynina, G., Tuzikov, F., Navakouski, M., Shilova, N., and Bovin, N.
(2011) Profiling of serum antibodies with printed glycan array: room
for data misinterpretation, Glycoconj. J., 28,
501-505, doi: 10.1007/s10719-011-9355-0.
26.Simon, A. K., Hollander, G. A., and McMichael, A.
(2015) Evolution of the immune system in humans from infancy to old
age, Proc. Biol. Sci., 282, 20143085, doi:
10.1098/rspb.2014.3085.
27.Walkovich, K., and Connelly, J. A. (2016) Primary
immunodeficiency in the neonate: early diagnosis and management,
Semin. Fetal. Neonatal. Med., 21, 35-43, doi:
10.1016/j.siny.2015.12.005.
28.Borghesi, J., Mario, L. C., Rodrigues, M. N.,
Favaron, P. O., and Miglino, M. A. (2014) Immunoglobulin transport
during gestation in domestic animals and humans – a review,
Open J. Anim. Sci., 4, 323-336, doi:
10.4236/ojas.2014.45041.
29.Kollmann, T. R., Levy, O., Montgomery, R. R., and
Goriely, S. (2012) Innate immune function by Toll-like receptors:
distinct responses in newborns and the elderly, Immunity,
37, 771-783, doi: 10.1016/j.immuni.2012.10.014.
30.Erkeller-Yuksel, F. M., Deneys, V., Yuksel, B.,
Hannet, I., Hulstaert, F., Hamilton, C., Mackinnon, H., Turner Stokes,
L., Munhyeshuli, V., Vanlangendonck, F., De Bruyere, M., Bach, B. A.,
and Lydyard, P. M. (1992) Age-related changes in human blood lymphocyte
subpopulations, J. Pediatr., 120, 216-222, doi:
10.1016/S0022-3476(05)80430-5.
31.Berni Canani, R., Gilbert, J. A., and Nagler, C.
R. (2015) The role of the commensal microbiota in the regulation of
tolerance to dietary allergens, Curr. Opin. Allergy Clin.
Immunol., 15, 243-249, doi:
10.1097/ACI.0000000000000157.
32.Wu, H. J., and Wu, E. (2012) The role of gut
microbiota in immune homeostasis and autoimmunity, Gut Microbes,
3, 4-14, doi: 10.4161/gmic.19320.
33.Brandtzaeg, P. (2009) Mucosal immunity:
induction, dissemination, and effector functions,
Scand. J. Immunol., 70, 505-515, doi:
10.1111/j.1365-3083.2009.02319.x.
34.Levy, O. (2007) Innate immunity of the newborn:
basic mechanisms and clinical correlates, Nat. Rev. Immunol.,
7, 379-390, doi: 10.1038/nri2075.
35.Field, C. J. (2005) The immunological components
of human milk and their effect on immune development in infants, J.
Nutr., 135, 1-4, doi: 10.1093/jn/135.1.1.
36.Viemann, D., Schlenke, P., Hammers, H. J.,
Kirchner, H., and Kruse, A. (2000) Differential expression of the B
cell-restricted molecule CD22 on neonatal B lymphocytes
depending upon antigen stimulation, Eur. J. Immunol., 30,
550-559, doi:
10.1002/1521-4141(200002)30:2<550::AID-IMMU550>3.0.CO;2-X.
37.Meffre, E., and Salmon, J. E. (2007) Autoantibody
selection and production in early human life, J. Clin. Invest.,
117, 598-601, doi: 10.1172/JCI31578.
38.Cooper, M. D. (2015) The early history of B
cells, Nat. Rev. Immunol., 15, 191-197, doi:
10.1038/nri3801.
39.Kraus, M., Alimzhanov, M. B., Rajewsky, N., and
Rajewsky, K. (2004) Survival of resting mature B lymphocytes depends on
BCR signaling via the Ig alpha/beta heterodimer, Cell,
117, 787-800, doi: 10.1016/j.cell.2004.05.014.
40.Bovin, N., Obukhova, P., Shilova, N., Rapoport,
E., Popova, I., Navakouski, M., Unverzagt, C., Vuskovic, M., and
Huflejt, M. (2012) Repertoire of human natural anti-glycan
immunoglobulins. Do we have auto-antibodies? Biochim.
Biophys. Acta, 1820, 1373-1382, doi:
10.1016/j.bbagen.2012.02.005.
41.Jacob, F., Goldstein, D. R., Bovin, N. V.,
Pochechueva, T., Spengler, M., Caduff, R., Fink, D., Vuskovic, M. I.,
Huflejt, M. E., and Heinzelmann-Schwarz, V. (2012) Serum antiglycan
antibody detection of nonmucinous ovarian cancers by using a printed
glycan array, Int. J. Cancer, 130, 138-146, doi:
10.1002/ijc.26002.
42.Kletter, D., Singh, S., Bern, M., and Haab, B. B.
(2013) Global comparisons of lectin–glycan interactions
using a database of analyzed glycan array data, Mol. Cell.
Proteomics, 12, 1026-1035, doi:
10.1074/mcp.M112.026641.
43.Gildersleeve, J. C., Wang, B., Achilefu, S., Tu,
Z., and Xu, M. (2012) Glycan array analysis of the antigen repertoire
targeted by tumor-binding antibodies, Bioorg. Med. Chem.
Lett., 22, 6839-6843, doi: 10.1016/j.bmcl.2012.09.055.
44.Alkan Ozdemir, S., Ozer, E. A., Kose, S., Ilhan,
O., Ozturk, C., and Sutcuoglu, S. J. (2016) Reference values of serum
IgG and IgM levels in preterm and term newborns, Matern. Fetal.
Neonatal. Med., 29, 972-976, doi:
10.3109/14767058.2015.1027680.
45.Madi, A., Hecht, I., Bransburg-Zabary, S., Merbl,
Y., Pick, A., Zucker-Toledano, M., Quintana, F. J., Cohen, I. R., and
Ben-Jacob, E. (2009) Organization of the autoantibody repertoire in
healthy newborns and adults revealed by system level informatics of
antigen microarray data, Proc. Natl. Acad. Sci. USA,
106, 14484-14489, doi: 10.1073/pnas.0901528106.
46.Vidarsson, G., Dekkers, G., and Rispens, T.
(2014) IgG subclasses and allotypes: from structure to effector
functions, Front. Immunol., 5, 520, doi:
10.3389/fimmu.2014.00520.
47.Einarsdottir, H. K., Selman, M. H., Kapur, R.,
Scherjon, S., Koeleman, C. A., Deelder, A. M., van der Schoot, C. E.,
Vidarsson, G., and Wuhrer, M. (2013) Comparison of the Fc glycosylation
of fetal and maternal immunoglobulin G, Glycoconj. J.,
30, 147-157, doi: 10.1007/s10719-012-9381-6.
48.Wuhrer, M., Stam, J. C., van de Geijn, F. E.,
Koeleman, C. A., Verrips, C. T., Dolhain, R. J., Hokke, C. H., and
Deelder A. M. (2007) Glycosylation profiling of immunoglobulin G (IgG)
subclasses from human serum, Proteomics, 7,
4070-4081, doi: 10.1002/pmic.200700289.
49.Ballard, O., and Morrow, A. L. (2013) Human milk
composition: nutrients and bioactive factors, Pediatr. Clin.
North Am., 60, 49-74, doi: 10.1016/j.pcl.2012.
50.Kieber-Emmons, T., Saha, S., Pachov, A.,
Monzavi-Karbassi, B., and Murali, R. (2014) Carbohydrate-mimetic
peptides for pan anti-tumor responses, Front. Immunol.,
5, 308, doi: 10.3389/fimmu.2014.00308.
51.Ohtaki, A., Kieber-Emmons, T., and Murali, R.
(2013) Structure-based peptide mimicry of tumor-associated
antigens, Monoclon. Antib. Immunodiagn. Immunother., 32,
1-5, doi: 10.1089/mab.
52.Cusick, M. F., Libbey, J. E., and Fujinami, R. S.
(2012) Molecular mimicry as a mechanism of autoimmune disease, Clin.
Rev. Allergy Immunol., 42, 102-111, doi:
10.1007/s12016-011-8294-7.
53.Umair, S., Deng, Q., Roberts, J. M., Shaw, R. J.,
Sutherland, I. A., and Pernthaner, A. (2016) Identification of peptide
mimics of a glycan epitope on the surface of parasitic nematode larvae,
PLoS One, 11, doi:
10.1371/journal.pone.0162016.
54.Vyas, N. K., Vyas, M. N., Chervenak, M. C.,
Bundle, D. R., Pinto, B. M., and Quiocho, F. A. (2003) Structural basis
of peptide-carbohydrate mimicry in an antibody-combining
site, Proc. Natl. Acad. Sci. USA, 100, 15023-15028, doi:
0.1073/pnas.2431286100.
55.Huflejt, M. E., Vuskovic, M., Vasiliu, D., Xu,
H., Obukhova, P., Shilova, N., Tuzikov, A., Galanina, O., Arun, B., Lu,
K., and Bovin, N. (2009) Anti-carbohydrate antibodies of
normal sera: findings, surprises and challenges, Mol. Immunol.,
46, 3037-3049, doi: 10.1016/j.molimm.2009.06.010.
56.Lee, E., Pandey, N. B., and Popel, A. S. (2014)
Lymphatic endothelial cells support tumor growth in breast cancer,
Sci. Rep., 4, 5853, doi: 10.1038/srep05853.
57.Muthana, S. M., Campbell, C. T., and
Gildersleeve, J. C. (2012) Modifications of glycans: biological
significance and therapeutic opportunities, ACS Chem. Biol.,
7, 31-43, doi: 10.1021/cb2004466.
58.Zhang, S. L., Zhang, H. S., Cordon Cardo, C.,
Reuter, V. E., Singhal, A. K., Lloyd, K. O., and Livingston, P. O.
(1997) Selection of tumor antigens as targets for immune attack using
immunohistochemistry: 2. Blood group-related antigens, Int. J.
Cancer, 73, 50-56, doi:
10.1002/(SICI)1097-0215(19970926)73:1<50::AID-IJC9>3.0.CO;2-0.
59.Smorodin, E. P., Kurtenkov, O. A., Sergeyev, B.
L., Chuzmarov, V. I., and Afanasyev, V. P. (2007) The relation of serum
anti-(GalNAc-beta) and para-Forssman disaccharide IgG levels to the
progression and histological grading of gastrointestinal cancer,
Exp. Oncol., 29, 1-6.
60.Hoja-Lukowicz, D., Lenczowski, P., Carpentieri,
A., Pochec, E., Artemenko, K. A., Bergquist, J., and Litynska, A.
(2013) L1CAM from human melanoma carries a novel type of N-glycan with
Galβ1-4Galβ1-motif. Involvement of N-linked glycans in
migratory and invasive behavior of melanoma cells, Glycoconj.
J., 30, 205-225, doi: 10.1007/s10719-012-9374-5.
Supplementary Material (ZIP)