Blood–Brain Barrier Unlocked
N. Goldstein1,2*, R. Goldstein1, D. Terterov3, A. A. Kamensky1, G. I. Kovalev4, Yu. A. Zolotarev5, G. N. Avakyan6, and S. Terterov7
1Department of Human and Animal Physiology, Biological Faculty, Lomonosov Moscow State University, 119991 Moscow, Russia; E-mail: dr.naum.goldstein@googlemail.com; roman.goldstein@gmail.com; Kamensky@5.Human.bio.msu.ru2Senior Experten Service (SES), Buschstrasse 2, 53113 Bonn, Germany
3Discipline of Surgery, Faculty of Medicine, Memorial University of Newfoundland, Health Sciences Centre, 300 Prince Philip Drive, St. John’s, NL A1B 3V6, Canada; E-mail: dterterov@mun.ca
4Zakusov Institute of Pharmacology, Russian Academy of Medical Sciences, ul. Baltiiskaya 8, 125315 Moscow, Russia; E-mail: kovalev_g@m9com.ru
5Institute of Molecular Genetics, Russian Academy of Sciences, pl. Kurchatova 2, 123182 Moscow, Russia; E-mail: zolya@img.ras.ru
6Department of Neurology and Neurosurgery, Russian State Medical University, ul. Ostrovityanova 1, 117997 Moscow, Russia; E-mail: gavakyan@yandex.ru
7Department of Neurosurgery, University of California Los Angeles, 18-228 Semel, Box 957039, Los Angeles, CA, 90095-7039, USA; E-mail: STerterov@mednet.ucla.edu
* To whom correspondence should be addressed.
Received January 15, 2012; Revision received January 23, 2012
The brain is protected by a physiological blood–brain barrier (BBB) against toxins and some metabolites circulating in the blood. At the same time, the BBB limits penetration into the brain of many neuroactive drugs. Efficient ways to increase BBB permeability for delivery of drugs of different chemical nature into the brain are unknown. This work deals with delivery into the brain of 10–2 M dopamine, a substance that does not penetrate the BBB under normal circumstances. It was studied in two independent experiments: (i) penetration of 3H-labeled dopamine from its mixture with 10–5 M H2O2 into hypothalamus and striatum structures of intact rat brain, and (ii) effect of unlabeled dopamine from a mixture with H2O2 on the rat motor activity in a haloperidol catalepsy model. It was shown that (i) at the third minute after nasal application of the dopamine + H2O2 mixture, the dopamine level increases 45-fold in the hypothalamus and almost 30-fold in the striatum and (ii) motility of animals in the catalepsy haloperidol model is recovered 90 sec after intranasal introduction of dopamine. No such effects were observed after replacement of H2O2 by 0.9% NaCl solution. Thus, it was shown on the example of dopamine that its introduction into the nasal cavity simultaneously with H2O2 provides for rapid delivery of the drug into the brain. These results expand our knowledge concerning the biological role of exoROS in modulating BBB permeability and may contribute to the development of a new therapeutic strategy for neurological diseases.
KEY WORDS: blood–brain barrier, reactive oxygen species, hydrogen peroxide, 3H-labeled dopamine, haloperidol induced catalepsyDOI: 10.1134/S000629791205001X
Abbreviations: BBB, blood–brain barrier; DA, dopamine; DOPAC, 3,4-dihydroxyphenylacetic acid; endoROS, endogenous (metabolic) ROS; exoROS, exogenous ROS; GS, gaseous superoxide; HBO, hyperbaric oxygenation; HPLC, high performance liquid chromatography; MAO-A and MAO-B, monoamine oxidase A and B, respectively; MPTP, 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine; 6-OHDA, 6-hydroxydopamine; ROS, reactive oxygen species; SOD, superoxide dismutase.
The concept of superoxide radical O2•–
as an endogenous reactive oxygen species (endoROS) accompanying
processes of cell metabolism has some history. Long-term increase in
endoROS levels can be a factor responsible for oxidative stress and
many diseases including nervous system pathology. Parkinson’s and
Alzheimer’s diseases, neurotoxin effects, stroke, craniocerebral
trauma, etc. are especially important [1-3]. In biology, the opinion has long been held that
complete removal of superoxide radical is an effective tool for
preservation of the normal vital activity of the organism. This opinion
has recently changed due to the recognition that
O2•– not only introduces “bad
changes” and that “in a healthy cell there is an optimal
balance between superoxide production and its removal” [4]. Present-day concepts of the role of endoROS are
considered in [5].
Recently proofs of vitally important biological role of the gaseous superoxide O2•– (GS) in surrounding air have appeared [6]. Unlike endoROS, GS together with the product of dismutation (H2O2) is exoROS. In toxicological investigation of exoROS, no pathological changes were revealed in rodents [7].
ExoROS applied onto the nasal mucosa are able to induce in animals inhibition of monoamine oxidase A and B (MAO-A and MAO-B) activities in hypothalamus, basal ganglia, and brain stem, increase in brain dopamine activity, enhancement of neuroprotector and neurorescue effects after action of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) neurotoxin, enhancement of resistance to toxic action of hyperbaric oxygenation (HBO), and enhancement of pain-relieving effects of narcotics and non-narcotic analgesics [7-10].
The latter effect served as the basis for the supposition concerning exoROS enhancing drug substance penetration into the brain after nasal application. In particular, this is supported by significantly more pronounced effects of enteral analgesia of different pain-relieving substances in combination with GS inhalation [9]. Introduction of superoxide dismutase (SOD) into the nasal cavity simultaneously with GS inhalation significantly enhanced the GS potentiating effects, which was considered by the authors as evidence of a determining role of micromolar (10–5 M) concentration of H2O2 as the superoxide dismutation product. In fact, an analogous effect was achieved by nasal introduction of equimolar H2O2 amounts, and this completely disappeared after nasal introduction of catalase [10].
Detected physiological and therapeutic properties of H2O2 after nasal application of micromolar amounts were used with a new anti-Parkinson drug [11, 12]. Taken together, these data served as the basis for a working hypothesis concerning the ability of exoROS, mainly H2O2, to modulate permeability of blood–brain barrier (BBB) structures and facilitate transport of substances from the nasal cavity to the brain. This also follows from data of other authors who showed that permeability of BBB structures for a number of substances could be modulated by mediators of inflammation [13] and oxidative stress [14-17]. These works do not consider the problem of possible use of these factors for delivery from nasal cavity to brain of drug substances, in particular, preparations not penetrating the BBB under the usual routs of introduction into the organism.
We supposed that nasal introduction of drug preparations in a pharmaceutical mixture with micromolar H2O2 can be an efficient and practical procedure for rapid and noninvasive delivery of drug substances from the nasal cavity into the brain. Results of the first experiments confirmed the therapeutic efficiency of this method [18].
The goal of this work was to investigate the effect of micromolar H2O2 on dopamine (DA) delivery into the brain in combined introduction into the nasal cavity. The choice of DA as a test substance was due to the fact that this substance does not penetrate into the brain through the BBB in generally accepted ways of introduction into the organism (except intrathecal). Penetration of 3H-labeled DA into the hypothalamus and striatum structures was estimated by measurement of radioactivity associated with DA and its metabolite 3,4-dihydroxyphenylacetic acid (DOPAC). In the second part of this work, we estimated the physiological activity of DA that penetrated into the striatum. To do this, we studied rat motor activity in the model of haloperidol catalepsy.
MATERIALS AND METHODS
Preparation of tritium-labeled dopamine. [3H-G]Dopamine was obtained in reaction of high-temperature solid-phase catalytic isotopic hydrogen exchange for tritium [19] in dopamine hydrochloride preparation. The resulting [3H]DA preparation was purified on a Kromasil C18 column (8 × 150 mm) in a concentration gradient of aqueous acetonitrile solution in the presence of 0.1% heptafluorobutyric acid. The preparation was quantitatively analyzed by HPLC using dopamine standard (Sigma-Aldrich, Germany). The specific radioactivity of the uniformly labeled DA was 20 Ci/mol. Aqueous stock solution of the preparation contained 10–2 M DA with volume activity 0.75 mCi/ml.
Preparation and nasal introduction of dopamine solution. The mixture for nasal introduction was prepared ex tempore by fusion of [3H]DA solution (concentration 4·10–2 M, volume activity 0.75 Ci/ml) with isotonic solution of stabilized hydrogen peroxide (Sigma-Aldrich). Final concentrations in solution for nasal introduction for DA and H2O2 were 10–2 and 10–5 M, respectively. In the experimental group of animals nasal introduction of [3H]DA + H2O2 was used. Animals of control group received solution [3H]DA + 0.9% NaCl.
Preparation of samples for chromatographic analysis. Three minutes after nasal introduction of [3H]DA solution, the brain was removed and the hypothalamus and both parts of the striatum were excised and placed onto a cold (4°C) support as described earlier [20]. The isolated brain structures were weighed for 35-40 sec, frozen in liquid nitrogen, and placed in Eppendorf tubes (Eppendorf AG, Germany). Frozen samples were lyophilized for 48 h, after which they were extracted by 200 µl of solution containing 0.1 M HClO4, 100 µg/ml DA, and 100 µg/ml DOPAC (Sigma-Aldrich). Samples were centrifuged for 15 min at 10,000g, and the supernatant was used for determination of [3H]DA and [3H]DOPAC.
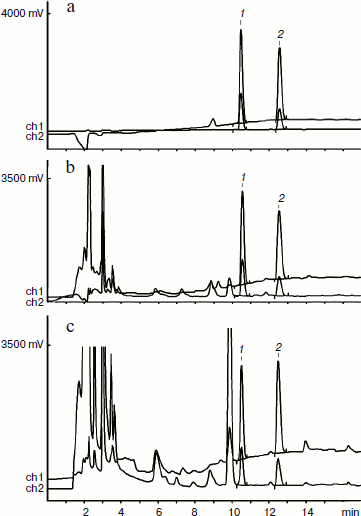
[3H]DA and [3H]DOPAC analysis by HPLC. Since final concentrations of DA and DOPAC in samples were not enough for UV detection of fractions containing radioactive derivatives of these substances against the background of blood plasma peaks, 10 µg of unlabeled DA and DOPAC standards were added to tissue extract before chromatographic separation. The extract in 0.1 M HClO4 was analyzed by chromatography on a Kromasil C18-5 µm (4 × 150 mm) column at 20°C using gradient elution by acetonitrile (4-24%) in 0.1% heptafluorobutyric acid. The eluate was simultaneously monitored at 254 and 220 nm with a Beckman model 165 (Altex) spectrophotometer, sample volume 100 µl. Fractions of brain structure extracts of each animal of experimental and control groups containing separately [3H]DA and [3H]DOPAC were analyzed quantitatively on a liquid scintillation counter. Figure 1 shows typical chromatograms of extracting solution, hypothalamus, and striatum extracts containing DOPAC and DA.
Quantitative analysis of rat hypothalamus and striatum extracts. Radioactivity bound with [3H]DA and [3H]DOPAC in chromatographic fractions of hypothalamus and striatum, corresponding to DA and DOPAC standards, was measured using a TriCarb 2900TR liquid scintillation counter (Perkin Elmer, USA). Radioactivity of extracts from each of eight rats of control and experimental groups were measured as dpm; to convert these values to µCi, the conversion factor 2.22·106 was used. Average counting efficiency was 49%. Conversion to DA amount was based on the [3H]DA stock solution concentration 10–2 M and volume activity 0.75 mCi/ml.Fig. 1. HPLC chromatograms of extracting solution containing DOPAC, DA, and brain tissue extracts. a) DOPAC (10 µg) (1) and DA (10 µg) (2) in extracting solution. b, c) Extracts of, respectively, hypothalamus and striatum of experimental animals containing DOPAC and DA standards. UV detection: ch1) 220 nm (1 A); ch2) 254 nm (0.2 A).
Haloperidol-induced catalepsy model in rats. The catalepsy state in rats was stimulated by a single intraperitoneal injection of haloperidol (Ratiopharm, Germany) at the dose of 0.25 mg/kg. After development of catalepsy determined as 85% decrease in spontaneous motor activity in the absence of reaction to external stimuli, animals of three separate groups received corresponding applications of solutions 10–2 M DA, 10–5 M H2O2, or DA + H2O2 mixture. The applied volume of solution per each nasal meatus was 50 µl. The single dose of applied DA was 0.8 mg/kg; the dose of H2O2 was 34 ng per animal. The “open field” test was used to estimate spontaneous activity of the animals. The test area was formed by a round field 80 cm in diameter with wooden floor ruled into 16 equal sectors subdivided by two concentric circles; the height of the area barrier was 40 cm. For measurement of spontaneous motor activity, each animal was placed into the center of the area and the number of horizontal transitions between sectors was registered for 2 min [21, 22]. Observations began 90 sec after application of the preparations.
Experimental animals. The work was carried out on laboratory animals in accordance with the requirements of the Institute Counsel on Ethics. Experimental results were obtained on 51 male Wistar rats of 220-250 g. The animals were kept under standard vivarium conditions with unlimited food and water. During three days before start of experiments, the rats were trained for contact with the researcher (“handling”).
Statistic analysis. Non-Gaussian distribution of radioisotope counts per minute of DA and DOPAC was supposed in the brain tissue, but due to the small sample for reliable check of distribution, the Kolmogorov–Smirnov test was inapplicable. For this reason, P values for this part of experiment were calculated using the one-sided nonparametric Mann–Whitney test. Results were considered as reliable at P < 0.05. During analysis of rat experimental catalepsy, spontaneous motor activity of animals was expressed in conditional units of visual registration, which made it possible to suppose non-Gaussian distribution of parameters.
RESULTS AND DISCUSSION
Quantitative analysis of [3H]DA and [3H]DOPAC in extracts. Radioactivity bound to [3H]DA and [3H]DOPAC and found in hypothalamus and striatum was much higher in experimental group compared to control. Calculated DA and DOPAC concentrations in hypothalamus and striatum of the experimental rats were much higher than in the control, as shown in the table.
Effect of micromolar H2O2 on nasal delivery of
dopamine into rat brain structures
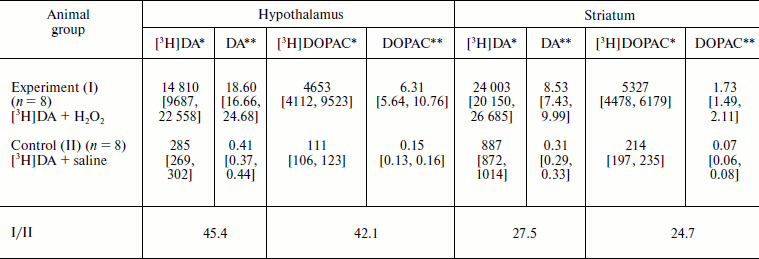
Notes: Values are given as medians [1st quartile; 3rd quartile].
P = 0.0004 (calculated using the one-sided Mann–Whitney
test). The volume of introduced solution was 2 ×
50 µl. The doses of introduced substances: DA,
0.8 mg/kg; H2O2, 34 ng.
*Counts per minute.
**pmol/mg tissue.
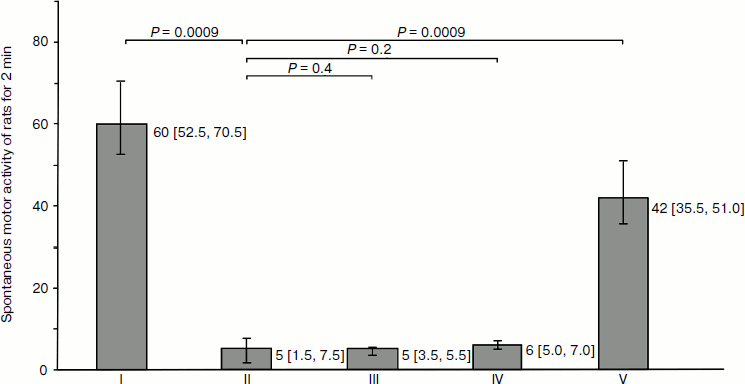
Physiological correlation. Injection of haloperidol caused significant inhibition of random activity of the animals. The latent period for development of catalepsy after intraperitoneal haloperidol injection was 9.4 [8.9; 9.8] min; the duration of catalepsy was estimated as 57.1 [54.8; 59.4] min. Nasal introduction of DA + H2O2 mixture caused during 90 sec significant recovery of spontaneous motor activity; injection of isotonic DA or H2O2 solutions in control groups did not result in recovery of motor activity in animals during the whole period of catalepsy (Fig. 2).
Drug delivery into the brain with minimal systemic side-effects is of great significance in therapy of neurological diseases such as Parkinson’s and Alzheimer’s diseases, epilepsy, migraine, depression, stroke, pain, brain tumors, etc. One of the problems in therapy of these diseases is associated with the low permeability of the BBB to most drugs. Therapeutic methods for crossing the BBB include decrease in mucociliary clearance [23], modulation of transporter expression [24], the use of anesthetics [25], ultrasound [26], etc. Recently there has been an increased number of investigations dealing with vascular permeability modulation under conditions of oxidative stress [27]. These works do not consider the use of these factors for delivery from the nasal cavity to the brain of drug substances that do not penetrate through the BBB in usual ways of introduction into the organism. Universal and safe methods for clinical drug delivery into the brain do not appear in the available literature [13, 28]. At the same time, the nasal introduction of drugs by an alternative way (through the BBB) has evident advantages in rate, dose decrease, and minimization of undesirable systemic effects.Fig. 2. Effect of nasal application of dopamine (DA) on spontaneous motor activity of rats in the model of haloperidol (HP) catalepsy. H2O2, hydrogen peroxide (10–5 M). One conventional unit corresponds to crossing of one sector in the “open field” test. Spontaneous motor activity of rats: group I, intact control; group II, after intraperitoneal introduction of HP; groups III and IV, after nasal application of DA or H2O2 isotonic solution; group V, after introduction of DA + H2O2 mixture. Indices in groups III-V were measured against the background of the effect of HP. The number of rats in each group n = 7. Animals of each group (I -V) were tested separately. Values and errors are expressed as medians [1st quartile; 3rd quartile]. P values were calculated using the one-sided Mann–Whitney test.
In this work, we investigated unlocking the BBB by nasal introduction of a composition of drug substance DA and 10–5 M H2O2. It was shown on the example of DA as a test substance that nasal introduction of H2O2 in micromolar concentration is able to rapidly switch off the protective mechanism of the BBB. Thus, only 3 min after nasal introduction, significant increase in DA and DOPAC content in the rat hypothalamus and striatum was observed, and in this case DA peaks on HPLC chromatograms of extracts completely coincide with the peak of introduced DA standard. In control animals, nasal introduction of [3H]DA in combination with physiological solution did not increase DA content in the brain structures.
It is seen in the table that introduction into the nasal cavity of DA + 10–5 M H2O2 resulted in a 45-fold increase in DA content in hypothalamus and almost 30-fold increase in striatum. Comparison with average DA and DOPAC content, respectively, of 5 and 3 pmol/mg tissue in rat hypothalamus and 70 and 7 pmol/mg in striatum [29], revealed significant difference in percent increase in DA and DOPAC levels between experimental and control animals. In the experimental group the DA and DOPAC content in hypothalamus increased, respectively, by 372.0 and 210.3%, which exceeds the corresponding increase by 8.2 and 5.0% in the control. Similarly, the DA and DOPAC content in striatum increased, respectively, by 12.2 and 24.7%, which is higher than the corresponding increase by 0.4 and 1.0% in the control. The DOPAC/DA ratio did not undergo pronounced changes and reached, respectively, in experimental and control groups 0.34 vs. 0.37 in hypothalamus and 0.20 vs. 0.23 in striatum. This shows that nasal introduction of H2O2 has no effect on DA metabolism in the studied brain structures.
Rapid development of the specific physiological activity of DA in striatum was revealed in the model of haloperidol catalepsy in rats. As shown in Fig. 2, motor activity in animals receiving only DA or only H2O2 did not change. On the contrary, the combination DA + H2O2 only 90 sec after application resulted in lowered catalepsy symptoms. These results show that DA penetrating into the striatum retains physiological activity by rapid relief of haloperidol blocking of dopamine (D2) receptors. Micromolar H2O2 undergoes rapid decomposition in the nasal mucosa and does not penetrate the brain structures.
Taking into account that H2O2 in low concentrations in vitro acts as a vasodilator and modulator of membrane permeability [17, 30], the vomeronasal organ, located at the anteroinferior part of the nasal septum [7, 31], and capillaries of the Kisselbach’s plexus can be the main points of action for the introduced H2O2 and “fenestras” from the nasal cavity into the BBB for drug substances.
Since mixture of DA and H2O2 solutions for nasal application was prepared ex tempore, formation of DA oxidation products was a fortiori excluded. It should also be taken into consideration that toxic product of DA oxidation – 6-hydroxydopamine (6-OHDA) – does not exhibit DA activity and is used in Parkinson’s disease modeling in experiments [32].
The difference in DA level increase in hypothalamus and striatum of experimental animals can be explained by time-dependent parameters of distribution of DA penetrating into the brain. However, it seems unlikely that the regions of radioactive label penetration are limited to the two studied structures. This follows from results of investigation of combined application of analgesics and nasal form of exoROS [7, 9, 10] showing that the higher subcortical centers of pain sensitivity in the thalamus can be involved in the physiological response. Different “traces” of exoROS were found in the brain stem [7] having afferent and efferent connections between thalamus, hypothalamus, limbic system, and cerebral cortex.
The described results confirm our earlier observations of prolonged action of substances introduced into the nasal cavity [18]. It can be supposed that ligands that were introduced via the nose and reached the appropriate brain receptors are temporarily retained in these structures. This can happen in presynaptic endings of DA synapses of the basal ganglia where excess DA undergoes reverse capture and is deposited in vesicles with gradual release into synapses.
Data of this work can be used to explain most physiological reactions caused by exoROS and correlate with the hypothesis about the important biological role of exoROS [7, 31], including therapeutic potential of micromolar hydrogen peroxide as a new vector for overcoming the protective BBB threshold. More complete understanding of the interaction between exoROS, nasal cavity structures, and BBB will stimulate development of new methods of drug delivery directly into the brain.
Authors are grateful to Prof. Y. A. Vladimirov for helpful discussions and comments on the manuscript and to Mr. A. S. Terterov for organization and coordination of this multicenter investigation and helpful participation in manuscript preparation.
REFERENCES
1.Halliwell, B., and Gutteridge, J. M. C. (1988)
Hum. Toxicol., 7, 7-13.
2.Cohen, G. (1994) Ann. NY Acad. Sci.,
738, 8-14.
3.McCord, J. M. (1993) Clin. Biochem.,
26, 351-357.
4.McCord, J. M. (1995) PSEBM, 209,
112-117.
5.Longo, V. D., Mitteldorf, J., and Skulachev, V. P.
(2005) Nat. Rev. Genet., 6, 866-872.
6.Goldstein, N. I., Goldstein, R. N., and Merzlyak,
M. N. (1992) Int. J. Biometeorol., 36, 118-122.
7.Goldstein, N. I. (2000) Biophysical Aspects of
the Exogenous Superoxide O2•–
Physiological Effect on Animals: Doctoral dissertation [in
Russian], Moscow State University, Moscow.
8.Arsavskis, V., Goldsteins, N., Aroncika, B.,
Konstantinova, O., and Raits, E. (1991) Latvijas Arsts,
2, 77-80 (in Latvian).
9.Goldstein, N. I., Voskresenskaya, O. G., Dubynin,
V. A., Levitskaya, N. G., and Kamensky, A. A. (2003) Byul. Eksp.
Biol. Med., 135, 253-256.
10.Goldstein, N., Baumann, S., Lewin, T., Kamensky,
A., Dubinin, V., and Konstantinova, O. (1996) Inflamm. Res.,
5, 473-478.
11.Golubev, V. L., Sadekov, R. A., Pilipocich, A.
A., and Goldstein, N. I. (2003) Lechenie Nervnykh Boleznei,
4, 26-30.
12.Goldstein, N. I., Naidin, V. L., and Fedorova, N.
V. (2002) Neurol. J. (Moscow), 6, 45-48.
13.Abbott, N. J. (2000) Cell Mol. Neurobiol.,
20, 131-147.
14.Miller, A. A., Drummond, G. R., and Sobey, C. G.
(2006) Antioxid. Redox Signal., 8, 1113-1120.
15.Kang, Y. S. (2006) Adv. Exp. Med. Biol.,
583, 291-298.
16.Faraci, F. M. (2006) J. Appl. Physiol.,
100, 739-743.
17.Lee, H. S., Namkoong, K., Kim, D. H., Kim, K. J.,
Cheong, Y. H., Kim, S. S., Lee, W. B., and Kim, K. Y. (2004)
Microvasc. Res., 68, 231-238.
18.Goldstein, N., and Goldstein, R. (2002) Patent DE
10248601.
19.Zolotarev, Yu. A., Dadayan, A. K., Borisov, Yu.
A., and Kozik, V. S. (2010) Chem. Rev., 110,
5425-5446.
20.Ehrenstrom, F., and Johansson, P. (1985) Life
Sci., 36, 867-879.
21.Levitskaya, N. G., Sebentsova, E. A., Andreeva,
L. A., Alfeeva, L. Y., Kamenskii, A. A., and Myasoedov, N. F. (2004)
Neurosci. Behav. Physiol., 34, 399-405.
22.Ashmarin, I. P., Levitskaya, N. G., Antonova, L.
V., Nezavibatko, V. N., Alfeeva, L. Yu., Dubinin, V. A., Golubovich, V.
P., Ponomareva-Stepnaya, M. A., and Kamensky, A. A. (1994) Regul.
Pept., 51, 49-54.
23.Al-Ghananeem, A. M., Malkawi, A. H., and Crooks,
P. A. (2011) Drug Dev. Ind. Pharm., 37, 329-334.
24.Haase, J., Killian, A. M., Magnani, F., and
Williams, C. (2001) Biochem. Soc. Trans., 29,
722-728.
25.Tetrault, S., Chever, O., Sik, A., and Amzica, F.
(2008) Eur. J. Neurosci., 28, 1330-1341.
26.Konofagou, E. E., Tung, Y. S., Choi, J.,
Deffieux, T., Baseri, B., and Vlachos, F. (2012) Curr. Pharm.
Biotechnol., [Epub ahead of print].
27.Neuwelt, E. A., Bauer, B., Fahlke, C., Fricker,
G., Iadecola, C., Janigro, D., Leybaert, L., Molnar, Z.,
O’Donnell, M. E., Povlishock, J. T., Saunders, N. R., Sharp, F.,
Stanimirovic, D., Watts, R. J., and Drewes, L. R. (2011) Nat. Rev.
Neurosci., 12, 169-182.
28.Su, Y., and Sinko, P. J. (2006) Expert Opin.
Drug Deliv., 3, 419-435.
29.Simansky, K. J., Bourbonais, K. A., and Smith, G.
P. (1985) Pharmacol. Biochem. Behav., 23, 253-258.
30.Lagrange, P., Romero, I. A., Minn, A., and
Revest, P. A. (1999) Free Radic. Biol. Med., 27,
667-672.
31.Goldstein, N. (2002) Biochemistry
(Moscow), 67, 161-170.
32.McCorvy, J. D., Watts, V. J., and Nichols, D. E.
(2012) Psychopharmacology (Berl.), January 6, [Epub ahead of
print].